Details of DPV and References
DPV NO: 117 July 1973
Family: Potyviridae
Genus: Potyvirus
Species: Hippeastrum mosaic virus | Acronym: HiMV
Hippeastrum mosaic virus
A. A. Brunt Glasshouse Crops Research Institute, Littlehampton, Sussex, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Disease described by Kunkel (1922); virus partially characterized by Brants
& Van den Heuvel (1965) and Brunt (1973).
- Selected synonym
- Amaryllis mosaic virus (Rev. appl. Mycol. 30: 403).
- A virus with flexuous filamentous particles c. 750 x 12 nm transmitted by aphids after brief acquisition and inoculation feeds. Only known to occur naturally in members of the Amaryllidaceae, but transmissible by mechanical inoculation of sap to a few species in three other families. World-wide distribution
Main Diseases
Causes mosaic diseases in Hippeastrum equestre and cultivated species and hybrids of Hippeastrum. It occurs also in Crinum, Hymenocallis, Isomene, Phaedranassa and Urceolina (Brunt, 1973), and possibly several related genera (Hannibal, 1942).
Geographical Distribution
Probably world-wide; reported from Asia (Bremer, 1926; Iwaki, 1967; Procenko & Procenko, 1964), Africa (P. J. Klesser, personal communication), Europe (Brants & Van den Heuvel, 1965) and America (Kunkel, 1922; Holmes, 1928).
Host Range and Symptomatology
Only known to occur naturally in members of the Amaryllidaceae, but will also infect the inoculated leaves only of a few species in Amaranthaceae, Chenopodiaceae and Solanaceae.
- Diagnostic species
- Hippeastrum hybridum. Irregular light and dark green mosaic patterns
on leaves and flower stalks, especially conspicuous in intolerant cultivars
(Fig. 1). Flowers are usually symptomless.
- Chenopodium murale. Fawn necrotic local lesions c. 2 mm in diameter after 3 weeks (Fig. 5). Not systemic.
- Chenopodium quinoa. Chlorotic or necrotic circular local lesions (Fig. 2) c. 2 mm in diameter after 2-3 weeks (Brunt, 1973). Not systemic.
- Gomphrena globosa. Few red circular necrotic local lesions (Fig. 4) 2-4 mm in diameter, sometimes after c. 10 days (Brants & Van den Heuvel, 1965) but often after 3-4 weeks. Not systemic. G. globosa is much less susceptible than other local lesion hosts, and is a very poor source of inoculum.
- Hyoscyamus niger. Numerous circular fawn or chlorotic local lesions (Fig. 3) c. 2 mm after 7 days (De Leeuw, 1972b). Not systemic.
- Nicotiana clevelandii. Numerous white etched rings 1-2 mm in diameter after 3-4 weeks (Fig. 6), later becoming diffuse and faintly chlorotic (Brunt, 1973). Not systemic.
- Tetragonia expansa. Circular chlorotic local lesions 1-2 mm in diameter after c. 3 weeks. Not systemic.
- Chenopodium murale. Fawn necrotic local lesions c. 2 mm in diameter after 3 weeks (Fig. 5). Not systemic.
- Propagation species
- Hippeastrum hybridum is a suitable host for maintaining the virus, but
inoculated leaves of Nicotiana clevelandii are a better source of virus for
purification.
- Assay species
- Chenopodium quinoa and Hyoscyamus niger are good hosts for local lesion assays, and Hippeastrum hybridum seedlings for aphid transmission tests.
Strains
None reported.
Transmission by Vectors
Transmitted readily after brief (2-10 min) acquisition and inoculation feeds by Aphis gossypii and Myzus persicae, but not Aphis craccivora or Macrosiphoniella sanborni (Brunt, 1973). No latent period.
Transmission through Seed
Not seed-borne in Hippeastrum hybridum.
Transmission by Dodder
No information.
Serology
The virus is a good immunogen; antisera with titres of 1/2000 have been produced in rabbits after one intravenous injection of c. 0.2 mg virus followed 11 and 19 days later by intramuscular injections of similar amounts of virus emulsified with Freund’s complete adjuvant. Antibodies and antigen react in tube precipitin tests to produce flagellar precipitates.
Relationships
The virus has properties typical of members of the potato virus Y (potyvirus) group, but is serologically unrelated to carnation vein mottle, turnip mosaic, tobacco severe etch, iris mild mosaic, iris severe mosaic, pepper veinal mottle, clover yellow vein, tobacco veinal necrosis, bean yellow mosaic, lettuce mosaic, pea mosaic and narcissus yellow stripe viruses (Brunt, 1973).
Stability in Sap
In Hippeastrum hybridum leaf extracts, infectivity is lost after dilution to 10-2-10-3, and after 10 min at 55-60°C, 1-4 days at 20°C or 16-32 days at 2°C (Brants & Van den Heuvel, 1965; Brunt, 1973).
