Details of DPV and References
DPV NO: 127 July 1974
Family: Tombusviridae
Genus: Carmovirus
Species: Elderberry latent virus | Acronym: ElLV
Elderberry latent virus
A. T. Jones Scottish Horticultural Research Institute, Invergowrie, Dundee, Scotland
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Jones (1972).
Synonym
- Elderberry virus B (Jones & Murant, 1971)
-
An RNA-containing virus found only in American elder (Sambucus canadensis) from eastern USA. The infective particles are isometric, c. 30 nm in diameter. The virus is readily sap-transmissible to several herbaceous species. Its vector is unknown.
Main Diseases
Causes line-pattern symptoms in American elder (Sambucus canadensis) (Fig. 1, Fig. 2), which develops symptoms following mechanical inoculation only when kept in cool conditions. The virus appears to cause no symptoms in experimentally infected S. nigra.
Geographical Distribution
Eastern USA.
Host Range and Symptomatology
Found in naturally infected Sambucus canadensis cv. Adams No. 2 imported into UK from USA. Probably other cultivars in USA also contain the virus but its presence may be masked by nepoviruses (Jones, 1972). In limited host range studies it infected 25 species in the families Amaranthaceae, Chenopodiaceae, Cucurbitaceae and Solanaceae. Almost all plants were infected symptomlessly and many became infected only in inoculated leaves.
-
Diagnostic species
- Chenopodium quinoa.
Large chlorotic local lesions 10-14 days after inoculation, coalescing to produce a general chlorosis of inoculated leaves (Fig. 4); occasionally, systemic chlorotic or necrotic spots develop (Fig. 3). - Symptomless systemic infection occurs in Nicotiana clevelandii,
N. tabacum cv. Xanthi-nc
and Petunia hybrida.
Propagation species
- Chenopodium quinoa. Nicotiana clevelandii
is useful for maintaining cultures. -
Assay species
- No satisfactory assay species found. Assays can be made by determining the maximum infective dilution of preparations in Chenopodium quinoa; suitable dilutions of inoculum sometimes induce countable local lesions in C. quinoa leaves.
Strains
No information.
Transmission by Vectors
No vector reported. The aphids Macrosiphum euphorbiae and Myzus persicae did not transmit the virus from Chenopodium quinoa to C. quinoa when allowed 5, 30 or 180 min acquisition access periods (Jones, 1972).
Transmission through Seed
No information.
Transmission by Dodder
No information.
Serology
The virus is moderately immunogenic giving antiserum with gel-diffusion titres of 1/512. Reacts well in micro-precipitin tests or in gel-diffusion tests in 1% agar. It produces a single line of precipitation in gel-diffusion tests.
Relationships
No serological reaction was obtained with antisera to any of thirty-one distinct isometric viruses and the virus seems distinct from previously described viruses (Jones, 1972 and unpublished). Its particle size, physical properties and the sizes of its RNA and protein molecules are similar to those reported for tomato bushy stunt virus (Martelli, Quacquarelli & Russo, 1971), but the two viruses are unrelated serologically, have different sedimentation coefficients, and possibly differ in the number and arrangement of their coat polypeptides (Mayo & Jones, 1973).
Stability in Sap
In Chenopodium quinoa sap, the virus lost infectivity after 10 min at 85-90°C, storage at room temperature for 7 days, or dilution to 10-5 - 10-6 (Jones, 1972). Most of its infectivity is lost after 10 min at 65°C.
Purification
The virus is easily purified from inoculated Chenopodium quinoa leaves by chloroform clarification of leaf extracts followed by precipitation of the virus at pH 4.8 and differential centrifugation; treatment with n-butanol causes much loss of infectivity (Jones, 1972).
Properties of Particles
Purified preparations sediment as a major infective component (B) and a minor non-infective component (T).
Sedimentation coefficients (s°20, w): 48 S (T) and 112 S (B) (Fig. 5).
Molecular weight (daltons): about 7 x 106 (B) (Mayo & Jones, 1973).
Isoelectric point: about pH 4.8.
A260/A280: 1.62(B).
Buoyant density: When centrifuged to equilibrium in CsCl solution, purified preparations of an uncloned isolate formed a major band at 1.36 g/cm3 and a minor band at 1.37 g/cm3 (Mayo & Jones, 1973).
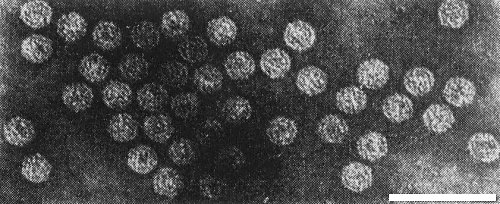
Particle Structure
Particles are disrupted in 2% sodium phosphotungstate but in uranyl formate (Fig. 6, Fig. 7) they are isometric with diameters of about 17 nm (T) or 30 nm (B). Particles are not penetrated by the stain (Jones, 1972). Calculations based on the M. Wt of the protein and RNA components suggest that the B component may contain 120 protein subunits (Mayo & Jones, 1973).
Particle Composition
Nucleic acid: RNA, single-stranded (Jones, 1972), about 23% of B particle weight. M. Wt, estimated from polyacrylamide gel electrophoresis, is 1.55 x 106 (Mayo & Jones, 1973).
Protein: In polyacrylamide gels the protein from B particles migrated as a single major component with an estimated M. Wt of 40,000 (Mayo & Jones, 1973).
Relations with Cells and Tissues
No information.
Notes
Because the virus caused no symptoms in S. canadensis or S. nigra after 18 months in a heated glasshouse it was called elderberry latent virus (Jones, 1972). However, in cooler conditions leaves of infected plants of S. canadensis, but not S. nigra, develop line-pattern symptoms.
The virus is distinct from other isometric viruses found in elder: the nepoviruses (arabis mosaic, cherry leaf roll, strawberry latent ringspot, tobacco ringspot and tomato ringspot), induce characteristic symptoms in Chenopodium quinoa and cucumber and have a different sedimentation behaviour; cucumber mosaic and tobacco necrosis viruses do not infect C. quinoa systemically. All are serologically unrelated to elderberry latent virus.
Figures
References list for DPV: Elderberry latent virus (127)
- Jones & Murant, Rep. Scott. hort. Res. Inst. 1970: 54, 1971.
- Jones, Ann. appl. Biol. 70: 49, 1972.
- Martelli, Quacquarelli & Russo, CMI/AAB Descriptions of Plant Viruses 69, 4 pp.,1971.
- Mayo & Jones, J. gen. Virol. 19: 245, 1973.