Details of DPV and References
DPV NO: 19 June 1970
Family: Bromoviridae
Genus: Ilarvirus
Species: Prune dwarf virus | Acronym: PDV
Prune dwarf virus
R. W. Fulton Department of Plant Pathology, University of Wisconsin, Madison, Wisconsin, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Thomas & Hildebrand (1936) and Moore & Keitt (1944).
- Selected synonyms
- Sour cherry yellows virus (Rev. appl. Mycol. 19: 30)
- Chlorogenus cerasae (Rev. appl. Mycol. 24: 235)
- Cherry chlorotic ringspot virus (Rev. appl. Mycol. 45: 2174)
- Peach stunt virus (Rev. appl. Mycol. 37: 91)
- Chlorogenus cerasae (Rev. appl. Mycol. 24: 235)
A virus with isometric particles about 22 nm in diameter. It is transmitted readily when sap from young leaves is used for inoculum and has a fairly wide host range. No vectors known.
Main Diseases
Causes dwarf (narrow, leathery leaves) in Italian prune (Fig. 1), Krikon and certain other plum varieties. Causes yellows in sour cherry (Fig. 2), especially when prunus necrotic ringspot virus is also present (Cropley et al., 1964). In Prunus avium the virus causes chlorotic ringspot, small etched necrotic spots, or yellow mosaic.
Geographical Distribution
Temperate regions where Prunus spp. are cultivated.
Host Range and Symptomatology
The experimental host range is fairly wide; the virus has been transmitted to species in 15 dicotyledonous families (Fulton, 1957a).
-
Diagnostic species
- Cucumis sativus
(cucumber). Small (1-2 mm) chlorotic primary lesions; systemic mosaic, which may be restricted to parts of leaves. No tip killing. - Cucurbita maxima cv. Buttercup (squash). Systemically infected leaves show
interveinal chlorotic areas, then become bright yellow
(Milbrath, 1956).
- Sesbania exaltata. Small dark local lesions in cotyledons (Fig. 3). Not systemic.
- Crotalaria spectabilis. Small dark local lesions; not systemic.
- Momordica balsamina. Chlorotic primary lesions; systemic mottle.
- Tithonia speciosa. Most isolates cause prominent chlorotic lines (Fig. 4); some do not infect.
- Prunus serrulata cv. Shirofugen (flowering cherry). Implanted virus-carrying buds cause local necrosis and gumming (Milbrath & Zeller, 1945).
- Sesbania exaltata. Small dark local lesions in cotyledons (Fig. 3). Not systemic.
-
Propagation species
- Vinca rosea, Prunus persica,
or P. mahaleb are good species for maintaining cultures. Cucurbita maxima cv. Buttercup is a good source of virus for purification.Assay species
- Sesbania exaltata
has been used for local lesion assay. Crotalaria spectabilis also appears suitable.
Strains
Many isolates differ in symptomatology and herbaceous host range. Some are more rapidly invasive in Prunus than others.
Transmission by Vectors
No vectors are known. The virus is pollen-borne in cherry and infects healthy trees, in low percentage, when they are pollinated with infected pollen (George & Davidson, 1964).
Transmission through Seed
Occurs in up to 70-80% of the seeds of Prunus species, although differentiation from prunus necrotic ringspot virus is not clear in some reports (Megahed & Moore, 1967).
Transmission by Dodder
Not tested.
Serology
The virus is moderately immunogenic. Antiserum in rabbits is produced more efficiently by intramuscular injection of virus emulsified in Freund’s incomplete adjuvant than by intravenous injections. Injections are more efficient at 3-4 day intervals than at longer intervals. The virus reacts well in agar double diffusion tests, giving a single line of precipitate, and gives a granular precipitate in liquid tests (Fulton & Hamilton, 1960).
Relationships
Chlorotic ringspot and yellow mosaic viruses of cherry are identical or closely related to prune dwarf virus (Kegler, 1965; Nemeth, 1965). It may occur with, and somewhat resembles, prunus necrotic ringspot virus, but there is no serological cross-reaction between these two viruses (Fulton & Hamilton, 1960).
