Details of DPV and References
DPV NO: 59 June 1971
Family: Potyviridae
Genus: Potyvirus
Species: Cocksfoot streak virus | Acronym: CSV
Cocksfoot streak virus
P. L. Catherall Welsh Plant Breeding Station, Aberystwyth, Wales
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Storey (1952) and Smith (1952).
- Synonym
- Cocksfoot mosaic virus (Rev. appl. Mycol. 37: 363)
- A virus with flexuous filamentous particles c. 752 x 13 nm infecting only a few species of Gramineae. It is transmitted by several species of aphid in the non-persistent manner and by inoculation of sap. Common in Britain and Europe.
Main Diseases
Causes mild or severe streaking and decreases tillering of cocksfoot grass. The virus is usually eliminated by inter-plant competition in grazed cocksfoot swards, but is frequent in cocksfoot seed crops.
Geographical Distribution
Widespread in Britain and Europe (Peto, 1955; Slykhuis, 1958; Klinkowski & Kreutzberg, 1958; Mühle & Schumann, 1959).
Host Range and Symptomatology
Host range is restricted to a few species of Gramineae. Transmissible by inoculation of sap, for example to the following:
- Diagnostic species
- Dactylis glomerata
(cocksfoot) and other Dactylis spp. Conspicuous longitudinal leaf streaks of dark and light green or yellow (Fig. 1). Infected plants are rarely stunted, but produce fewer vegetative tillers. They flower earlier than healthy plants, but produce fewer fertile seeds (Catherall & Griffiths, 1966). - Lolium multiflorum (Italian ryegrass) and L. perenne (perennial
ryegrass). Indistinct pale green or yellow streaks (Fig. 2).
- Paspalum membranaceum and Setaria macrostachia. Very conspicuous yellow streaks (Ohmann-Kreutzberg, 1963).
- Phalaris paradoxa and Lamarkia aurea. Yellow spots.
- Anthoxanthum aristatum, Avena strigosa, Bromus mollis, Festuca capillata, Hordeum murinum, Lagurus ovatus and Setaria viridis. Symptomless infection.
- Paspalum membranaceum and Setaria macrostachia. Very conspicuous yellow streaks (Ohmann-Kreutzberg, 1963).
- Propagation species
- Cocksfoot is suitable for maintaining cultures and for propagating virus for
purification.
- Assay species
- No known local lesion hosts. Cocksfoot and Paspalum membranaceum have been used as test plants in host range studies and for testing aphids in vector studies. Ohmann-Kreutzberg (1963) found that Paspalum membranaceum was the more reliable because the incubation period of the virus in this species was shorter (14 days) and less variable than in cocksfoot (30-50 days).
Strains
No variants have been distinguished.
Transmission by Vectors
Transmissible by several species of aphids including Myzus persicae, Macrosiphum euphorbiae, Macrosiphum avenae, Metopolophium dirhodum and Hyalopteroides humilis (Smith, 1952; Watson & Mulligan, 1960). Virus can be acquired in 10 sec and inoculated in less than 1 min. No latent period. Feeding vectors cease to transmit within 1 hr after acquisition.
Transmission through Seed
Not seed-transmitted.
Transmission by Dodder
Not tested.
Serology
In double diffusion tests in l% agar partially purified virus preparations form a single band of precipitate with antisera prepared by intravenous injection.
Relationships
Serological tests have failed to show any relationship between cocksfoot streak and two viruses with particles of similar shape and size, namely ryegrass mosaic virus (Mulligan, 1960) and anthoxanthum mosaic virus (Catherall, unpublished).
Stability in Sap
Infectivity of cocksfoot extracts in water or neutral 0.1 M phosphate buffer is low. Transmission is greatly increased if a slightly acid (pH 5.9) phosphate buffer is used. In extracts of cocksfoot in pH 5.9 phosphate buffer, the thermal inactivation point (10 min) is about 55°C, dilution end-point about 3 x 10-3, and infectivity is retained at 20°C for up to 16 days (Ohmann-Kreutzberg, 1963).
Purification
No detailed information. Differential centrifugation (5000 g for 15 min, 50,000 g for 2 hr) has been used to prepare partially pure preparations of the virus for use as immunogen.
Properties of Particles
No information.
