Details of DPV and References
DPV NO: 1 June 1970
Family: Bromoviridae
Genus: Cucumovirus
Species: Cucumber mosaic virus | Acronym: CMV
There is a more recent description of this virus: DPV 213
Cucumber mosaic virus
A. J. Gibbs Rothamsted Experimental Station, Harpenden, Hertfordshire, England
B. D. Harrison Scottish Horticultural Research Institute, Invergowrie, Dundee, Scotland
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Doolittle (1916) and Jagger (1916).
- Selected synonyms
- Cucumber virus 1 (Rev. appl. Mycol. 6: 501)
- Cucumis virus 1 (Rev. appl. Mycol. 17: 52)
- Marmor cucumeris (Rev. appl. Mycol. 28: 514)
- Spinach blight virus (J. agric. Res. 14: 1)
- Tomato fern leaf virus (Rev. appl. Mycol. 9: 417)
An RNA-containing virus with isometric particles about 30 nm in diameter. It has a wide host range, is transmitted by many species of aphid in the non-persistent manner and is readily transmissible by inoculation of sap. It is widespread and common in temperate regions.
Main Diseases
Causes mosaic of cucumber and other cucurbits, blight of spinach, fern leaf of tomato, mosaic of celery, woodiness of passion fruit (in part), mosaics of many dicotyledonous and monocotyledonous ornamental species; also occurs in many other crop species.
Geographical Distribution
World-wide, especially in temperate regions.
Host Range and Symptomatology
Host range is wide; species in more than 40 dicotyledonous and monocotyledonous families are susceptible to infection (Price, 1940). Readily transmissible by inoculation of sap, for example to the following:
-
Diagnostic species
- Cucumis sativus
(cucumber). Green or yellow/green systemic mosaic (Fig. 1). - Nicotiana tabacum (tobacco;
Fig. 3 and
Fig. 4),
N. glutinosa and N. clevelandii.
Inoculated leaves are symptomless, or develop chlorotic or necrotic lesions;
green or yellow/green
systemic mosaic or ringspots, usually without necrosis.
- Lycopersicon esculentum (tomato). Systemic mosaic and much narrowed leaf laminae (fern leaf, Fig. 6).
- Phaseolus vulgaris (French bean). In U.K., pin-point necrotic local lesions develop in winter but not in summer; not systemic (Fig. 5).
- Chenopodium amaranticolor and C. quinoa. Chlorotic or necrotic local lesions; not systemic. Vigna sinensis (cowpea). Local lesions in some cultivars; most isolates not systemic.
- Lycopersicon esculentum (tomato). Systemic mosaic and much narrowed leaf laminae (fern leaf, Fig. 6).
-
Propagation species
- Nicotiana glutinosa
or N. tabacum cv. Xanthi-nc are suitable plants for maintaining cultures; N. tabacum, N. clevelandii and Cucurbita pepo are good sources of virus for purification.Assay species
- Vigna sinensis, Phaseolus vulgaris, Chenopodium amaranticolor
and C. quinoa are useful local-lesion hosts. Some strains cause necrotic local lesions in Nicotiana tabacum.
Strains
Many minor variants can be distinguished. The best known major variants are:
Yellow strain of Price (1934) = strain 6 of Price. Produces brilliant yellow mosaics in Nicotiana spp. and necrotic lesions in inoculated leaves of Zinnia elegans. Source USA.
Y strain> of Price (1934). Symptoms produced in Nicotiana spp. resemble those of the yellow strain but are usually less intense. Systemic in V. sinensis. Source USA.
Spinach strain of Bhargava (1951). Necrotic local lesions in N. tabacum; systemic green mosaic or ringspots with distortion and some necrosis of the veins. Severe systemic symptoms in several species. Source UK.
Transmission by Vectors
Transmissible by more than 60 spp. of aphids, notably Aphis gossypii and Myzus persicae (Kennedy, Day & Eastop, 1962). All instars can transmit. Virus can be acquired in less than 1 min and inoculated in less than 1 min. No latent period. Feeding vectors retain virus for less than 4 h. Not transmitted to progeny aphids. Some isolates can lose their transmissibility by one aphid species but retain their transmissibility by another (Badami, 1958).
