Details of DPV and References
DPV NO: 99 October 1972
Family: Rhabdoviridae
Genus: Cytorhabdovirus
Species: Wheat American striate mosaic virus | Acronym: WASMV
American wheat striate mosaic virus
R. C. Sinha Chemistry and Biology Research Institute, Canada Department of Agriculture, Research Branch, Ottawa, Ontario KIA 0C6, Canada
R. M. Behki Chemistry and Biology Research Institute, Canada Department of Agriculture, Research Branch, Ottawa, Ontario KIA 0C6, Canada
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Slykhuis (1953) and Lee (1964).
An RNA-containing virus with bacilliform or bullet-shaped particles, mostly 200-250 x 75 nm. The particles also contain protein, lipid and probably some carbohydrate. The virus has a narrow host range, is transmitted by two species of leafhopper in the persistent manner and occurs in some parts of North America. Not transmissible by inoculation of sap.
Main Diseases
The virus is one of several that cause striate mosaic of wheat. Incidence of the disease in Canada was not above 1% in any wheat fields examined (Slykhuis, 1962), but in USA up to 25% infection was observed in some fields (Timian, 1960). High temperatures during early summer favour vector transmission and are a prerequisite for severe epidemics of the disease in spring wheat (Slykhuis & Sherwood, 1964).
Geographical Distribution
May be indigenous in S. Dakota; found occasionally in other parts of central USA and Canada (Slykhuis, 1963; Hamilton, 1964).
Host Range and Symptomatology
Host range is narrow; about 20 species in the family Gramineae are susceptible to infection, the severity of symptoms often depending on the variety (Slykhuis, 1962, 1963). Readily transmitted by the leafhopper Endria inimica, for example to the following:
-
Diagnostic species
- Triticum durum
(wheat) cv. Ramsey (Fig. 1) and Avena sativa (oat) cv. Victory. Obvious leaf striation (yellow to white parallel streaks); chlorosis, stunting and necrosis of older leaves. -
Eragrostis cilianensis (stink grass) cv. Lutati and Panicum capillare
(witch grass). Leaf striation and chlorosis.
- Hordeum vulgare (barley) cv. Vantage and Zea mays (corn) cv. Gaspe Flint. Slight to moderate leaf striation and stunting.
-
Propagation and assay species
- Triticum durum
cv. Ramsey is suitable for assaying transmission by leafhoppers (systemic infections) and for maintaining cultures. It is also a good source of virus for purification.
Strains
None reported.
Transmission by Vectors
Transmissible by the leafhoppers, Endria inimica and Elymana virescens. Endria inimica (Fig. 2) is an efficient vector. All instars can acquire and inoculate the virus. Few insects can acquire the virus in less than 1 min; minimum and maximum incubation periods in leafhoppers are about 4 and 24 days, respectively. Virus can be inoculated in about 15 min by some viruliferous insects. Most insects continue to transmit virus for several weeks but some cease to do so after 5 weeks. Not transmitted to progeny insects (Slykhuis, 1963). Leafhoppers readily become infective after injection with the virus. Virus multiplies in the vector, probably in several tissues (Sinha & Chiykowski, 1967, 1969). Elymana virescens is an inefficient vector; the minimum incubation period is about 15 days (Sinha, 1970).
Transmission through Seed
Probably none or rare. Infected wheat plants of highly susceptible varieties produce shrivelled seeds or none at all.
Transmission by Dodder
Attempts to transmit the virus using Cuscuta campestris were unsuccessful (Timian, 1964).
Serology
The virus is weakly immunogenic. Antiserum prepared by intramuscular injection using partially purified preparations had titres of 1/160, 1/40, and 1/5 in ring-precipitin, tube-precipitin and gel-diffusion tests, respectively. Specific precipitates of the virus in serological tests in liquids are floccular (Sinha, 1968). Antiserum obtained by injecting about 4 mg of purified virus weekly for 8 weeks had a titre of 1/320 in the ring-precipitin test. Increasing the dose of virus to 8 mg did not increase the titre (R. C. Sinha, unpublished).
