Details of DPV and References
DPV NO: 45 June 1971
Family: Alphaflexiviridae
Genus: Potexvirus
Species: Narcissus mosaic virus | Acronym: NMV
Narcissus mosaic virus
W. P. Mowat Scottish Horticultural Research Institute, Invergowrie, Dundee, Scotland
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by
van Slogteren & de Bruyn Ouboter (1946),
Cremer & van der Veken (1964)
and
Brunt (1966).
Selected synonym
- Narcissus mild mosaic virus (Rev. appl. Mycol. 45: 3028)
An RNA-containing virus with flexuous filamentous particles c. 550 nm long and 13 nm in diameter. Readily transmitted by inoculation of sap to several hosts. Prevalent in commercial stocks of many cultivars of narcissus, the only known natural host. No vector known.
Main Diseases
Causes indistinct to severe mosaic symptoms in leaves of many Narcissus cultivars of the Royal Horticultural Society Divisions I, II & IV. Effect on crop performance may be slight (Rees, 1966).
Geographical Distribution
Reported from the Netherlands and Britain, and probably occurs in all countries to which Narcissus stocks are exported.
Host Range and Symptomatology
Sap-transmissible to herbaceous species in 10 dicotyledonous families (Brunt, 1966). In most hosts, inoculated leaves are symptomless or develop inconspicuous symptoms; few hosts become systemically infected.
Diagnostic species
- Chenopodium amaranticolor.
Chlorotic local lesions (Fig. 1) becoming necrotic; infection sometimes partially systemic.- Pisum sativum (pea). Necrotic local lesions
(Fig. 2).
- Trifolium incarnatum (crimson clover). Systemic mosaic symptoms and stunting.
- Gomphrena globosa. White and/or maroon lesions in inoculated (Fig. 5) and systemically infected leaves.
- Non-hosts include Nicotiana tabacum cv. White Burley, N. glutinosa and Brassica pekinensis.
Propagation species
- High yields of virus are obtained from inoculated leaves of Chenopodium quinoa (in about 10 days) and from inoculated and systemically infected leaves of Nicotiana clevelandii (in about 4-6 weeks). N. clevelandii and Gomphrena globosa are suitable plants for maintaining cultures.
- Trifolium incarnatum (crimson clover). Systemic mosaic symptoms and stunting.
Assay species
- C. amaranticolor
and Tetragonia expansa are good local lesion hosts. For more virulent isolates (see below) C. quinoa (Fig. 7) and Phaseolus vulgaris (Fig. 6) are also suitable.
Strains
The isolate studied by Tollin et al. (1967), now called isolate M, produces more severe symptoms in some experimental hosts than does Brunt’s (1966) type isolate; isolate M causes chlorotic lesions in C. quinoa and necrotic lesions in Phaseolus vulgaris, whereas the type strain produces inconspicuous symptoms in both hosts.
Transmission by Vectors
None known; not transmitted by 4 species of aphid tested (Brunt, 1966; Mowat, 1966). It is possibly transmitted mechanically in commercial stocks of narcissus during flower harvesting.
Transmission through Seed
None found in narcissus seedlings, Gomphrena globosa or Nicotiana clevelandii (Brunt, 1966).
Transmission by Dodder
No information.
Serology
Antisera with titres of up to 1/4096 are readily prepared. Flagellar precipitates form in tube tests. Sap from virus-infected test plants or purified preparations of the virus often react with antisera in gel-diffusion tests.
Relationships
There was only a slight difference between the type isolate and isolate M in serological tests using an antiserum to isolate M (Mowat, unpublished). Properties and particle morphology place the virus in the potato virus X group, but no serological relationship was found to any of seven other viruses in this group using antisera of moderate titre (Brunt, 1966).
Stability in Sap
Sap from infected Gomphrena globosa lost infectivity after dilution to 10-5-10-6 or heating for 10 min at 75°C, but was still infective after storage for 12 weeks at 18°C, or 36 weeks at 0-4°C, and after freezing or freeze-drying (Brunt, 1966).
Purification
Two methods have been used:
1. Brunt (1966): a modification of the n-butanol method of Tomlinson et al.
2. Tollin et al. (1967): sap clarified by addition of an equal volume of diethyl ether followed by several cycles of differential centrifugation. Inoculated leaves of C. quinoa may yield about 500 mg virus per kg leaf tissue; virus readily disperses in water, or at pH 7 in 0.07 M phosphate buffer or Tris-EDTA buffer. Preparations are strongly birefringent.
Properties of Particles
Sedimentation coefficient (s20,w) at infinite dilution: 114 S. No accessory viral components are found by analytical centrifugation.
A260/A280: 1.2.
Particle Structure
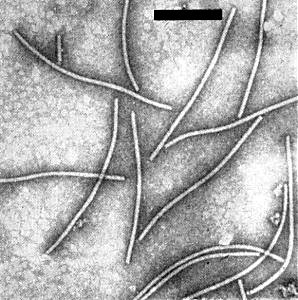
Helically constructed flexuous particles c. 550 nm long and 13 nm diameter (Fig. 8). The pitch of the helix, measured by X-ray diffraction, ranges from 3.3 to 3.6 nm depending on the water content of the specimen. True repeat is in five turns of the helix. Cross-banding is found by electron microscopy, but only in specimens stained with uranyl formate (Tollin et al., 1967; Tollin, Wilson & Young, 1968).
Particle Composition
No information.
Relations with Cells and Tissues
Spindle-shaped inclusions are formed in epidermal cells of leaves of Chenopodium quinoa, Nicotiana clevelandii and Gomphrena globosa (Fig. 3, Fig. 4) (A. Z. Stefanac & W. P. Mowat, unpublished).
Notes
Narcissus mosaic virus is the only 550 nm filamentous virus known to occur in Narcissus. The same name has sometimes been used as a synonym for the aphid-transmitted virus, narcissus yellow stripe, which has filamentous particles c. 750 nm long (see Brunt, 1966).
Acknowledgements
Photographs: courtesy of Scottish Horticultural Research Institute.
Figures
References list for DPV: Narcissus mosaic virus (45)
- Brunt, Ann. appl. Biol. 58: 13, 1966.
- Cremer & van der Veken, Neth. J. Pl. Path. 72: 105, 1964.
- Mowat, Rep. Scott. hort. Res. Inst. for 1964 & 1965: 51, 1966.
- Rees, Ann. appl. Biol. 58: 25, 1966.
- Tollin, Wilson, Young, Cathro & Mowat, J. molec. Biol. 26: 353, 1967.
- Tollin, Wilson & Young, J. molec. Biol. 34: 189, 1968.
- van Slogteren & de Bruyn Ouboter, Daffodil Tulip Yb. 12: 3, 1946.