Details of DPV and References
DPV NO: 110 October 1972
Family: Betaflexiviridae
Genus: Carlavirus
Species: Chrysanthemum virus B | Acronym: CVB
Chrysanthemum virus B
M. Hollings Glasshouse Crops Research Institute, Littlehampton, Sussex, England
Olwen M. Stone Glasshouse Crops Research Institute, Littlehampton, Sussex, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Noordam (1952).
- Chrysanthemum mild mosaic virus (Rev. appl. Mycol. 37: 238)
- A virus with rather straight rod-shaped particles about 685 nm long. It has a limited host range, is transmitted by aphids in the non-persistent manner and by inoculation of sap. Widespread throughout the world in cultivated varieties of chrysanthemum.
Main Diseases
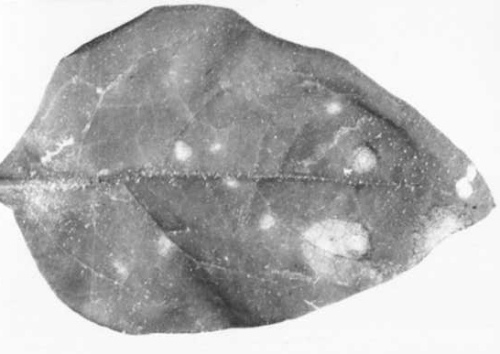
Causes very mild leaf-mottling or vein-clearing (Fig. 5) in some cultivars; some infected varieties show slight loss of flower quality compared with virus-free plants, and a few varieties sometimes develop brown necrotic streaks (Fig. 4) on the florets (Brierley, 1955). Many cultivars are entirely infected, often without visible symptoms.
Geographical Distribution
Widespread wherever chrysanthemums are grown.
Host Range and Symptomatology
Host range narrow; infects chrysanthemum and about 10 other species in 5 dicotyledonous families (Hollings, 1957). Transmitted with difficulty under long day conditions of the N.W. European summer.
- Diagnostic species
- Petunia hybrida.
Local yellow spots after 2-5 weeks; some isolates from chrysanthemum induce necrotic or green spots or yellow rings (Fig. 1, Fig. 2, Fig. 3). Symptomless systemic infection occurs rarely. - Nicotiana glutinosa. Local chlorotic spots after 2-3.5 weeks; no
systemic infection.
- N. clevelandii. Very mild systemic mottle and vein-clearing after 3 weeks (Fig. 7); high concentrations of virus may be produced.
- Tetragonia expansa. Diffuse local chlorotic spots up to 3 mm diameter after 2.5-4 weeks; no systemic infection.
- N. clevelandii. Very mild systemic mottle and vein-clearing after 3 weeks (Fig. 7); high concentrations of virus may be produced.
- Propagation species
- Nicotiana clevelandii
is suitable for maintaining cultures and as a source of virus for purification.- Assay species
- Petunia hybrida
has been used as an assay host but best results are obtained by serology and electron microscopy (Hakkaart, 1969).
Strains
Hollings (1957) distinguished three different strains, based on difference of reaction in P. hybrida (Fig. 1, Fig. 2, Fig. 3). The strains have not been compared serologically.
Transmission by Vectors
Transmitted in the non-persistent manner by the aphids Myzus persicae, Macrosiphum euphorbiae, Aulacorthum solani, Coloradoa rufomaculata, and Macrosiphoniella sanborni (Hollings, 1957). The virus was acquired in 2-5 min. Feeding vectors retained infectivity for 30-45 min and non-feeding aphids for up to 1 hr. Frequency of transmission was increased by starving the aphids for 3 hr before the acquisition feed.
Transmission through Seed
None reported.
Transmission by Dodder
Not recorded.
Serology
The virus is a good immunogen and, following injections of freshly prepared virus preparations, serum titres up to 1/250,000 can be obtained in precipitin tube tests. The precipitates are flocculent (flagellar). Micro-precipitin and ring tests can also be used satisfactorily, but not double diffusion in agar gel.
Relationships
Serologically distantly related to carnation latent virus, potato virus S and potato virus M (Hakkaart, van Slogteren & de Vos, 1962); all these viruses are placed in the carnation latent virus (carlavirus) group (Brandes, 1964; Harrison et al., 1971).
Stability in Sap
The thermal inactivation point (10 min) is between 70 and 75°C in chrysanthemum sap, between 75 and 80°C in P. hybrida sap; the difference is probably caused by differences in virus concentration. The dilution end-point ranges between 10-2 and 10-4. Infectivity is lost after 2-6 days at 20°C. Lyophilized sap of Nicotiana clevelandii stored under vacuum at 20°C retained infectivity for at least 18 months.
