Details of DPV and References
DPV NO: 32 October 1970
Family: Luteoviridae
Genus: Unassigned Luteoviridae
Species: Barley yellow dwarf virus | Acronym: BYDV
Isolates previously assigned to this virus are now classified as 6 separate viruses: BYDV-MAV, BYDV-PAV, BYDV-GPV, BYDV-SGV, CYDV-RPV, CYDV-RMV.
Barley yellow dwarf virus
W. F. Rochow Plant Pathology Dept., Cornell University, Ithaca, N.Y. 14850, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Oswald & Houston (1951, 1953a and 1953b).
- Selected synonyms
- Cereal yellow dwarf virus (Rev. appl. Mycol. 33: 74)
- Hordeumvirus nanescens (Rev. appl. Mycol. 38: 677)
- Oat red leaf virus (Rev. appl. Mycol. 36: 238)
- Hordeumvirus nanescens (Rev. appl. Mycol. 38: 677)
A virus with isometric particles c. 20-24 nm in diameter, transmitted by aphids in the persistent (circulative) manner, but not by sap inoculation. Apparently confined to the phloem. Many variants exist that differ in vector specificity, virulence, and serological properties. Distributed throughout the world, probably more widely than any other virus affecting cereals.
Main Diseases
Causes stunting and chlorosis of a wide range of monocotyledonous species, including oats, barley, wheat, and many grasses (Bruehl, 1961; Rochow, 1961; Slykhuis, 1967). Losses may be great if infection occurs early in the growing season.
Geographical Distribution
World wide.
Host Range and Symptomatology
About 100 monocotyledonous spp. are known to be susceptible. Such knowledge is limited, however, by the great variability among virus isolates, by the necessity to use feeding aphids for inoculation, and by genetic heterogeneity in many grass species. Symptoms are usually less severe in barley than in oats, and mildest of all in wheat. Some infected grasses develop no symptoms. Most infected plants are stunted (Fig. 1). Blasting of florets is common, especially in oats. Leaves may appear water-soaked and develop chlorotic stripes, blotches, or mottle, starting at the tip. Leaf chlorosis is often accompanied in barley by a brilliant yellow colour; oats may become red or purple. Some leaves may be serrated. Cool temperatures and supplementary light are often needed for development of obvious symptoms in the greenhouse.
- Diagnostic species
- Avena sativa cvs. Clintland, Clintland 64 and Blenda (spring oats).
- Avena byzantina cvs. Coast Black and California Red (winter oats).
- Hordeum vulgare cv. Black Hulless (barley).
- Avena byzantina cvs. Coast Black and California Red (winter oats).
- Propagation species
- Avena byzantina cv. Coast Black has been used as a source of virus for purification.
- Assay species
- Same as diagnostic species.
Strains
Many variants have been identified on the basis of virulence, host range, and vector specificity. Recent evidence suggests that some of the variants are so different that they should perhaps be considered as distinct viruses, for not only do they seem serologically unrelated but their interactions in mixed infections and in cross-protection tests resemble those of unrelated viruses (Aapola, 1968). Four isolates (Fig. 1) that have been studied for some years (Rochow, 1969b) are as follows:
RMV - Weakly virulent in Coast Black oats, transmitted regularly by R. maidis, but infrequently by R. padi, M. avenae, and S. graminum.
RPV - Weakly virulent in Coast Black oats, transmitted regularly by R. padi, erratically by S. graminum, but rarely by R. maidis and M. avenae.
MAV - Moderately virulent in Coast Black oats, transmitted regularly by M. avenae, but rarely by R. padi, R. maidis, and S. graminum.
PAV - Strongly virulent in Coast Black oats, transmitted regularly by R. padi and M. avenae, erratically by S. graminum, but rarely by R. maidis.
Three of these virus isolates (MAV, RPV, and PAV) have also been differentiated by serological techniques.
Gill (1969) described an additional vector-specific isolate:
SGV - Weakly virulent in Clintland 64 oats, transmitted regularly by S. graminum, but rarely if at all by M. avenae, R. padi, or R. maidis.
Transmission by Vectors
Transmissible by about 14 spp. of aphids that feed on cereals and grasses. Some of the most important vectors are Metopolophium (Acyrthosiphon) dirhodum, Macrosiphum avenae, Rhopalosiphum maidis, Rhopalosiphum padi and Schizaphis graminum. Transmission is in the persistent (circulative) manner and the virus persists in the vector for 2-3 weeks. Virus transmission to nymphs does not occur. A latent period can be shown when acquisition feeding times are shorter than 1 day (Rochow, 1963), but transmission readily occurs when acquisition and inoculation feeding times are 1-2 days (Watson & Mulligan, 1960). Transmission depends greatly on many factors, including aphid species or clone, virus isolate, temperature, test plant species, and age of source plant (Rochow, 1969a). The marked specificity between virus and vector is an important method of identifying variants of the virus.
Transmission is greatly affected by interactions among isolates of the virus (Jedlinski & Brown, 1965). Studies of mixed infections of two isolates have suggested that vector specificity is a function of the virus coat protein and that phenotypic mixing between two isolates can be an important factor in aphid transmission (Rochow, 1970).
Transmission through Seed
Not known to occur.
Transmission by Dodder
Virus has been recovered by aphids feeding on dodder established on infected barley (Orlob, Arny & Medler, 1961), and virus has been transmitted from barley to barley by Cuscuta campestris (Timian, 1964).
