Details of DPV and References
DPV NO: 8 June 1970
Family: Potyviridae
Genus: Potyvirus
Species: Turnip mosaic virus | Acronym: TuMV
Turnip mosaic virus
J. A. Tomlinson National Vegetable Research Station, Wellesbourne, Warwickshire, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Gardner & Kendrick (1921) and Schultz (1921).
- Selected synonyms:
- Brassica
virus 1 (Rev. appl. Mycol. 17: 52) - Selected synonyms:
- Cabbage virus A (Rev. appl. Mycol. 24: 428)
- Cabbage black ringspot virus (Rev. appl. Mycol. 14: 669)
- Cabbage black ring virus (Rev. appl. Mycol. 18: 365)
- Daikon mosaic virus (Rev. appl. Mycol. 44: 896)
- Horseradish mosaic virus (Rev. appl. Mycol. 28: 43)
- Marmor brassicae (Rev. appl. Mycol. 28: 514)
- Radish P and R viruses (Rev. appl. Mycol. 39: 643)
- Cabbage black ringspot virus (Rev. appl. Mycol. 14: 669)
- A virus with flexuous filamentous particles approximately 720 nm long, sap-transmissible to a wide range of species and transmitted by many aphid species in the non-persistent manner. World-wide distribution.
Main Diseases
Causes mottling and black necrotic spots and ringspots in cabbage, cauliflower and Brussels sprout; mosaic with leaf distortion and stunting in turnip, swede, radish, rape, mustard, Chinese cabbage, watercress and horseradish; mottles and ringspots in rhubarb; and flower-breaks in wallflower, stock, Zinnia, Tropaeolum, Petunia and Anemone.
Geographical Distribution
World-wide. Frequently reported in temperate regions of Africa, Asia, Europe and N. America.
Host Range and Symptomatology
Host range is wide, susceptible species occurring in 20 dicotyledonous families. Transmissible by sap inoculation (e.g. from infected Brassica perviridis) to the following:
- Diagnostic species
- Nicotiana tabacum
cv. White Burley (tobacco). Chlorotic local spots (5-6 days) enlarging up to 5 mm, becoming centrally necrotic, brown-rimmed and surrounded by a chlorotic halo (Fig. 5). Not systemic. - Brassica rapa (turnip). Pale chlorotic or necrotic local lesions.
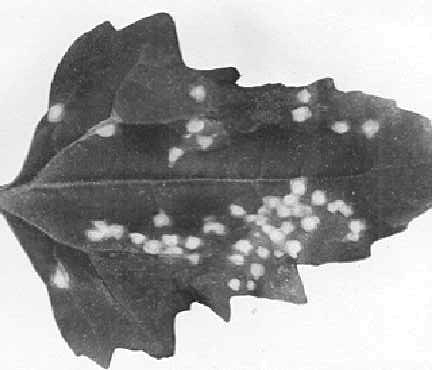
Systemic vein clearing and veinal flecking (Fig. 1) developing into severe
mosaic with light and dark green patches or blisters (Fig. 2). Severe distortion
and stunting.
- Brassica oleracea var. capitata (cabbage). Small local black necrotic rings or spots (14 days). Usually no vein clearing. Systemic black ringspots best seen on under side of leaf.
- Brassica oleracea var. botrytis (cauliflower). Diffuse systemic mottle composed of small pale green spots or rings which may look like blisters. On older leaves, black necrotic spots may appear (Fig. 4).
- Matthiola incana (stock). Systemic leaf distortion and slight marbling. Outstanding symptom is ‘flower-breaking’ i.e. appearance of white or pale-coloured sectors in coloured flowers (Fig. 6).
- Cheiranthus cheiri (wallflower). Similar to Matthiola incana: ‘broken’ flowers conspicuous, with yellow stripes and flecks in the petals of red coloured varieties.
- Chenopodium amaranticolor. Chlorotic spots 5-10 days after inoculation (with some strains) enlarging into semi-necrotic red or brown-rimmed lesions after 2-3 weeks (Fig. 7).