Particle Structure
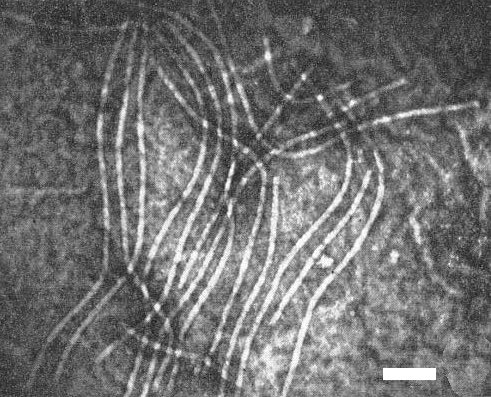
Particles are flexuous filaments c. 752 x 13 nm (Brandes, 1959) (Fig. 3).
Particle Composition
No information.
Relations with Cells and Tissues
No information.
Notes
A virus with flexuous filamentous particles causing streaking of cocksfoot and ryegrass in Germany was called cocksfoot streak virus (Schumann, 1969) although no serological tests or insect transmissions were made to substantiate this conclusion. The virus was easily transmissible to many species of Gramineae, including Avena sativa, Festuca pratensis and Triticum aestivum, which are immune to cocksfoot streak virus in Britain.
McKinney (1956) described a virus, orchard grass mosaic, causing streaking of cocksfoot in USA. Its vector and serological relationship with cocksfoot streak virus are not known, but unlike cocksfoot streak it infects Avena sativa, causing blue-green dwarfing.
Several viruses that differ serologically from cocksfoot streak virus can cause streaking of cocksfoot. Ryegrass mosaic virus (Mulligan, 1960) is the most difficult to distinguish from cocksfoot streak. It is easily transmissible by inoculation of sap to many varieties of cocksfoot, but whether it occurs naturally in cocksfoot has not been established with certainty. It has flexuous filamentous particles c. 703 x 19 nm (Brandes, 1964), but is transmitted by eriophyid mites and not by aphids, and is transmissible to Avena sativa and Festuca pratensis. Cocksfoot mottle (Serjeant, 1964) and cocksfoot mild mosaic viruses (Huth, 1968) sometimes occur in combination with cocksfoot streak virus, but both have isometric particles c. 25-30 nm in diameter. Cocksfoot mottle virus is transmissible by beetles to Triticum aestivum and cocksfoot mild mosaic virus by aphids to Setaria italica (immune to cocksfoot streak virus). Two other isometric viruses, brome mosaic (McKinney, l944), which occurs in North America, and the related ryegrass streak (Proll & Richter, 1965), which occurs in Europe, infect cocksfoot rarely. Both cause local lesions in Datura stramonium and there is an unconfirmed report that ryegrass streak virus is transmitted by nematodes.
Figures
References list for DPV: Cocksfoot streak virus (59)
- Brandes, Phytopath. Z. 35: 205, 1959.
- Brandes, Mitt. biol. BundAnst. Ld- u. Forstw. 110, 130 pp., 1964.
- Catherall & Griffiths, Ann. appl. Biol. 57: 141, 1966.
- Huth, Phytopath. Z. 62: 300, 1968.
- Klinkowski & Kreutzberg, Phytopath. Z. 32: 1, 1958.
- McKinney, Phytopathology 34: 993, 1944.
- McKinney, Pl. Dis. Reptr 40: 524, 1956.
- Mühle & Schumann, Phytopath. Z. 36: 314, 1959.
- Mulligan, Ann. appl. Biol. 48: 575, 1960.
- Ohmann-Kreutzberg, Phytopath. Z. 47: 113, 1963.
- Peto, J. Br. Grassld. Soc. 10:193, 1955.
- Proll & Richter, Naturwissenschaften 52: 145, 1965.
- Schumann, Arch. PflSchutz 5: 318, 1969.
- Serjeant, Pl. Path. 13: 23, 1964.
- Slykhuis, Pl. Prot. Bull. FAO. 6: 129, 1958.
- Smith, Pl. Path. 1: 118, 1952.
- Storey, Pl. Path. 1: 101, 1952.
- Watson & Mulligan, Rep. Rothamsted exp. Stn, 1959: 101, 1960.