Transmission through Seed
Probably not common. Reported for instance in White Acre pea, Vigna sesquipedalis and V. sinensis (4-18%) (Anderson, 1957).
Transmission by Dodder
At least 10 species of Cuscuta can transmit. The virus infects the dodder (Schmelzer, 1956).
Serology
The virus is poorly immunogenic. It is precipitated by 0.15 M (0.85%) NaCl (Francki et al., 1966), which should not be used as a diluent in tube precipitin tests; 0.05 M phosphate buffer (pH 8), although not ideal, can be used. Agarose (1%), prepared in water, is satisfactory for gel-diffusion tests. In tests with virus protein, 1.5 M KCl should be used as the diluent in tube precipitin tests, and for preparing agarose. Specific precipitates of virus in serological tests in liquids are granular (somatic). In gel-diffusion tests two bands of precipitate may develop when antisera prepared by intramuscular injection are used: a curved band near the antigen well (intact virus) and a straighter one nearer to the antiserum well (degraded virus). Antisera prepared by intravenous injection may react only with the slower diffusing antigen (Scott, 1968).
Relationships
Using narrow-spectrum antisera, the strains of cucumber mosaic virus fall into two broad groups. The first group contains Price’s yellow and Y strains, and the second contains most of the virus strains isolated from crop and garden plants in the U.K. When broad-spectrum antisera are used, serological relationships can be shown between some viruses of the two groups. Some strains of the second group are distantly serologically related to strains of tomato aspermy virus from chrysanthemum and tomato (Brunt, Hollings and Stone, personal communication; Lawson, 1967).
In plant-protection tests, strains protect against the effects of one another either completely or partially. Price’s yellow strain (necrotic lesions in Zinnia elegans) and the spinach strain (necrotic lesions in tobacco) are often used as the challenge viruses. Some workers have found evidence of protection between cucumber mosaic virus and tomato aspermy virus but several others have not.
Stability in Sap
In tobacco sap, the thermal inactivation point (10 min) is about 70°C, dilution end-point about l0-4, and infectivity is retained at 20°C for 3-6 days. Infectivity is greatly stabilised by adding reducing agents (for example 0.1% thioglycollic acid) or chelating agents (for example 0.01 M sodium diethyl dithiocarbamate) to the leaf extraction medium.
Purification
Two methods seem satisfactory. Inoculated tobacco leaves may yield 100-400 mg virus per kg
leaf
tissue.
1. Scott (1963).
Grind tissue in 0.5 M citrate buffer (pH 6.5; containing 0.1% thioglycollic acid)
together with an equal volume of chloroform. Dialyse aqueous phase against 0.005 M borate
buffer (pH 9).
Sediment and clarify by three cycles of high- and low-speed centrifugation, resuspending
the pellets
obtained at high speed in the borate buffer. Work at 0-4°C.
2. Murant (1965).
Extract tissue in 0.5 M phosphate buffer (pH 7.5) containing 0.1% thioglycollic acid.
Filter, and shake filtrate with an equal volume of ether. Centrifuge at low speed.
Sediment and clarify
by two cycles of high- and low-speed centrifugation, resuspending the pellets obtained at
high speed in
0.06 M phosphate buffer (pH 7.5). Centrifuge at low speed, adjust supernatant fluid to
pH 5 and
centrifuge again at low speed. Resuspend pellet in 0.06 M buffer and centrifuge at
low speed. Work at
0-4°C.
Zone electrophoresis can be used to free partially purified virus from host materials (Van Regenmortel, 1966).
Properties of Particles
Sedimentation coefficient (s20, w) at infinite dilution: about 98 S.
Molecular weight: 5.8-6.7 x 106 daltons.
Diffusion coefficient (D20 x 10-7cm2/sec): 1.23.
Isoelectric point: about pH 4.7.
Partial specific volume (calculated): 0.701.
Electrophoretic mobility: -8 x 10-5 cm2/sec/volt at pH 7 in 0.1 M buffer (S strain); other strains probably differ.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 5.0.
A260/A280: 1.65.
