Details of DPV and References
DPV NO: 137 July 1974
Family: Unallocated ssRNA+ viruses
Genus: Umbravirus
Species: Carrot mottle virus | Acronym: CMoV
Carrot mottle virus
A. F. Murant Scottish Horticultural Research Institute, Invergowrie, Dundee, Scotland
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Stubbs (1948,
1952) and
Watson, Serjeant & Lennon (1964).
Synonym
- Carrot motley dwarf virus (in part) (Rev. appl. Mycol. 28: 435)
-
An RNA-containing virus with enveloped isometric particles c. 52 nm in diameter. It is transmitted in a persistent manner by the aphid Cavariella aegopodii but usually only from umbelliferous plants that also contain carrot red leaf virus. It is sap-transmissible but has only a moderate host range. Reported from four continents.
Main Diseases
In the field, carrot mottle virus probably occurs only in mixed infections with carrot red leaf virus (Watson et al., 1964). The virus complex causes yellowing and reddening of carrot (Daucus carota) leaves with variable amounts of mottling and severe stunting (Fig. 1), hence the disease name ‘motley dwarf’ (Stubbs, 1948); the disease causes serious reduction in yield of carrots in some years (Watson, 1960; Watson & Serjeant, 1964). The virus complex causes a similar disease in parsley (Petroselinum crispum) (Frowd & Tomlinson, 1972). Carrot plants infected with carrot red leaf virus alone may lack the mottle symptom and the severe stunting, but plants infected experimentally with carrot mottle virus alone are symptomless.
Geographical Distribution
Motley dwarf disease is reported from Australia (Stubbs, 1948), New Zealand (Anon., 1959), Japan (Komuro & Yamashita, 1956), USA (Stubbs, 1956) and Europe (Watson, 1960; Heinze, 1968). So far as can be judged from the descriptions given, the carrot mottle virus component of the complex was probably present in all these countries. Carrot mottle virus itself is reported from UK (Watson et al., 1964) and Germany (Heinze, 1968; Wolf, 1970; Wolf & Schmelzer, 1972).
Host Range and Symptomatology
Carrot mottle virus, unlike carrot red leaf virus, is transmissible by inoculation of sap. It infects a limited range of dicotyledonous plants, but some only with difficulty, especially in glasshouse conditions in summer. Hosts have been reported in the Amaranthaceae, Chenopodiaceae, Papilionaceae, Solanaceae and Umbelliferae. In transmission experiments using the aphid vector, species of Umbelliferae may become infected with carrot red leaf virus as well as carrot mottle virus, whereas non-umbelliferous hosts, which are immune to carrot red leaf virus, become infected with carrot mottle virus alone. The following descriptions of symptoms are for carrot mottle virus alone.
-
Diagnostic species
- Chenopodium quinoa.
Within 7 days, pinpoint chlorotic spots in inoculated leaves, becoming necrotic, developing poorly in summer (Fig. 2). Not systemically infected. - Coriandrum sativum (coriander). Systemic chlorotic mottle or yellowing with
varying
amounts of necrotic flecking. Moderate stunting.
- Nicotiana clevelandii. Local lesions, when present, are yellow or slightly necrotic. Systemic fine vein-etching 9-10 days after inoculation (Fig. 4) followed by partial recovery to give a mild light and dark green mottle with slight stunting.
- N. tabacum (tobacco) cv. Xanthi-nc. Local silver-grey necrotic broken rings, systemic necrotic ring and line patterns.
- Phaseolus vulgaris (French bean) cv. The Prince. In winter, dark brown local lesions 0.5-1 mm diameter, 2-3 days after inoculation (Fig. 3). Not systemically infected.
- Nicotiana clevelandii. Local lesions, when present, are yellow or slightly necrotic. Systemic fine vein-etching 9-10 days after inoculation (Fig. 4) followed by partial recovery to give a mild light and dark green mottle with slight stunting.
-
Assay species
- Chenopodium quinoa
and Nicotiana tabacum cv. Xanthi-nc have been used as local lesion assay hosts (Watson et al., 1964; Murant et al., 1969). However, a better test plant is Phaseolus vulgaris cv. The Prince grown before and after inoculation in simulated winter conditions, e.g. 20°C, with illumination at 2500 lux for 8 h per day (Murant, Roberts & Goold, 1973). For tests using the aphid vector, coriander is probably the best species, but carrot and chervil (Anthriscus cerefolium) may also be used. All three species develop severe yellowing and reddening symptoms mainly due to infection with carrot red leaf virus and the presence of carrot mottle virus must be detected by manual inoculation e.g. to Nicotiana clevelandii.Propagation species
- For studies on virus properties and purification the virus may be propagated in Nicotiana clevelandii. For long term maintenance, virus isolates are best kept in carrot together with carrot red leaf virus, and transferred by means of the aphid vector.