Serology
The virus is strongly immunogenic but, because of its small concentration in plants, concentrated, partially purified virus preparations must be used in all serological methods tested so far. Antisera against MAV, PAV, and RPV have been obtained by intramuscular and intravenous injection of rabbits (Aapola, 1968). Three serological methods have been useful. In one the infectivity is neutralized, thereby blocking transmissions by aphids that feed through membranes on treated virus preparations (Rochow & Ball, 1967; Rochow, 1970). The others involve micro agar double-diffusion and latex agglutination (Aapola, 1968; Aapola & Rochow, 1968).
Relationships
All three serological methods show a close relationship between MAV and PAV but suggest that RPV is different from the other two. The apparent unrelatedness of RPV was confirmed in cross-protection tests and other in vivo interactions (Aapola, 1968). Possible relationships with other viruses are not yet known.
Stability in Sap
MAV has been recovered from crude sap (diluted as much as 1/1000) by M. avenae feeding through membranes on sap containing added sucrose. The thermal inactivation point (10 min) for RPV and MAV both in crude and in partially purified preparations is between 65 and 70°C (Heagy & Rochow, 1965).
Purification
MAV has been purified by chloroform clarification, differential centrifugation, and density gradient centrifugation (Rochow & Brakke, 1964). Yields of MAV are usually less than 100 µg/l of juice. Purified preparations of RPV, PAV and RMV have also been obtained by the same method, but yields of virus are usually less, especially with PAV and RMV. Precipitation by polyethylene glycol (M. Wt = 6000) (Hebert, 1963) has shown promise in preliminary tests. Growth of source plants at temperatures below 20°C and thorough extraction of virus from tissue are two critical steps in purification.
Properties of Particles
Sedimentation coefficient (s20,w) is c. 115-118 S for MAV. Other isolates studied appear to have a similar coefficient.
A260/A280: c. 1.92 (MAV), 1.72 (RPV) (Brakke & Rochow, unpublished data).
Particle Structure
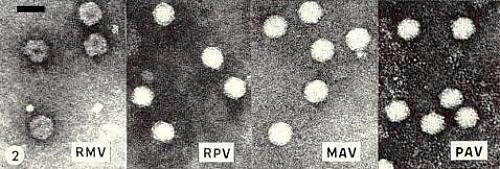
Particles are isometric; diameter c. 30 nm in shadowed preparations (Rochow & Brakke, 1964), c. 24 nm in thin sections of host tissue (Jensen, 1969), and c. 20 nm in negatively stained virus preparations (Fig. 2). Particles of four isolates appear similar (Fig. 2), although RPV is less stable than MAV in phosphotungstate at pH 6.85. The particle may be an octahedron (Israel & Rochow, unpublished).
Particle Composition
No information.
Relations with Cells and Tissues
Particles appear to be confined to the phloem (Jensen, 1969).
Notes
Because symptoms of barley yellow dwarf in the field can easily be confused with those caused by other factors, recovery of virus by aphids is an indispensable part of diagnosis. The disease especially resembles aster yellows in small grains (Banttari, 1965; Gill, Westdal & Richardson, 1969).
Much effort has been directed toward breeding varieties tolerant of the virus; use of such varieties is an important way of controlling barley yellow dwarf.
Studies on barley yellow dwarf virus have been facilitated by the relative ease with which two bioassay techniques can be applied: injection of virus into aphids and acquisition of virus by aphids feeding through membranes.
Figures

Symptoms caused by four isolates of barley yellow dwarf virus in single plants of Coast Black oats 56 days after inoculation by Rhopalosiphum maidis (RMV), R. padi (RPV and PAV) and Macrosiphum avenue (MAV). H is a comparable healthy plant originally infested with virus-free aphids as a control. Photograph by H. H. Lyon, Jr. (From Phytopathology 59: 1582, 1969).
References list for DPV: Barley yellow dwarf virus (32)
- Aapola, Ph.D. Thesis, Cornell Univ., 1968.
- Aapola & Rochow, Phytopathology 58: 398, 1968.
- Banttari, Phytopathology 55: 838, 1965.
- Bruehl, Monogr. Am. phytopath. Soc. 1, 1961.
- Gill, Can. J. Bot. 47: 1277, 1969.
- Gill, Westdal & Richardson, Phytopathology 59: 527, 1969.
- Hebert, Phytopathology 53: 362, 1963.
- Heagy & Rochow, Phytopathology 55: 809, 1965.
- Jedlinski & Brown, Virology 26: 613, 1965.
- Jensen, Virology 38: 83, 1969.
- Orlob, Arny & Medler, Phytopathology 51: 515, 1961.
- Oswald & Houston, Pl. Dis. Reptr 35: 471, 1951.
- Oswald & Houston, Phytopathology 43: 128, 1953a.
- Oswald & Houston, Phytopathology 43: 309, 1953b.
- Rochow, Adv. Agron. 13: 217, 1961.
- Rochow, Phytopathology 53: 355, 1963.
- Rochow, in Viruses, Vectors, & Vegetation, Ed. K. Maramorosch, Interscience, New York, p.175, 1969a.
- Rochow, Phytopathology 59: 1580, 1969b.
- Rochow, Science, N.Y. 167: 875, 1970.
- Rochow & Ball, Virology 33: 359, 1967.
- Rochow & Brakke, Virology 24: 310, 1964.
- Slykhuis, Rev. appl. Mycol. 46: 401, 1967.
- Timian, Phytopathology 54: 910, 1964.
- Watson & Mulligan, Ann. appl. Biol. 48: 711, 1960.