Particle Structure
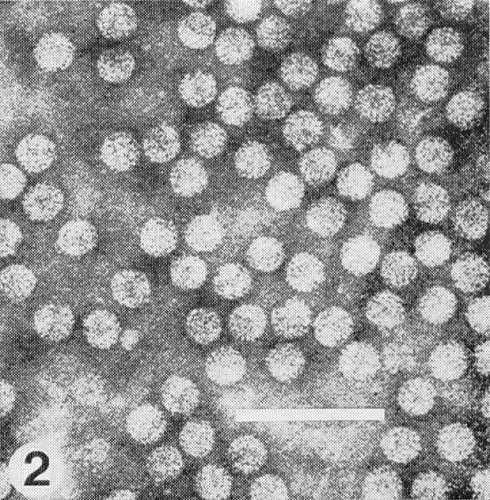
Particles are isometric, about 30 nm in diameter (Fig. 2) and are built of 180 subunits in pentamer-hexamer clusters; particle centre is hollow (Finch et al., 1967). The particles of most strains disintegrate when mounted in phosphotungstate for electron microscopy, unless previously fixed (for instance, 2% formaldehyde for 30 min). The particles are penetrated by phosphotungstate and contrast poorly.
Particle Composition
RNA: Molecular weight about 1 x 106, about 18% of particle weight, probably single stranded. Molar percentages of nucleotides: G23.4; A24.3; C23.2; U29.1. Sedimentation coefficient of RNA (s20, w) is 23 S in 0.02 M KH2PO4 - NaOH buffer of pH 7.2, but subsidiary components of 13 S and 20 S reported (Kaper et al., 1965).
Protein: Subunits have molecular weight about 3.2 x 104, and contain about 287 amino acid residues. Amino acid composition (moles %): ala 6.0; arg 8.4; asx 10.4; cys 0; glu 7.1; gly 5.6; his 1.4; ile 5.5; leu 9.2; lys 6.3; met 2.7; phe 2.5; pro 6.4; ser 11.1; thr 6.1; trp 0; tyr 3.8; val 7.6 (Van Regenmortel, 1967).
Other components: None reported.
Relations with Cells and Tissues
All tissues infected except, possibly, meristematic regions. No inclusion bodies found. Virus-like particles occur in cytoplasm.
Notes
Tomato aspermy virus has a host range and properties similar to those of cucumber mosaic virus. However, it causes more distortion of systemically infected leaves of Nicotiana glutinosa, and enations develop. Unlike cucumber mosaic virus it is confined to inoculated leaves of Cucumis sativus and Datura stramonium. It occurs commonly in chrysanthemum, in which cucumber mosaic virus is uncommon (Hollings, 1955).
Alfalfa mosaic virus may be confused with cucumber mosaic virus, because it too is widespread, causes similar symptoms in some hosts and is also transmitted by aphids in the non-persistent manner. However, most strains of alfalfa mosaic virus, unlike cucumber mosaic virus, give local lesions in Phaseolus vulgaris at all times of the year, and infect Chenopodium amaranticolor and C. quinoa systemically. Also, alfalfa mosaic virus has distinctive bacilliform particles.
Figures
References list for DPV: Cucumber mosaic virus (1)
- Anderson, Phytopathology 47: 515, 1957.
- Badami, Ann. appl. Biol. 46: 554, 1958.
- Bhargava, Ann. appl. Biol. 38: 377, 1951.
- Doolittle, Phytopathology 6: 145, 1916.
- Finch, Klug & Van Regenmortel, J. mol. Biol. 24: 303, 1967.
- Francki, Randles, Chambers & Wilson, Virology 28: 729,1966.
- Hollings, Ann. appl. Biol. 43: 86, 1955.
- Jagger, Phytopathology 6: 148, 1916.
- Kaper, Diener & Scott, Virology 27: 54, 1965.
- Kennedy, Day & Eastop, A conspectus of aphids as vectors of plant viruses. London, Commonwealth Institute of Entomology, 1962.
- Lawson, Virology 32: 357, 1967.
- Murant, Virology 26: 538, 1965.
- Price, Phytopathology 24: 743, 1934.
- Price, Amer. J. Bot. 27: 530, 1940.
- Schmelzer, Phytopath. Z. 28: 1, 1956.
- Scott, Virology 20: 103, 1963.
- Scott, Virology, 34: 79, 1968.
- Van Regenmortel, Virology 23: 495, 1966.
- Van Regenmortel, Virology 31: 391, 1967.