Stability in Sap
In undiluted cucumber sap about half the infectivity is lost in 30 sec. In sap diluted with phosphate buffer the maximum longevity is 15-18 hr. Infectivity is stabilized in extracts by including both Na-diethyldithiocarbamate (0.01 M) and cysteine hydrochloride (0.005 M) in the extracting buffer. With infectivity chemically stabilized, thermal inactivation points (10 min) ranged from 45 to 54°C for different isolates (Waterworth & Fulton, 1964). Virus in tissue withstands rapid freezing to -78°C, but not slow freezing (Fulton, 1957b).
Purification
The method used for prunus necrotic ringspot virus (Fulton, 1968) is effective. Homogenize systemically infected squash leaves, cold, in 0.02 M, pH 8.0 phosphate buffer, 1.5 ml/g tissue. Buffer is 0.02 M with respect to 2-mercaptoethanol and Na-diethyldithiocarbamate (Hampton & Fulton, 1961). After low speed centrifugation, mix the supernatant liquid thoroughly with 0.8 vol of hydrated calcium phosphate and again centrifuge at low speed for 10-20 min. Sediment the virus by centrifuging 3 hr at 78,000 g . Resuspend pellets in 0.02 M, pH 8.0 phosphate buffer, bring to pH 4.8-5.0 with citric acid, remove precipitate by centrifugation, neutralize the supernatant liquid and concentrate the virus by high speed centrifugation. Precipitation of host protein by anti-host serum has been used for further purification.
Properties of Particles
The virus has at least two kinds of particles, sedimenting at different rates. Only the faster sedimenting particles are infective.
A260/A280: c. 1.56.
Particle Structure
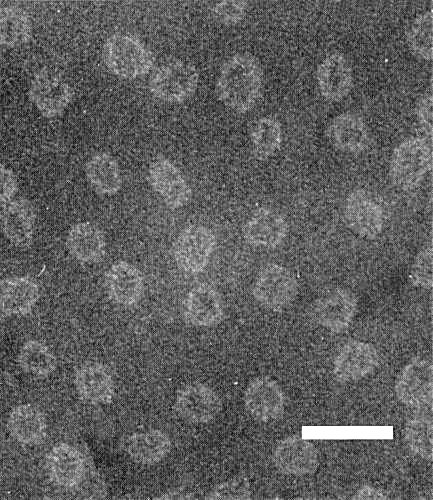
Particles are isometric, about 22 nm in diameter (Fig. 5) (Fulton, 1959).
Particle Composition
Not determined.
Relations with Cells and Tissues
No information.
Notes
Prune dwarf virus causes shock symptoms in sour cherry resembling those caused by prunus necrotic ringspot virus but they are usually milder and involve only leaves that are unfolded and partially expanded, whereas those caused by prunus necrotic ringspot virus appear in small leaves before they unfold.
Figures
References list for DPV: Prune dwarf virus (19)
- Cropley, Gilmer & Posnette, Ann. appl. Biol. 53: 325, 1964.
- Fulton, Phytopathology 47: 215, 1957a.
- Fulton, Phytopathology 47: 683, 1957b.
- Fulton, Virology 9: 522, 1959.
- Fulton, Phytopathology 58: 635, 1968.
- Fulton & Hamilton, Phytopathology 50: 635, 1960.
- George & Davidson, Can. J. Pl. Sci. 44: 383, 1964.
- Hampton & Fulton, Virology 13: 44, 1961.
- Kegler, Phytopath. Z. 54: 305, 1965.
- Megahed & Moore, Phytopathology 57: 821, 1967.
- Milbrath, Phytopathology 46: 638, 1956.
- Milbrath & Zeller, Science, N.Y. 101: 114, 1945.
- Moore & Keitt, Phytopathology 34: 1009, 1944.
- Nemeth, Zast., Bilja 16 (85-88): 441, 1965.
- Thomas & Hildebrand, Phytopathology 26: 1145, 1936.
- Waterworth & Fulton, Phytopathology 54: 1155, 1964.