- Chenopodium quinoa. Chlorotic and necrotic lesions 5-10 days after inoculation (Fig. 8). Systemic veinal flecks and spots.
- Brassica oleracea var. capitata (cabbage). Small local black necrotic rings or spots (14 days). Usually no vein clearing. Systemic black ringspots best seen on under side of leaf.
- Propagation species
- Isolates can be maintained in Brassica perviridis (tendergreen
mustard), Petunia hybrida or Nicotiana glutinosa; B. perviridis, B.
pekinensis (Chinese cabbage) and B. rapa are good sources of virus
for purification.
- Assay species
- Nicotiana tabacum, Chenopodium amaranticolor, C. quinoa.
Strains
Symptom variations caused by differences in virulence between isolates have been noted in stock (Tompkins, 1939), Iceland poppy (McClean & Cowin, 1953) and cauliflower (Broadbent, 1957). Isolates failing to infect a number of Brassica species including cabbage were obtained from stock (Tompkins, 1939) and Anemone (Hollings, 1957). A cabbage isolate was reported to infect cucumber (Larson & Walker, 1941). The relationships of some of these isolates have been discussed by Pound & Walker (1945) and Sylvester (1953).
Susceptibility of certain lettuce cultivars is related to their resistance to downy mildew (Zink & Duffus, 1969).
Transmission by Vectors
Transmissible by 40-50 species of aphids, notably Myzus persicae and Brevicoryne brassicae (Kennedy, Day & Eastop, 1962). All instars can transmit. Virus can be acquired in less than 1 min and inoculated in less than 1 min (Sylvester, 1953). No latent period. Virus is retained by feeding vectors for less than 4 hr (Sylvester, 1954).
Transmission through Seed
No reports.
Transmission by Dodder
Cuscuta californica and C. campestris failed to transmit the virus from infected to healthy lettuce (Zink & Duffus, 1969).
Serology
Antisera with titres of 1/512 were prepared by Shepherd & Pound (1960) and Tomlinson & Walkey (1967) using purified virus isolates obtained respectively from cabbage and rhubarb. In precipitin tube tests, 0.85% NaCl is used as diluent. Reactions in such tests give flagellar-type precipitates. In gel-diffusion tests the virus normally gives no reaction but after ultrasonic treatment (Tomlinson et al., 1965; Tomlinson & Walkey, 1967) diffusion of fragmented antigen results in a precipitation line.
Relationships
Close relationships have been established between cabbage black ringspot, cabbage virus A and horseradish mosaic isolates (Larson, Matthews & Walker, 1950); cabbage black ringspot and anemone mosaic isolates (Hollings, 1957); and isolates from cabbage, rhubarb and statice (Niblett, Paulus & Semancik, 1969).
In plant protection tests isolates may protect against each other, as with cabbage black ring and cabbage virus A in cabbage and Solanum integrifolium ( Pound & Walker, 1945). In Petunia, Salpiglossis and Chinese cabbage, Hollings (1957) obtained no protection between cabbage black ringspot virus and anemone mosaic virus isolates.
Stability in Sap
Properties in vitro differ between isolates but, in general, the thermal inactivation point is below 62°C, the dilution end point in sap is between 10-3 and 10-4 and infectivity is retained at 20°C for 3-4 days. Infective sap kept at 2°C retains infectivity for several months.
Purification
Two methods have been used:
1. Shepherd & Pound (1960). Extract tissue in 0.5 M potassium phosphate buffer (pH 7.5) and filter. Add n-butanol to 8.0%. Clarify and sediment by differential centrifugation, resuspending pellets in 0.02 M borate buffer (pH 7.5). The preparations can be further freed from residual host components by acidification to pH 5.3 and further differential centrifugation.
2. Tomlinson (1964). Extract tissue at pH 7.5 in 0.5 M borate buffer containing 0.001 M NaEDTA and 0.1% thioglycollic acid. Add n-butanol to 8.5%. Centrifuge twice at low speed. Sediment by high-speed centrifugation and resuspend pellets in 0.05 M borate (pH 7.5). Do all steps at 0-4°C. Preparations made in phosphate buffer were less infective than those made in borate buffer (Tomlinson, 1963), probably because of virus aggregation as found by Shepherd & Pound (1960).
