Details of DPV and References
DPV NO: 134 July 1974
Family: Potyviridae
Genus: Potyvirus
Species: Cowpea aphid-borne mosaic virus | Acronym: CABMV
Cowpea aphid-borne mosaic virus
K. R. Bock Overseas Development Administration, Crop Virology Project, EAAFRO, Nairobi, Kenya
M. Conti Laboratorio di Fitovirologia Applicata, Consiglio Nazionale delle Ricerche, Turin, Italy
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Lovisolo & Conti (1966) and Bock (1973).
- Synonyms (see also under Relationships)
- Cowpea common mosaic virus (Rev. appl. Mycol. 48: 2838)
- Cowpea mosaic virus (in part) (Rev. appl. Mycol. 20: 444; 26: 524). This name is now used for the isometric virus that is the type member of the comovirus group - see Van Kammen (1971)
- Cowpea mosaic virus (in part) (Rev. appl. Mycol. 20: 444; 26: 524). This name is now used for the isometric virus that is the type member of the comovirus group - see Van Kammen (1971)
- A distinctive virus with flexuous filamentous particles c. 750 nm long. It is seed-borne in cowpea, has a wide experimental host range, is transmitted by several common species of aphid, and occurs in many countries where cowpea is grown.
Main Diseases
Causes a severe mosaic of cowpea (Vigna unguiculata), the severity depending on host cultivar and virus strain. Diseased cowpea plants show variable amounts of dark green vein banding or interveinal chlorosis, leaf distortion, blistering and stunting (Fig. 1, Fig. 2, Fig. 3, Fig. 4). Viruses that may be related cause mosaic diseases of adzuki bean (Phaseolus angularis) and asparagus bean (V. sesquipedalis).
Geographical Distribution
Occurs in Africa (Kenya, Uganda and Nigeria), Europe (Italy, and probably Rumania), and Asia (India, Iran, Japan, and probably China). Viruses that are probably related occur in the USA (Florida) and in the south-west Pacific area.
Host Range and Symptomatology
Infects many species in the Leguminosae and most strains also infect members of the Amaranthaceae, Chenopodiaceae, Cucurbitaceae, Labiatae and Solanaceae (Lovisolo & Conti, 1966; Bos, 1970; Bock, 1973).
- Diagnostic species
- Chenopodium amaranticolor
. Local chlorotic or necrotic lesions (Fig. 5); not systemic. - Glycine max (soybean). Mild systemic mottle with more or less pronounced
veinbanding.
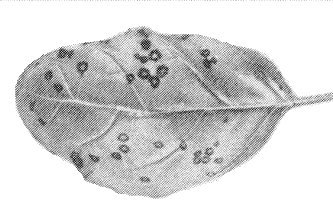
- Ocimum basilicum (basil). Local necrotic lesions with whitish centres (Fig. 7); not systemic.
- Petunia hybrida. Systemic infection, either symptomless or with dark green patches surrounding mid-ribs of leaves.
- Phaseolus vulgaris (French bean). Local chlorotic or necrotic lesions which spread along the veins (Fig. 6). Normally restricted to inoculated leaves (in cvs. The Prince, Canadian Wonder, Saxa, Scotia) but systemic infection may follow in cv. Bataaf (European (type) strain; Bos, 1970) or in cv. The Prince (African mild strain; Bock, 1973).
- Pisum sativum (pea). Systemic mottle in many cvs. (Onward, Greenfeast, Koroza), but symptomless systemic infection in others (Laxton).
- Ocimum basilicum (basil). Local necrotic lesions with whitish centres (Fig. 7); not systemic.
- Propagation species
- Cultures are best maintained in cowpea, soybean or petunia. The virus should
be propagated in cowpea for virus purification; soybean is unsatisfactory for
this purpose.
- Assay species
- Chenopodium amaranticolor
is a good local lesion host.
Strains
1. European (type) strain (Lovisolo & Conti, 1966). Causes a severe distorting mosaic in cowpea (Fig. 1).
2. African (neo-type) strain (Bock, 1973). Closely resembles the European strain in symptom expression. In cowpea cv. Mak 1, induces irregular angular broken mosaic (Fig. 2).
3. African mild strain (Bock, 1973). In cowpea cv. Mak 1, induces a very mild mottle, with little or no effect on growth of the plants.
4. African veinbanding strain (Bock, 1973). In cowpea cv. Mak 1, induces a broad dark-green veinbanding (Fig. 3).
Transmission by Vectors
The virus is transmitted in the stylet-borne, non-persistent manner by Aphis craccivora (Bock, 1973), A. fabae, A. gossypii, A. medicaginis, Macrosiphum euphorbiae and Myzus persicae (Vidano & Conti, 1965). Non-feeding aphids can retain infectivity up to 15 h after virus acquisition (M. Conti, unpublished data).
Transmission through Seed
Seed transmission is usually slight (0-3 %) but instances of 21.5% have been recorded in cowpea cv. Kurodane 16 for a virus which resembles the type strain and 14.7% for a virus which may be related, adzuki bean mosaic (Tsuchizaki, Yora & Asuyama, 1970a). Seed transmission ranged from 3 to 19% in cv. Pusa Phalguni for an Indian isolate serologically related to the African (neo-type) virus (H. C. Phatak, personal communication). Plants infected through the seed show bright mosaic symptoms in primary leaves (Fig. 4).
Transmission by Dodder
Not reported.
Serology
The virus is moderately to strongly immunogenic; an antiserum with a tube precipitin titre of 1/8192 has been obtained (Bock, 1973).