Relationships
Relationships with other viruses have not been studied. Other viruses that cause superficially similar diseases in wheat and are transmitted by leafhoppers (Jassidae) are Russian winter wheat mosaic (bacilliform particles, 260 ± 20 x 60 ± 5 nm; Razvyazkina & Polyakova, 1967) and Australian wheat striate mosaic virus (Grylls, 1963; particle morphology unknown). European wheat striate mosaic virus (Slykhuis & Watson, 1958), also with unknown particle morphology, causes similar symptoms in wheat; however, it is transmitted by a planthopper (Delphacidae), not by Endria inimica, the main leafhopper vector of American wheat striate mosaic virus (Slykhuis, 1963).
Stability in Sap
Properties were determined by injecting extracts of viruliferous leafhoppers into virus free insects and then testing the latter for ability to transmit. The thermal inactivation point (10 min) is about 55°C, dilution end-point 10-5, and infectivity is retained at 4 or -10°C for about 3 days (Lee & Bell, 1962). Infective virus can be preserved in leaves by freezing them in liquid nitrogen.
Purification
The following method seems satisfactory (Ahmad, Sinha & Hochster, 1970). Infected leaves yield about 4 mg virus per 100 g leaf tissue.
Extract the juice from leaves that have been infiltrated under vacuum with 0.1 M MgCl2 and 0.3 M glycine solution (pH 8.0). Centrifuge at low speed. Adjust pH of the supernatant fluid to 8.0 and centrifuge at low speed again. Add 5% activated charcoal to the supernatant fluid and stir for 1 min, then add 5% Celite and stir for another minute. Pass the mixture through a Celite pad (0.5 inch thick) and collect 20-ml fractions. Pool those that are opalescent and centrifuge for 90 min at 15,000 g. Resuspend the pellet in MgCl2-glycine solution (pH 7.0) and pass through a column of 2% agarose beads. Fractions containing virus are pooled and further clarified by centrifugation in rate zonal (20-40% sucrose) and quasi-equalibrium (30-60% sucrose) density gradients. Sediment the visible virus zone and clarify by two cycles of high and low speed centrifugation, resuspending the pellets obtained at high speeds in the MgCl2-glycine solution (pH 7.0). Zone electrophoresis in sucrose gradients can be used to free partially purified virus from host materials. Work at 0-4°C.
Preparations purified in this way are highly infective but recovery of infectivity is only 5-10% (R. C. Sinha, unpublished).
Properties of Particles
(R. C. Sinha, R. M. Behki & V. R. Harwalker, unpublished).
Sedimentation coefficient (s20,w) of the main component: about 900 S; preparations also contained two minor components, one of which had a higher and the other a lower s value than the main component.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 3.1.
A260/A280: 1.25.
Particle Structure
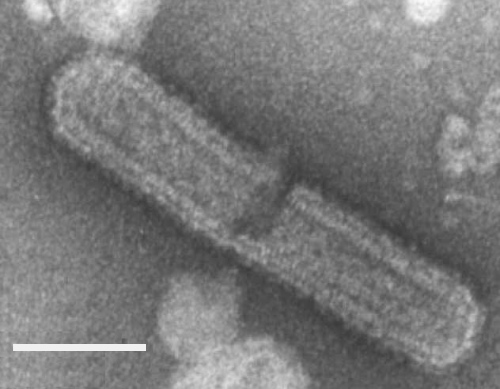
The virus particles were at first considered to be rod-shaped (Lee, 1964) and later bacilliform (Lee, 1967). Actually three types of membrane-bounded particles can be recognised both in situ and in preparations from wheat plants (Sinha, 1971): (1) bacilliform, 240-250 x 75 nm; (2) bullet-shaped, 200-215 x 75 nm; and (3) long bacilliform 405-415 x 75 nm. The first two types are most common (Fig. 3). The particles easily break during extraction from tissues or when mounted in phosphotungstate for electron microscopy unless previously fixed with glutaraldehyde (Fig. 4). The particles show distinct transverse striations and evenly spaced bead-like projections on the outer membrane.
