Details of DPV and References
DPV NO: 118 July 1973
Family: Potyviridae
Genus: Rymovirus
Species: Agropyron mosaic virus | Acronym: AgMV
Agropyron mosaic virus
J. T. Slykhuis Ottawa Research Station, Canada Department of Agriculture, Ottawa, Ontario, Canada
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
First reported by McKinney (1937).
Selected synonyms
- Agropyron green mosaic virus (Rev. appl. Mycol. 24: 136)
- Agropyron streak mosaic virus (Rev. appl. Mycol. 33: 285)
- Agropyron yellow mosaic virus (Rev. appl. Mycol. 24: 136)
- Couch grass streak mosaic virus (Rev. appl. Mycol. 36: 303)
- Wheat virus 2 (McKinney, 1937)
- Marmor agropyri (Rev. appl. Mycol. 24: 136)
- Agropyron streak mosaic virus (Rev. appl. Mycol. 33: 285)
-
A virus with filamentous particles c. 717 nm long and 15 nm in diameter infecting only species of Gramineae. It has been found naturally in Agropyron repens and wheat. It is transmitted by an eriophyid mite, and by rubbing leaves with infective sap.
Main Diseases
Causes light green to yellow mosaic in Agropyron repens (Fig. 1) and wheat. A strain found in Ontario caused a yellow mosaic, moderate stunting and loss in yield of wheat.
Geographical Distribution
USA (McKinney, 1937; Shepard, 1968; Slykhuis, 1952), Canada (Slykhuis, 1962), Finland (Bremer, 1964), Germany (Schumann, 1969).
Host Range and Symptomatology
Only species in the Gramineae have been reported systemically infected, generally developing light green to yellow mosaic on the leaves. In North America, wheat (Triticum aestivum and T. durum) was reported highly susceptible and Agropyron repens, A. elongatum and A. intermedium were moderately susceptible. Less susceptible species included Elymus canadensis, E. virginicus, Hordeum vulgare cvs. Brant and Husky, Lolium multiflorum cv. S22 and Secale cereale (Slykhuis & Bell, 1966). Additional hosts reported in Europe include Bromus japonicus and Hordeum murinum (Bremer, 1964) and Aegilops aegilopoides, Ae. crassa, Agropyron acutum, A. caninum, A. elmeri, A. pungens, A. sibiricum, A. villosum, Apera spicaventi, Bromus racemosus, B. secalinus, Cynosurus cristatus, Deyeuxia sylvatica, Hordeum maritimum, Lamarkia aurea, Panicum crus-galli, Phalaris paradoxa, Poa pratensis, Setaria glauca, S. viridis and Triticum bicorne (Schumann, 1969). Bremer (1964) also reported that the virus produced local lesions in a dicotyledonous species, Chenopodium quinoa.
-
Diagnostic species
- Systemic mosaic symptoms develop in wheat, Lolium multiflorum cv. S22, rye and some cultivars of barley; all these hosts are also susceptible to wheat streak mosaic virus, hordeum mosaic and ryegrass mosaic viruses. Agropyron mosaic virus is differentiated from these viruses, which have similar particles, by its ability to infect Agropyron repens but not oats.
-
Propagation species
- Agropyron repens
is the best host for maintaining the virus, but a number of cultivars of wheat, e.g. Kent, are more suitable as sources of virus for purification.Assay species
- Assays based on proportion of plants infected have been done using Kent wheat. Other cultivars of wheat should also be suitable.
Strains
Mild (green) and virulent (yellow) strains have been reported in the USA (McKinney, 1944) and in Canada (Slykhuis, 1962). The mild strain had little apparent effect on wheat, but the virulent strain caused significant stunting and loss of yield.
Transmission by Vectors
Eriophyid mites were usually present on Agropyron repens or wheat found infected with agropyron mosaic virus in the field, but mites transferred singly to test plants did not transmit the virus. However, a low percentage of wheat test plants became infected when colonies of the mite Abacarus hystrix (Fig. 2) were reared on Agropyron repens or wheat infected with the virus and the mites then blown to the test plants by a fan. No transmission occurred in similar tests with Aculus mckenziei or Aceria tulipae (Slykhuis, 1969).