Properties of Particles
No accessory viral components are found by analytical ultra-centrifugation. In borate buffer the virus sediments as a single component but in phosphate buffer it produces two peaks, the faster consisting of aggregated virus (Shepherd & Pound, 1960).
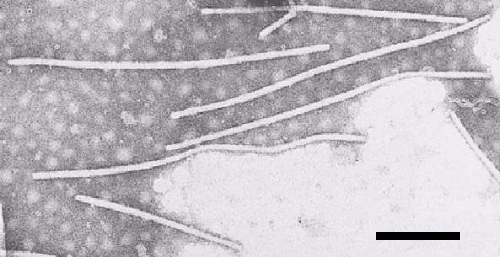
Particle Structure
The virus consists of flexuous filamentous particles (Fig. 3). The mean particle length is variously estimated as 680 nm (Shepherd & Pound, 1960), 722 nm (Tomlinson & Walkey, 1967), and 754 nm (Bode & Brandes, 1958).
Particle Composition
No reports.
Relations with Cells and Tissues
With the light microscope Stefanac & Milicic (1965) observed different types of cellular inclusions with three virus strains. In ultra-thin sections of infected Brassica perviridis, virus particles were associated with intracellular fibrous masses and band structures (Kamei, Honda & Matsui, 1969).
Notes
Symptoms of turnip mosaic virus infection in cabbage and cauliflower are sometimes confused with those caused by cauliflower mosaic virus. Cauliflower mosaic virus can usually be recognized by producing, initially, vein clearing of the youngest leaves, followed by dark green vein banding and necrotic spotting (Broadbent, 1957). Most strains of turnip mosaic virus infect many non-cruciferous species such as Nicotiana tabacum, Chenopodium amaranticolor and C. quinoa, which are not susceptible to cauliflower mosaic virus.
Figures
References list for DPV: Turnip mosaic virus (8)
- Bode & Brandes, Phytopath. Z. 34: 103, 1958.
- Broadbent, Investigation of virus diseases of Brassica crops, Cambridge Univ. Press, 1957.
- Gardner & Kendrick, J. agric. Res. 22: 123, 1921.
- Hollings, Ann. appl. Biol. 45: 44, 1957.
- Kamei, Honda & Matsui, Phytopathology 59: 139, 1969.
- Kennedy, Day & Eastop, A conspectus of aphids as vectors of plant viruses, London, Commonwealth Institute of Entomology, 1962.
- Larson & Walker, J. agric. Res. 62: 475, 1941.
- Larson, Matthews & Walker, Phytopathology 40: 955, 1950.
- McClean & Cowin, Sci. Bull. Dep. Agric. S. Afr., 332, 30 pp, 1953.
- Niblett, Paulus & Semancik, Phytopathology 59: 1166, 1969.
- Pound & Walker, J. agric. Res. 71: 255, 1945.
- Schultz, J. agric. Res. 22: 173, 1921.
- Shepherd & Pound, Phytopathology 50: 797, 1960.
- Stefanac & Milicic, Phytopath. Z. 52: 349, 1965.
- Sylvester, Phytopathology 43: 541, 1953.
- Sylvester, Hilgardia 23: 53, 1954.
- Tomlinson, Nature, Lond. 200: 93, 1963.
- Tomlinson, Ann. appl. Biol. 53: 95, 1964.
- Tomlinson, Walkey, Hughes & Watson, Nature, Lond. 207: 495, 1965.
- Tomlinson & Walkey, Virology 32: 267, 1967.
- Tompkins, J. agric. Res. 58: 63, 1939.
- Tompkins, Gardener & Thomas, J. agric. Res. 57: 929, 1938.
- Zink & Duffus, J. Am. Soc. Hort. Sci. 94: 403, 1969.