Details of DPV and References
DPV NO: 135 July 1974
Family: Reoviridae
Genus: Fijivirus
Species: Rice black streaked dwarf virus | Acronym: RBSDV
Rice black streaked dwarf virus
E. Shikata Department of Botany, Faculty of Agriculture, Hokkaido University, Sapporo, Japan
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
First described by
Kuribayashi & Shinkai (1952).
A virus with isometric particles c. 75-80 nm in diameter. The virus is not mechanically transmissible but is transmitted in a persistent manner by planthoppers. Host range of the virus is restricted to species of Gramineae. Infected plants show severe stunting, with neoplastic proliferations along the veins. The disease causes serious damage to rice, maize, wheat and barley in Japan.
Main Diseases
Causes stunting of plants, deformation of leaves, and white waxy or black-streaked swellings along the veins in rice, maize, wheat, oat and barley (Fig. 1, Fig. 2, Fig. 3, Fig. 4, Fig. 5).
Geographical Distribution
Japan.
Host Range and Symptomatology
All known host plants are members of the Gramineae, in which 25 species, including rice, maize, wheat, oat and barley can be infected by means of planthoppers (Shinkai, 1962). Not sap-transmissible.
-
Diagnostic species
- Oryza sativa
(rice). Most japonica varieties of rice are very susceptible; indica varieties are more or less resistant (Morinaka & Sakurai, 1968). Japanese paddy rice usually shows pronounced stunting (Fig. 1), darkening of leaves, and twisting of the distal portions of young leaves. The typical symptoms in rice are white waxy swellings along the veins on the underside of the leaf blades, sheaths and culms; later they become dark brown, forming black-streaked tumours of various lengths (Fig. 2). Diseased plants produce poor heads or none at all; sometimes the heads are retained within the sheaths. Dark brown blotches appear on some of the grains. Symptoms appear after an incubation period of 14-24 days (Shinkai, 1962). Zea mays (maize). Infected plants are dwarfed with darkened leaves. At an early stage of infection, white streaks appear along the veins and white waxy swellings occur on the veins on the under surfaces of the leaves (Fig. 4). Margins of old affected leaves split (Fig. 5). Incubation period in maize plants is 7-10 days (Shinkai, 1962).
Triticum sativum (wheat), Avena sativa (oat) and Hordeum vulgare (barley) are severely stunted (Fig. 3) and produce twisted leaves, sometimes with white waxy swellings on the veins on the under surfaces of leaves and culms.
-
Propagation species
- Japanese rice varieties, wheat, oat and barley are good plants for maintaining the virus, but maize cannot be used because it is a poor host for the vector (see Assay species). For virus purification, rice and maize plants are good sources. Virus contents are high in wheat and barley, but contamination by bacteriophages sometimes occurs (Kitagawa & Shikata, 1974).
-
Assay species
- Maize (cv. Golden Cross Bantam) is highly susceptible to the virus (Ishii, Yoshimura & Soejima, 1969), showing obvious white streaks on the leaves 7-10 days after inoculation. When fed on maize, virus-carrying vector insects (Laodelphax striatellus) usually die within 2-3 days and never lay eggs, but about 80-100% of the plants may be infected. Thus diseased maize cannot be used as a propagation species (Ishii & Yoshimura, 1969; Kitagawa & Shikata, 1973).
Strains
No strains reported.
Transmission by Vectors
Three planthopper species are known to transmit the virus in a persistent manner: Laodelphax striatellus (Kuribayashi & Shinkai, 1952; Shinkai, 1962) (Fig. 6), Unkanodes sapporona (Shinkai, 1966), and U. albifascia (Shinkai,1967). No transmission occurs through the eggs (Shinkai, 1962). Insects acquire the virus after feeding on infected rice plants for 30 min. Minimum inoculation feeding time is about 5 min (Shinkai, 1962). Incubation period in feeding insects is 7-35 days (Shinkai, 1962), after which the insects are viruliferous until death. A large proportion of insects injected with extracts from diseased plants or insects transmit the virus (Kitagawa & Shikata, 1969a). By injecting insects serially with insect extracts, it was shown that the virus multiplies in its vector (Kitagawa & Shikata, 1974).
Transmission through Seed
None found.
Transmission by Dodder
None found.
Serology
Rabbits receiving intravenous injections of purified preparations containing particles 60 nm in diameter yielded antiserum with a titre of 1/8192 in the ring precipitin test, and 1/1024 in the agar gel-diffusion test (Luisoni et al., 1973).
Relationships
The morphology of the particles indicates that this virus probably belongs to the same group as wound tumor, rice dwarf, maize rough dwarf and sugarcane Fiji disease viruses. In host range and symptomatology, properties in vitro and transmission by planthoppers, rice black streaked dwarf virus closely resembles maize rough dwarf virus, and Luisoni et al. (1973) reported that these two viruses are serologically related.
Stability in Sap
Thermal inactivation point (10 min) of the virus in rice extracts is 60°C. Longevity in vitro at 4°C is 7 days in rice sap, 6 days in insect extracts. Infectivity survived more than 232 days in frozen rice leaves. Dilution end-point is 10-5 in rice leaf extracts, 10-6 in insect extracts. The virus in crude rice leaf extracts retained its infectivity after treatment at pH 5-9, or with 0.01 M ethylenediamine-tetraacetic acid, 0.1% sodium deoxycholate, 40% carbon tetrachloride or 20% fluorocarbon, but lost infectivity after treatment with 10-50% chloroform, 3-10% n-butanol or 10-20% of a 1:1 mixture of chloroform and n-butanol. However, purified virus preparations were not inactivated by treatment with 50% chloroform or 50% n-butanol (Kitagawa & Shikata, 1973).
Purification
Grind vector insects or diseased leaves of rice, maize, wheat or barley in cold 0.1 M phosphate buffer, pH 7.0. Clarify the extracts with 30% carbon tetrachloride, followed by differential centrifugation (8000 rev/min for 30 min, 20,000 rev/min for 60 min). Resuspend the pellets after high speed centrifugation in 0.001 M phosphate buffer, pH 7.0. Further purification is by sucrose density gradient centrifugation; infectivity is associated with a single light-scattering zone. Virus concentrations are highest in barley and in insect extracts (Kitagawa & Shikata, 1969b).
Properties of Particles
Not investigated.
Particle Structure
Virus particles in purified preparations are isometric, c. 60 nm in diameter (Fig. 9), but two kinds of particles, c. 75-80 nm and c. 50-55 nm in diameter, occur in cells of infected plants and insects. Leaf extracts in phosphate buffer or distilled water contain particles c. 60 nm in diameter without obvious outer projections, identical to those seen in purified virus preparations. Leaves fixed in 2% osmium tetroxide or 2% paraformaldehyde solutions before extraction yield large particles c. 75-80 nm in diameter with outer projections (Fig. 10). Probably the virus particles lose the outer projections during the purification procedures (Shikata, 1973).
Particle Composition
Double-stranded RNA was detected by the acridine orange test described by Bradley (1965) (Y. Kitagawa & T. Kodama, personal communication).
Relations with Cells and Tissues
Swellings on the leaves result from hyperplasia and hypertrophy of phloem cells of infected plants. Inclusion bodies, about 6.5 µm in diameter and stainable with giemsa, azure II, pyronin, trypan blue or acetocarmine, appear within neoplastic cells (Kashiwagi, 1966) (Fig. 8). Ultrathin sections of diseased leaves reveal the inclusions as granular structures (‘viroplasm’) within hypertrophied phloem cells (Fig. 7). Small particles of c. 50-55 nm occur in the viroplasms and large particles c. 75-80 nm are scattered in the surrounding cytoplasm (Fig. 11). Large particles are sometimes seen arranged within tubular structures (Fig. 10). Crystalline aggregates composed of large particles are also found in the infected cells (Shikata, 1969).
In infected insect cells, large areas of the cell cytoplasm are occupied by viroplasms, identical with those in infected plant cells. Large particles are usually scattered in the cytoplasm around the viroplasms, or arranged within tubular structures and in crystalline arrays (Fig. 12). Virus accumulations are found in cells of the fat bodies, salivary glands and intestines (Shikata, 1969).
Notes
Although maize rough dwarf virus is reported to be related to rice black streaked dwarf virus it seems to differ in being unable to infect Japanese or Italian varieties of rice and in being transmitted through the egg of the planthopper vector. Induction of swellings or black-streaked tumours on the veins distinguishes rice black streaked dwarf virus from other viruses in rice.
Figures