Various insects (including aphids) failed to transmit the virus (Bremer, 1964; Schumann, 1969; Slykhuis, 1962; 1969).
Transmission through Seed
None reported.
Transmission by Dodder
No reports.
Serology
Antisera with precipitin titres of 1/1280 were prepared by injecting rabbits intra-muscularly with partially purified preparations of a yellow strain of the virus emulsified with Freund's incomplete adjuvant. The precipitin titre of clarified sap from infected Agropyron repens was about 1/16, and from infected wheat, 1/32 (Slykhuis & Bell, 1966).
Relationships
The mild and virulent strains tested in Canada were indistinguishable from each other in serological tube and micro-precipitin tests. Staples & Brakke (1963) suggested that agropyron mosaic virus was a mild strain of wheat streak mosaic virus, but Slykhuis & Bell (1966) found that although the particles of agropyron mosaic virus resembled those of hordeum mosaic and wheat streak mosaic viruses, they were slightly longer (717 nm as compared with 700 nm and 683 nm) and appeared more flexuous in leaf dip preparations. The viruses had some antigens in common, but were considered to be distantly related serotypes rather than closely related strains. Brakke (1971) found no serological relationship using low-titred antisera. Despite similarities in particle morphology and vector transmission, there is no evidence of serological relationship between any of these viruses and ryegrass mosaic virus (Slykhuis & Paliwal, 1972).
In plant protection tests, agropyron mosaic virus did not protect wheat against wheat streak mosaic virus but gave partial protection against hordeum mosaic virus.
Stability in Sap
In wheat sap, the thermal inactivation point (10 min) is about 50°C, dilution end-point about 10-3, and infectivity is retained at 20°C for 2-4 days.
Purification
The following method was used to partially purify the virus for the preparation of antigen (Slykhuis & Bell, 1966):
Inoculate wheat at the 3-leaf stage, harvest leaves 12-14 days later, freeze overnight at -20°C, thaw, grind at 4°C, express juice, acidify at pH 4.9 by adding 0.5 N HCl while stirring, allow to stand at 4°C for 30 min, centrifuge at 8000 g for 10 min, dilute 1:1 with 0.5 M neutral phosphate-buffered saline, centrifuge at 8000 g for 10 min, sediment and clarify by high and low speed centrifugation, resuspend pellets obtained at high speed in neutral phosphate-buffered saline. Work at 4°C.
Properties of Particles
Sedimentation rate the same as for wheat streak mosaic virus (Staples & Brakke, 1963) which has a sedimentation coefficient (s20, w) about 165 S (Brakke, 1971).
Particle Structure
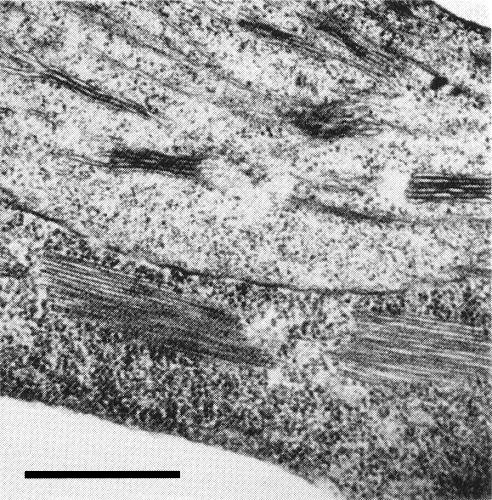
Particles are flexuous filaments (Fig. 3) about 15 nm in diameter and with a modal length of about 717 nm (Bremer, 1964; Slykhuis & Bell, 1966).
Particle Composition
No reports.
