Details of DPV and References
DPV NO: 113 July 1973
Family: Tymoviridae
Genus: Tymovirus
Species: Scrophularia mottle virus | Acronym: ScrMV
Scrophularia mottle virus
R. Bercks Biologische Bundesanstalt für Land- und Forstwirschaft, Braunschweig, Germany
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Hein (1959) and
Bercks et al. (1971).
Synonym
- Scrophularia-Scheckungsvirus (Rev. appl. Mycol. 51: 1241)
-
An RNA-containing virus with isometric particles about 25-27 nm in diameter. It has a rather wide host range, is transmitted by beetles of the genus Cionus and is readily transmissible by inoculation of sap. Found in Germany.
Main Diseases
Causes variable amounts of mottling in Scrophularia nodosa (Fig. 1) sometimes with vein-clearing.
Geographical Distribution
So far known only in Germany; possibly more widely distributed.
Host Range and Symptomatology
Scrophularia nodosa is the only natural host known. Species in 7 out of 19 dicotyledonous families develop systemic symptoms. Species in 9 families develop local or latent infections. Transmissible by inoculation of sap.
-
Diagnostic species
- Datura stramonium.
Inoculated leaves develop circular bright yellow spots, which tend to fuse (Fig. 3); systemically infected leaves show light green spots along the veins, or mosaic. - Vicia faba. Local lesions or brown dots or rings
(Fig. 4, right), sometimes with
irregular and poorly defined outlines (Fig. 4, left).
- Chenopodium quinoa. Inoculated leaves show irregularly distributed bright yellow dots (Fig. 5). Mild mosaic on systemically infected leaves.
- Antirrhinum majus. Systemically infected leaves develop light green spots (Fig. 6).
- Chenopodium quinoa. Inoculated leaves show irregularly distributed bright yellow dots (Fig. 5). Mild mosaic on systemically infected leaves.
-
Propagation species
- Antirrhinum majus
and Datura stramonium are good sources of virus for purification.Assay species
- Vicia faba
can be used as a local lesion host.
Strains
No information. Isolates differ in symptom expression on Scrophularia nodosa (Fig. 1) and on herbaceous test plants.
Transmission by Vectors
The vectors, which transmit the virus only from Scrophularia to Scrophularia, are beetles belonging to the family Curculionidae (Cionus tuberculosis, C. scrophulariae, C. hortulanus, C. alauda) and unidentified larvae of Cionus spp. About 20% of the individuals of these species transmit the virus (Weidemann, 1972, and unpublished).
Transmission through Seed
No information.
Transmission by Dodder
No information.
Serology
The virus is strongly immunogenic in rabbits; antisera with titres of 1/1000-1/2000 in agar gel diffusion tests are readily obtained. The virus produces one line of precipitate.
Relationships
The virus belongs to the tymovirus group and has serological relationships with Andean potato latent, belladonna mottle, dulcamara mottle, eggplant mosaic, ononis yellow mosaic and turnip yellow mosaic viruses. The closest serological relationship is to ononis yellow mosaic virus. However, Bercks et al. (1971) detected no serological relationship between ononis yellow mosaic virus and turnip yellow mosaic virus, and R. Koenig (unpublished) found only a very distant one. Scrophularia mottle virus has many other properties in common with the viruses of the tymovirus group.
Stability in Sap
The virus is inactivated when heated to 92-94°C for 10 min (Hein, 1959). In sap from infected Datura stramonium, the virus loses infectivity when diluted 10-5 (assayed on D. stramonium) or 10-6 (assayed on Antirrhinum majus); inactivation at room temperature takes about 30 days when D. stramonium is used as a test plant (R. Bercks, unpublished).
Purification
The virus is easily purified by Steere’s butanol-chloroform method from A. majus or D. stramonium whole plants harvested 4-6 weeks after inoculation. Preparations free of ribonuclease are obtained using the bentonite method as recommended for turnip yellow mosaic virus by Dunn & Hitchborn (1965).
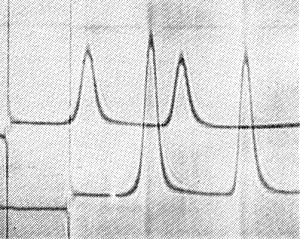
Properties of Particles
Virus from D. stramonium sediments as two components, empty protein shells (T) and nucleoprotein (B) (Fig. 7). Sedimentation coefficients (s20, w) at infinite dilution (svedbergs): 54 (T), 116 (B). Preparations from Antirrhinum contain three additional components sedimenting at 85, 169 and 200 S. The nature of these components is unknown but the 169 S component possibly consists of dimers of the B component.
Partial specific volumes (calculated): 0.74 ml/g (T), 0.69 ml/g (B).
Electrophoretic behaviour: in immunoelectrophoresis in 1% agarose containing 0.05 M phosphate buffer, pH 7, the virus migrates slowly towards the cathode.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): about 8 for the normal mixture of T and B components (H. L. Paul, unpublished).
A260/A280: 1.79; corrected and uncorrected values give the same results for the normal mixture of T and B components from D. stramonium.
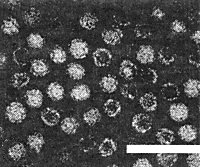
Particle Structure
The particles are isometric, c. 26 nm in diameter, usually with poorly defined hexagonal outlines (Fig. 2). The empty shells are penetrated by stains used for electron microscopy. In structure the virus apparently resembles other tymoviruses.
Particle Composition
RNA: Single-stranded, M. Wt 2.13 x 106. Molar percentages of nucleotides: G15.9±0.3; A21.3±0.2; C33.7±0.3; U29.3±0.3 (W. Huth, unpublished). RNA is about 37% of particle weight.
Protein: The subunits have M. Wt 21,600. The amino acid composition is (moles percent): ala 6.4; arg 3.8; asx 6.8; cys ?; glx 7.8; gly 5.8; his 1.7; ile 6.8; leu 8.5; lys 3.7; met 2.2; phe 2.5; pro 10.8; ser 13.3; thr 10.2; try 0.8; tyr 1.7; val 7.7 (H. L. Paul & B. Wittmann, unpublished).
Relations with Cells and Tissues
No information.
Notes
Scrophularia mottle virus and ononis yellow mosaic virus (Gibbs et al., 1966) are closely related serologically but the former is the more closely related to turnip yellow mosaic virus. During electrophoresis at pH 7 scrophularia mottle virus migrates towards the cathode whereas ononis yellow mosaic virus migrates towards the anode. The two viruses also differ in host range and thermal inactivation point (65°C for ononis yellow mosaic virus).
Acknowledgements
Photographs: courtesy of H. Schlobach, Braunschweig.
Figures
References list for DPV: Scrophularia mottle virus (113)
- Bercks, Huth, Koenig, Lesemann, Paul & Querfurth, Phytopath. Z. 71: 341, 1971.
- Dunn & Hitchborn, Virology 25: 171, 1965.
- Gibbs, Hecht-Poinar, Woods & McKee, J. gen. Microbiol. 44: 177, 1966.
- Hein, Phytopath. Z. 36: 290, 1959.
- Weidemann, Jber. biol. BundAnst. Land- u. Forstw. Braunschweig, 1971: 70, 1972.