Particle Composition
(R. C. Sinha & R. M. Behki, unpublished):
RNA: About 5% of particle weight, probably single-stranded. Approximate molar percentages of nucleotides: G27; A20; C24; U29.
Protein: About 68% of particle weight. Amino acid composition (moles %): ala 9.2; arg 3.0; asp 10.3; cys 1.6; glu 9.5; gly 7.6; his 1.1; ile 5.5; leu 7.4; lys 5.2; met 2.8; phe 4.1; pro 5.2; ser 8.3; thr 7.8; trp 2.2 (assumed); tyr 4.1; val 6.5.
Other components: About 24% lipid (neutral lipids and polar lipids - both with and without phosphate); 3% of the particle weight is suspected to be carbohydrate.
Relations with Cells and Tissues
The particles are present in all parts of infected plants, namely leaves, sheaths, culms, awns and roots (Sinha, 1971). They are found both in the cytoplasm and in the perinuclear space of parenchyma and phloem cells (Fig. 5). The virus particles could not be found in extracts of viruliferous Endria inimica or in thin sections of their various tissues. Infectivity bioassays show that virus is present in about the same concentration (per g tissue) in plants as in leafhoppers (R. C. Sinha, unpublished).
Notes
It is difficult to differentiate this disease from other mosaic diseases of wheat by symptoms alone. Fine chlorotic parallel streaks caused in susceptible plants distinguish it from some virus diseases of cereals but not from wheat striate mosaic diseases described from Europe and Australia, nor from winter wheat mosaic from Russia. It is probably safe to assume that the European virus transmitted by delphacids is distinct, because no virus has been shown to be transmitted both by delphacids and by jassids. The Australian virus, transmitted by the leafhopper Nesoclutha obscura (Grylls, 1963), differs from the American one in host range, but neither has been tested for transmission by the vector of the other (Slykhuis, 1967). Speculation about whether the two are related is probably premature until the Australian virus is characterized morphologically. The only other jassid-transmitted cereal virus with bacilliform particles is Russian winter wheat mosaic, which is transmitted by a different species, Psammotettix striatus (Zazhurilo & Sitnikova, 1941). However, this does not exclude the possibility of relationship, because a single virus can be transmitted by several different leafhopper species. American wheat striate mosaic virus belongs to the rhabdovirus group (sensu Howatson, 1970).
Figures
References list for DPV: American wheat striate mosaic virus (99)
- Ahmad, Sinha & Hochster, Virology 41: 768, 1970.
- Grylls, Aust. J. agric. Res. 14: 143, 1963.
- Hamilton, Pl. Dis. Reptr 48: 68, 1964.
- Howatson, Adv. Virus Res. 16: 196, 1970.
- Lee, Virology 23: 145, 1964.
- Lee, Virology 33: 84, 1967.
- Lee & Bell, Can. J. Bot. 41: 767, 1962.
- Razvyazkina & Polyakova, Dokl. Akad. Nauk SSSR 174: 1435, 1967.
- Sinha, Phytopathology 58: 452, 1968.
- Sinha, Can. Pl. Dis. Surv. 50: 118, 1970.
- Sinha, Virology 44: 342, 1971.
- Sinha & Chiykowski, Virology 32: 402, 1967.
- Sinha & Chiykowski, Virology 38: 679, 1969.
- Slykhuis, Phytopathology 43: 537, 1953.
- Slykhuis, Can. Pl. Dis. Surv. 42: 135, 1962.
- Slykhuis, Can. J. Bot. 41: 1171, 1963.
- Slykhuis, Rev. appl. Mycol. 46: 401, 1967.
- Slykhuis & Sherwood, Can. J. Bot. 42: 1123, 1964.
- Slykhuis & Watson, Ann. appl. Biol. 46: 542, 1958.
- Timian, Pl. Dis. Reptr 44: 771, 1960.
- Timian, Phytopathology 54: 910, 1964.
- Zazhurilo & Sitnikova, Dokl. Akad. SSSR 6: 27, 1941.