Details of DPV and References
DPV NO: 130 July 1974
Family: Tombusviridae
Genus: Carmovirus
Species: Pelargonium flower break virus | Acronym: PFBV
Pelargonium flower break virus
M. Hollings Glasshouse Crops Research Institute, Littlehampton, Sussex, England
Olwen M. Stone Glasshouse Crops Research Institute, Littlehampton, Sussex, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Stone & Hollings (1973).
A virus with isometric particles c. 30 nm in diameter. Found occasionally in pelargoniums (Pelargonium domesticum) in England, and causes flower-breaking in some pelargonium types. It is transmitted by inoculation of sap, and has a rather limited host range.
Main Diseases
Causes flower-breaking in some pelargonium seedling lines (Fig. 3), but is symptomless in many cultivars. Occurs in pelargoniums less frequently than other viruses.
Geographical Distribution
Found in several pelargonium cultivars in England.
Host Range and Symptomatology
Infected 15 of 100 species tested, in 6 of 35 plant families (Amaranthaceae, Aizoaceae, Caryophyllaceae, Chenopodiaceae, Geraniaceae and Solanaceae).
-
Diagnostic species
- No single test species gives diagnostic reactions.
Inoculation of sap from pelargonium is much
more effective when 1-2% of polyethylene glycol M. Wt 6000 is added to the buffer.
- Chenopodium quinoa. Numerous chlorotic and necrotic local lesions c. 1 mm diameter after 7-10 days (Fig. 1); occasional systemic infection, with leaf mottling.
- Gomphrena globosa. Few, brown necrotic local lesions c. 1 mm diameter; no systemic infection. Inocula with low virus concentration cause symptomless infection in inoculated leaves.
- Nicotiana clevelandii. Diffuse chlorotic, occasionally necrotic, local lesions after 15-21 days (Fig. 2); no systemic infection.
- Tetragonia expansa. Few chlorotic local lesions 2-3 mm diameter, visible after 4 weeks as the inoculated leaves age; no systemic infection.
- Seedlings of pelargonium type Nittany Lion (Kemp, 1969) are apparently immune to this virus.
- Chenopodium quinoa. Numerous chlorotic and necrotic local lesions c. 1 mm diameter after 7-10 days (Fig. 1); occasional systemic infection, with leaf mottling.
-
Propagation species
- The virus can be maintained in Nicotiana megalosiphon, which develops symptomless systemic infection, or in pelargonium seedlings; inoculated leaves of N. clevelandii are a much better source of virus for purification.
-
Assay species
- Chenopodium quinoa
seems the best local lesion host.
Strains
Isolates of the virus differ little.
Transmission by Vectors
Not transmitted by the aphid Myzus persicae after short acquisition feeds.
Transmission through Seed
Apparently not transmitted through seed of pelargonium seedling clones.
Transmission by Dodder
No transmission by Cuscuta campestris.
Serology
The virus is a good immunogen; rabbits immunized by one intravenous plus two intramuscular injections (with Freund’s complete adjuvant) over a 3-week period gave antisera with specific titres in precipitin tube tests of 1/2048 (Stone & Hollings, 1973). The precipitates are granular (somatic). In gel-diffusion tests, a single reaction line is formed. Although satisfactory reactions in gel can be obtained with crude infective sap from N. clevelandii (and less satisfactory ones from C. quinoa), tannins in pelargonium sap produce non-specific precipitates with normal serum.
Relationships
No serological relationships were found to any of 42 other isometric viruses (Stone & Hollings, 1973). However, on the basis of sedimentation characteristics and biochemical criteria, pelargonium flower-break virus might be grouped with certain other single-component viruses such as carnation mottle and turnip crinkle.
Stability in Sap
In crude sap of N. clevelandii or C. quinoa, the thermal inactivation point is 85-90°C (though over 50% of infectivity is lost at 75°C). The dilution end-point is 10-5 to 10-6, compared with 10-3 to 10-4 in pelargonium sap, and infectivity survives for about 3 weeks at laboratory temperature (20°C). Lyophilized sap of C. quinoa, containing 7% dextrose + 7% peptone, was still infective after 4 years’ storage in vacuo at laboratory temperature.
Purification
The following method gives good yields (Stone & Hollings, 1973); preparations from N. clevelandii may yield 100 mg virus per kg leaf tissue. Harvest inoculated leaves of N. clevelandii about 3 weeks after inoculation. Homogenize leaves at room temperature with 0.05 M phosphate buffer (pH 7.6) containing 0.1% thioglycollic acid, using 4 ml buffer/g leaf. Express juice through cheesecloth, and add 9.3 ml n-butanol to every 100 ml extract. Shake the extract for 5 min and store it at c. 2°C for 24 h. Concentrate the virus by one or more cycles of differential centrifugation (12,000 g for 25 min, then 100,000 g for 2 h). Re-suspend the final pellets in 0.03 M phosphate buffer and, after 2-3 h, remove insoluble material by brief centrifugation. Purify further by density-gradient centrifugation and re-sedimenting the extracted virus zone.
Properties of Particles
One specific light-scattering band in density-gradient centrifugation. One sedimenting component with s°20, w: 125 S.
In immunoelectrophoresis at 4°C, using Ionagar in 0.03 M phosphate buffer pH 7.6, the virus migrates as a single antigenic component towards the cathode.
A260/A280: 1.64; Amax(260)/Amin(246): 1.08 (values corrected for light-scattering).
Particle Structure
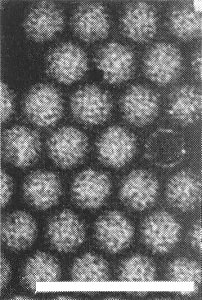
Particles are isometric, the diameter ranging from 27 nm in 1% ammonium molybdate (pH 6.8) to 34 nm in 2% uranyl acetate; particles are more or less disrupted in potassium phosphotungstate or 2% uranyl nitrate (Fig. 4, Fig. 5, Fig. 6, Fig. 7) (Stone & Hollings, 1973).
Particle Composition
Nucleic acid: RNA, single-stranded (M. Hollings & O. M. Stone, unpublished data).
Protein: Particles contain a protein of M. Wt 41,200 (Stone & Hollings, 1973).
Relations with Cells and Tissues
All tissues are infected; no inclusion bodies have been reported.
Notes
Several isometric viruses occur in pelargoniums: the pelargonium leaf curl strain of tomato bushy stunt virus induces local lesions in Nicotiana glutinosa and N. tabacum; cucumber mosaic virus and four nepoviruses (tomato black ring, tomato ringspot, tobacco ringspot and arabis mosaic) all cause symptoms in N. tabacum and Cucumis sativus. Pelargonium flower-break virus does not infect any of these plants. Pelargonium ringspot, another isometric virus (M. Hollings & O. M. Stone, unpublished data) systemically infects N. clevelandii, but without causing symptoms. All these viruses can be distinguished serologically and by sedimentation behaviour from pelargonium flower-break virus.
Pelargoniums can be freed from flower-break virus by heat-treatment (4 weeks at 35°C) followed by rooting small cuttings from the stem tips, but pelargoniums do not withstand thermotherapy well.
Figures
References list for DPV: Pelargonium flower break virus (130)
- Stone & Hollings, Ann. appl. Biol. 75: 15, 1973.
- Kemp, Can. Pl. Dis. Surv. 49: 1, 1969.