Purification
Infected Hippeastrum hybridum leaves have a high virus content but also contain mucilaginous substances that hinder purification; Nicotiana clevelandii is a better source of virus, yielding up to 10 mg/kg leaf tissue by the following procedure (Brunt, unpublished). Leaves with numerous local lesions 3-4 weeks after inoculation are extracted (1 g/4 ml) in 0.1 M sodium citrate containing 0.05 M disodium ethylenediaminetetraacetate and 0.2% 2-mercaptoethanol (pH 7.2), and the fluid subjected to one cycle of differential centrifugation; high speed pellets are dispersed in 0.033 M phosphate buffer (pH 7.6), stirred for c. 30 min with 25% (v/v) carbon tetrachloride, virus sedimented from the aqueous phase by centrifugation at 75,000 g for 90 min, and then redispersed in 0.03 M phosphate buffer (pH 7.6). Further purification is achieved by centrifuging for 75 min at 78,000 g in 10-40% sucrose gradient columns; extracted zones are dialysed for up to 48 hr against 0.03 M phosphate (pH 7.6) containing 0.05% (w/v) sodium chloride, virus sedimented by high speed centrifugation and finally dispersed in 0.03 M phosphate buffer (pH 7.6).
Properties of Particles
Sedimentation coefficient (s°20,w): 155 S. No accessory particles are detected by analytical ultracentrifugation.
Ultraviolet absorption: max 258-260, min 242-246 nm. A260/A280 = 1.21. A260/A244 = 1.24.
Particle Structure
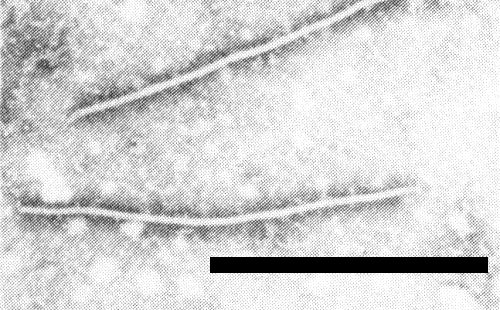
Particles are flexuous filaments (Fig. 7), previously reported to be 600-800 nm long (Herbas, 1964; Iwaki, 1967; Brants & Van den Heuvel, 1965), but mostly measuring c. 750 x 12 nm in neutral 2% phosphotungstate (Brunt, 1973).
Particle Composition
No information.
Relations with Cells and Tissues
Ovoid or spherical intracellular inclusion bodies up to 20 µm in diameter occur in infected plants (Kunkel, 1922; Bremer, 1926; Holmes, 1928); most cells in chlorotic areas of leaves contain a single inclusion (Fig. 8), usually near or apposed to the nucleus, and chloroplasts that are smaller than those in normal cells (Bremer, 1926).
Infected cells also contain numerous pinwheel and bundle inclusions similar to those induced by many other morphologically similar viruses.
Notes
Tomato spotted wilt, cucumber mosaic and tobacco mosaic viruses also occur naturally in Hippeastrum hybridum, sometimes together with hippeastrum mosaic virus (De Leeuw, 1972a; Herbas, 1964; Iwaki, 1967; Kahn & Scott, 1964; Smith, 1935), but they are easily distinguished from hippeastrum mosaic virus in differential hosts and by differences in particle morphology. Another virus with particles c. 650 x 13 nm has been detected in Hippeastrum hybridum cultivated in Europe (Brants, Fokkema & De Bode, 1970) but this has yet to be identified or characterized. A virus tentatively designated ‘hippeastrum streak’ infects H. vittatum in Papua and New Guinea (Van Velsen, 1967), but has been insufficiently characterized to permit comparisons with viruses infecting Hippeastrum species cultivated elsewhere.
Although hippeastrum mosaic virus is now widespread in Hippeastrum hybridum, virus-free plants are readily obtainable; c. 6% of bulblets developing on pieces of infected flower stem are virus-free (Holmes, 1965).
It is now generally accepted that the genus Amaryllis contains only the single S. African species A. belladonna whilst Hippeastrum is reserved for numerous species and hybrids originating in the USA (Moore, 1963). Before the nomenclature of the two genera had been resolved, Amaryllis was sometimes used in America as a synonym for Hippeastrum (Herbas, 1964; Johnson, 1951; Kahn & Scott, 1964); ‘amaryllis mosaic virus’ is thus an invalid synonym.
Figures
References list for DPV: Hippeastrum mosaic virus (117)
- Brants & Van den Heuvel, Neth. J. Pl. Path. 71: 145, 1965.
- Brants, Fokkema & De Bode, Neth. J. Pl. Path. 76: 171, 1970.
- Bremer, Meded. Proefstn Java-Suik Ind. 11: 337, 1926.
- Brunt, Rep. Glasshouse Crops Res. Inst. 1972: 103, 1973.
- De Leeuw, Neth. J. Pl. Path. 78: 69, 1972a.
- De Leeuw, Neth. J. Pl. Path. 78: 107, 1972b.
- Hannibal, Herbertia, La Jolla 9: 149, 1942.
- Herbas, Turrialba 14: 140, 1964.
- Holmes, Bot. Gaz. 86: 50, 1928.
- Holmes, Phytopathology 55: 504, 1965.
- Iwaki, Ann. phytopath. Soc. Japan 33: 237, 1967.
- Johnson, Phytopathology 41: 78, 1951.
- Kahn & Scott, Phytopathology 54: 360, 1964.
- Kunkel, Science, N.Y. 55: 73, 1922.
- Moore, Baileya 11: 15, 1963.
- Procenko & Procenko, Proc. 5th Conf. Czechosl. Pl. Virol. 1962: 241, 1964.
- Smith, Jl R. hort. Soc. 60: 304, 1935.
- Van Velsen, Papua New Guin. agric. J. 19: 13, 1967.