Relations with Cells and Tissues
Pinwheel inclusions, and bundles of virus particles (Fig. 4, Fig. 5), are present in the parenchyma cells of infected wheat (W. B. Langenberg, unpublished).
Notes
Brome mosaic virus might be confused with agropyron mosaic virus because it is also readily transmitted to Agropyron repens in which it occurs in Russia (Larina, 1968) and Jugoslavia (Milicic et al., 1966). It is readily distinguished because it produces severe effects on a much wider range of hosts, including Zea mays and oats, and induces local lesions in some dicotyledonous plants including Chenopodium hybridum and Datura stramonium. Also, it has much higher thermal inactivation and dilution end-points, and has isometric particles (Bancroft, 1970).
In wheat, agropyron mosaic virus can be confused with wheat streak mosaic or hordeum mosaic viruses. The symptoms can be differentiated by careful comparison under controlled conditions. Agropyron mosaic is readily distinguished from these viruses by its transmissibility to Agropyron repens but not to oats. Also it is not transmitted by Aceria tulipae, the efficient vector of wheat streak mosaic virus.
Agropyron mosaic virus is widely distributed in northern USA, southern Canada, and some areas of northern Europe. Its occurrence in wheat appears to depend on nearness to infected Agropyron repens. It occurs together with wheat streak mosaic virus in some areas, but the two viruses seem to spread entirely independently. Normally the symptoms in wheat are very mild and the incidence very low, but some strains in Ontario, Canada, can cause severe stunting and loss of yield. If such strains were to become prevalent and to spread rapidly in wheat they could cause severe losses. The possibility of this has been demonstrated in experimental plots in which high rates of spread occurred in immature wheat during the summer (Slykhuis, 1969).
Acknowledgements
Figs 3, 4 & 5 courtesy W. G. Langenberg, United States Department of Agriculture, Lincoln, Nebraska.
Figures

Leaves of Agropyron repens showing various symptoms of agropyron mosaic. The leaf on the left is from a healthy plant.
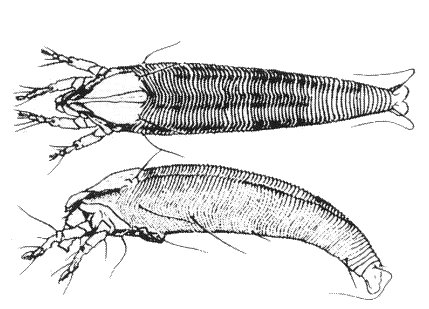
The mite vector, Abacarus hystrix, dorsal and lateral views. The adult mites are about 250 µm long (Keifer, 1952).
References list for DPV: Agropyron mosaic virus (118)
- Brakke, CMI/AAB Descriptions of Plant Viruses 48, 4 pp., 1971.
- Bancroft, CMI/AAB Descriptions of Plant Viruses 3, 3 pp., 1970.
- Bremer, Ann. Agric. Fenn. 3: 324, 1964.
- Keifer, Bull. Calif. Insect Surv. 2, 123 pp., 1952.
- Larina, Byull. vses. nauchno-issled. Inst. Zashch. Rast. 3: 60, 1968.
- McKinney, Circ. U.S. Dep. Agric. 442, 23 pp., 1937.
- McKinney, J. Wash. Acad. Sci. 34: 322, 1944.
- Milicic et al., Zast. Bilja 17: 213, 1966.
- Schumann, Phytopath. Z. 64: 258, 1969.
- Shepard, Pl. Dis. Reptr 52: 139, 1968.
- Slykhuis, Tech. Bull. S. Dak. agric. Exp. Stn 11, 29 pp., 1952.
- Slykhuis, Can. J. Bot. 40: 1439, 1962.
- Slykhuis, Phytopathology 59: 29, 1969.
- Slykhuis & Bell, Can. J. Bot. 44: 1191, 1966.
- Slykhuis & Paliwal, CMI/AAB Descriptions of Plant Viruses 86, 4 pp., 1972.
- Staples & Brakke, Phytopathology 53: 969, 1963.