Black-streaked and white waxy tumours on leaves (right) and culms (left and middle) of infected rice plants. (Courtesy of Dr A. Shinkai.)

One of the insect vectors, Laodelphax striatellus. Male (left), female (middle), 4th-instar nymph (right) (Courtesy of Dr A. Shinkai.)
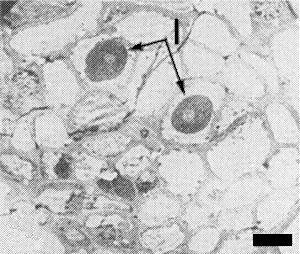
Low magnification electron micrograph showing inclusions (I) within infected maize cells. Bar represents 5 µm.

Virus particles, 60 nm in diameter, in purified preparation, stained with phosphotungstate. Bar represents 100 nm.

Large virus particles 75-80 nm in diameter within a tubular structure, in dip preparation from OsO4-fixed leaves. Bar represents 100 nm.
References list for DPV: Rice black streaked dwarf virus (135)
- Bradley, Nature, Lond. 205: 1230, 1965.
- Ishii & Yoshimura, Ann. phytopath. Soc. Japan 35: 388, 1969.
- Ishii, Yoshimura & Soejima, Ann. phytopath. Soc. Japan 35: 361, 1969.
- Kashiwagi, Ann. phytopath. Soc. Japan 32: 168, 1966.
- Kitagawa & Shikata, Mem. Fac. Agric. Hokkaido Univ. 6: 439, 1969a.
- Kitagawa & Shikata, Mem. Fac. Agric. Hokkaido Univ. 6: 446, 1969b.
- Kitagawa & Shikata, Rep. Tottori mycol. Inst. 10: 787, 1973.
- Kitagawa & Shikata, Ann. phytopath. Soc. Japan 40: 329, 1974.
- Kuribayashi & Shinkai, Ann. phytopath. Soc. Japan 16: 41, 1952.
- Luisoni, Lovisolo, Kitagawa & Shikata, Virology 52: 281, 1973.
- Morinaka & Sakurai, Bull. Chugoku agric. exp. Stn Ser. E 2: 1, 1968.
- Shikata, in The Virus Diseases of the Rice Plant, compiled by Int. Rice Res. Inst., Johns Hopkins, Baltimore p. 223, 1969.
- Shikata, Ann. phytopath. Soc. Japan 39: 221, 1973.
- Shinkai, Bull. natn. Inst. agric. Sci., Tokyo Ser. C, 14: 1, 1962.
- Shinka, Ann. phytopath Soc. Japan 32: 317, 1966.
- Shinkai, Ann. phytopath. Soc. Japan 35: 318, 1967.