Relationships
Among the African strains, the neo-type and mild strains are serologically identical but the veinbanding strain is distinguishable although related. No serological tests have been made between the African isolates and the European type strain but they are considered to be strains of the same virus because both are distantly serologically related to bean common mosaic virus (Lovisolo & Conti, 1966: Bock, 1973) and they resemble one another in particle size and morphology, physical properties, host range and symptomatology, and in transmission by aphids. Although cowpea aphid-borne mosaic virus belongs to the potyvirus group, no direct serological relationship was detected between the African neo-type strain and potato virus Y, bean yellow mosaic, pea seed-borne mosaic, clover yellow vein, soybean mosaic, sugarcane mosaic, tobacco severe etch or iris mosaic viruses (Bock, 1973).
In symptomatology, properties in vitro, and transmission by aphids and through seed, cowpea aphid-borne mosaic virus resembles the cowpea mosaic viruses of McLean (1941), Yu (1946), Van Velsen (1962) and Quantz (see Brandes, 1964), the asparagus bean mosaic virus of Snyder (1942) and Hino (1960), and the Blackeye cowpea mosaic virus of Anderson (1955). It may be related to or synonymous with some or all of these. Another virus that is possibly related is adzuki bean mosaic virus, which occurs in Japan (Hino, 1962) and is apparently confined to species in the Leguminosae. No serological comparisons have been made but it protects adzuki bean plants against infection with cowpea aphid-borne mosaic virus (Tsuchizaki et al., 1970a).
Stability in Sap
Isolates differ somewhat in stability, but in cowpea sap the thermal inactivation point lies between 57°C and 60°C, the dilution end-point is between 10-3 and 10-4 and infectivity is retained at 20°C for 1-3 days. Frozen infected leaves retain infectivity for at least 7 weeks (Bock, 1973; Lovisolo & Conti, 1966).
Purification
Bock (1973). Homogenize systemically infected cowpea leaves in 0.5 M sodium citrate buffer containing 1% 2-mercapto-ethanol pH 8.1 (2 ml buffer/1g tissue), clarify by treatment with 8.5 ml n-butanol/100 ml sap for 45 min centrifuge differentially; resuspend in 0.01 M phosphate buffer pH 7.7.
Properties of Particles
Sedimentation coefficient (s°20, w): 150 S for unaggregated particles.
Particle Structure
Particles are flexuous filaments c. 750 nm long (Fig. 8) (Lovisolo & Conti, 1966; Bock, 1973).
Particle Composition
No information.
Relations with Cells and Tissues
The virus induces numerous granular inclusions in epidermal cells of pea but few in petunia (Bos, 1970). The virus infects most parts of cowpea plants, including pollen, anthers and ovaries (Tsuchizaki et al., 1970b) and embryos (H. C. Phatak, personal communication).
Notes
Several viruses induce mosaic symptoms in cowpea in the field; although vector type and biophysical properties are useful in diagnosis, particle morphology and serology are usually necessary for unequivocal identification.
Both cowpea mosaic (Van Kammen, 1971) and cowpea chlorotic mottle (Bancroft, 1971) viruses are readily distinguishable from cowpea aphid-borne mosaic virus by their isometric particles and transmission by leaf beetles. Cowpea mild mottle virus (Brunt & Kenten, 1973) has 650 nm filamentous particles and, unlike cowpea aphid-borne mosaic virus, apparently is not transmitted by aphids.
The cowpea strains of tobacco mosaic (Lister & Thresh, 1955) and southern bean mosaic (Shepherd & Fulton, 1962) viruses both survive temperatures in plant sap greatly in excess (more than 20°C) of that which inactivates cowpea aphid-borne mosaic virus. Although the cowpea strain of bean yellow mosaic virus (Brierley & Smith, 1962) is best distinguished from cowpea aphid-borne mosaic virus serologically, it induces severe systemic yellow spotting in Chenopodium amaranticolor in contrast to the known strains of cowpea aphid-borne mosaic virus, which do not invade this host systemically.
Figures
References list for DPV: Cowpea aphid-borne mosaic virus (134)
- Anderson, Pl. Dis. Reptr 39: 349, 1955.
- Bancroft, CMI/AAB Descriptions of Plant Viruses 49, 4 pp., 1971.
- Bock, Ann. appl. Biol. 74: 75, 1973.
- Bos, Neth. J. Pl. Path. 76: 8, 1970.
- Brandes, Mitt. biol. BundAnst. Ld.- u. Forstw. 110, 130 pp., 1964.
- Brierley & Smith, Pl. Dis. Reptr 46: 335, 1962.
- Brunt & Kenten, Ann. appl. Biol. 74: 67, 1973.
- Hino, Ann. phytopath. Soc. Japan 25: 178, 1960.
- Hino, Ann. phytopath. Soc. Japan 27: 138, 1962.
- Lister & Thresh, Nature, Lond. 175: 1047, 1955.
- Lovisolo & Conti, Neth. J. Pl. Path. 72: 265, 1966.
- McLean, Phytopathology 31: 420, 1941.
- Shepherd & Fulton, Phytopathology 52: 489, 1962.
- Snyder, Phytopathology 32: 518, 1942.
- Tsuchizaki, Yora & Asuyama, Ann. phytopath. Soc. Japan 36: 112, 1970a.
- Tsuchizaki, Yora & Asuyama, Ann. phytopath. Soc. Japan 36: 237, 1970b.
- Van Kammen, CMI/AAB Descriptions of Plant Viruses 47, 4 pp., 1971.
- Van Velsen, Papua New Guin. agric. J. 14: 153, 1962.
- Vidano & Conti, Atti. Accad. Sci. Torino 99: 1041, 1965.
- Yu, Ann. appl. Biol. 33: 450, 1946.