Purification
1. Oertel (1969). Harvest Nicotiana clevelandii whole plants 3 weeks after inoculation. Grind in medium containing 0.2% sodium sulphite and 0.2% ascorbic acid (wt:vol = 1:1). Centrifuge at 2500 g for 15 min. Shake supernatant fluid with an equal volume diethyl ether for 2 min. Separate and discard the ether phase. Shake the aqueous phase with an equal volume of carbon tetrachloride for 2 min then centrifuge at 2500 g for 15 min. Retain the aqueous phase and centrifuge at 5600 g for 15 min. Sediment the virus by centrifuging for 2 hr at 54,000 g. Resuspend pellet in 0.01 M phosphate buffer pH 7.5, and remove insoluble matter by brief centrifugation.
2. Hollings et al. (1970). Harvest N. clevelandii whole plants 3-4 weeks after inoculation. Homogenize each 100 g in 200 ml 0.05 M phosphate buffer (pH 7.6) containing 0.1% thioglycollic acid, and let the slurry stand c. 24 hr at 2°C. Express the juice, add n-butanol to 8.5% (v/v) and shake the mixture 1 hr. Separate the virus by one or more cycles of differential centrifugation (10,000 g for 25 min; 80,000 g for 90 min). Resuspend the final pellets in 0.03 M phosphate buffer and, after 3-4 hr, remove insoluble material by brief centrifugation (10,000 g).
Preparations made by Oertel’s method have higher infectivity but are less pure, and give non-specific precipitates in saline (Hollings & Stone, unpublished).
Properties of Particles
Sedimentation coefficient (s20,w) at infinite dilution: about 168 S.
A260/A280: 1.55.
Amax(260)/Amin(280): 1.20.
Particle Structure
Particles in leaf extracts are rather straight rods c. 685 nm long and 12 nm wide, when stained in neutral phosphotungstate (Fig. 6, Fig. 8) with cucumber green mottle mosaic virus as internal standard. Purified preparations had particles of similar dimensions (Hollings et al., 1970). Particles have a straighter, less flexuous appearance than carnation latent virus, and tend to fragment in phosphotungstate in a characteristic manner (Fig. 6).
Particle Composition
Nucleic acid: No information, but by analogy with serologically related viruses, probably RNA.
Protein: No information.
Relations with Cells and Tissues
In leaves of varieties showing mottle, the chlorotic areas have smaller palisade cells and fewer chloroplasts than usual. No intracellular inclusions have been detected (Hollings, 1957). The virus is present in all parts of the plant though in the summer months it may not be readily detectable.
Notes
Chrysanthemum plants can be freed from the virus by heat treatment or meristem-tip culture, and very easily by a combination of both techniques (Hakkaart & Quak, 1964; Hollings & Stone, 1968; Oertel, 1969).
Although chrysanthemum virus B is serologically related to carnation latent virus and potato virus S and potato virus M, the host ranges do not overlap. Symptoms in the chrysanthemum varieties Good News and Mistletoe readily distinguish virus B from other filamentous viruses of chrysanthemum, such as chrysanthemum vein mottle. Whereas the latter cause severe mottle, sometimes with distortion and dwarfing, in 6-10 weeks, plants infected with virus B are usually almost symptomless (Hollings, 1957). Identification of virus B is best done by electron microscopy or, in the winter months, by serology (Hakkaart, 1969).
Figures
References list for DPV: Chrysanthemum virus B (110)
- Brandes, Mitt. biol. Bund Anst. Ld- u. Forstw. Berlin-Dahlem 110, 130 pp., 1964.
- Brierley, Phytopathology 45: 2, 1955.
- Hakkaart, Neth. J. Pl. Path. 75: 355, 1969.
- Hakkaart & Quak, Neth. J. Pl. Path. 70: 154, 1964.
- Hakkaart, van Slogteren & de Vos, Tijdschr. PlZiekt. 63: 126, 1962.
- Harrison, Finch, Gibbs, Hollings, Shepherd, Valenta & Wetter, Virology 45: 356, 1971.
- Hollings, Ann. appl. Biol. 45: 589, 1957.
- Hollings & Stone, Rep. Glasshouse Crops Res. Inst. 1967: 106, 1968.
- Hollings, Stone, Thorne & Atkey, Rep. Glasshouse Crops Res. Inst. 1969: 128, 1970.
- Noordam, Tijdschr. PlZiekt. 58: 121, 1952.
- Oertel, Nova Acta Leopoldina 34: 69, 1969.