Strains
No strains have been distinguished but isolates differ somewhat in virulence, and tend to become milder when propagated continuously in Nicotiana clevelandii in the glasshouse.
Transmission by Vectors
Transmitted by the carrot-willow aphid, Cavariella aegopodii, but not by C. pastinacae, C. theobaldi, Myzus persicae or several other aphid species (Stubbs, 1948; Watson et al., 1964; A. F. Murant & R. A. Goold, unpublished data). In Japan, a virus culture causing a disease thought to be motley dwarf was transmitted by Brachycolus (Semiaphis) heraclei (Komuro & Yamashita, 1956).
In general, Cavariella aegopodii transmits carrot mottle virus only from plants that also contain carrot red leaf virus (Watson et al., 1964), although A. F. Murant & R. A. Goold (unpublished data) noted rare instances of transmission from plants apparently containing carrot mottle virus alone. Carrot mottle virus therefore usually ceases to be aphid- transmissible following manual inoculation or when transferred by means of aphids to plants that are not hosts of carrot red leaf virus. C. aegopodii continues to transmit both viruses for many days after acquisition and retains them after moulting (Stubbs, 1955; Watson et al., 1964). The aphid can acquire both viruses after 30 min access to source plants; aphids given acquisition access feeds of 24 h can inoculate the viruses to test plants in feeds of 2 min (S. El Nagar & A. F. Murant, unpublished data). There is a short latent period and the minimum total time for acquisition and inoculation is about 9 h (Watson et al., 1964).
Cavariella aegopodii carrying carrot red leaf virus cannot transmit carrot mottle virus from a singly infected plant (A. F. Murant, S. El Nagar & R. A. Goold, unpublished data). This behaviour contrasts with that found for some non-persistent viruses of the potyvirus group (Kassanis & Govier, 1971a, 1971b) and with a semi-persistent virus, parsnip yellow fleck (El Nagar & Murant, 1973, and unpublished data). Nor did aphids transmit carrot mottle virus from plants simultaneously infected with anthriscus yellows virus, which assists transmission of parsnip yellow fleck virus by C. aegopodii (Murant & Goold, 1968; 1971). Aphids transmitted carrot mottle and carrot red leaf viruses after injection with haemolymph from aphids carrying both viruses, but it is not known whether either virus multiplies in the vector; aphids injected with partially purified preparations of carrot mottle virus did not transmit (El Nagar & Murant, 1974).
The reason why carrot mottle virus depends on a ‘helper’ virus for transmission by aphids is not known, but a plausible hypothesis (‘heterologous encapsidation’; Rochow, 1972) is that encapsidation of the nucleic acid of the dependent virus by protein consisting partially or completely of the protein of the helper virus confers on the dependent virus the aphid transmission characteristics of the helper.
Transmission through Seed
None found (Stubbs, 1948).
Transmission by Dodder
Not reported.
Serology
Serum from a rabbit injected with a partially purified virus preparation reacted with highly concentrated virus in double diffusion tests in 0.5% agarose gels to produce two lines of precipitate; the antibody titres were 1/8 and 1/16. It is thought that both lines were virus-specific, but because of difficulty in obtaining enough virus to react, this conclusion is tentative (A. F. Murant & R. A. Goold, unpublished data).
Relationships
In the properties and morphology of its particles, carrot mottle virus differs from all other well-characterized plant viruses. In its ability to be transmitted both manually and by aphids in the persistent manner, and in its dependence on a helper virus for transmission by aphids, it resembles tobacco mottle virus (Smith, 1946a, 1946b) and groundnut rosette virus (Okusanya & Watson, 1966; Watson & Okusanya, 1967; Hull & Adams, 1968).
Stability in Sap
The virus loses infectivity when Nicotiana clevelandii leaf extracts in pH 7.0 phosphate buffer are diluted more than 10-3, heated for 10 min at 70°C, stored for 9-24 h at room temperature, frozen and thawed, or freeze-dried. Dimethyl sulphoxide (10%) protects the virus against damage caused by freezing and thawing. Infectivity is abolished by treatment with ether, chloroform, n-butanol, carbon tetrachloride, 1,1,2-trichloro-1, 2,2-trifluoroethane, or sodium deoxycholate (Murant et al., 1969; Murant & Goold, 1969). N. clevelandii leaf extracts in pH 8.8 Tris buffer contain both ‘stable’ and ‘labile’ forms of infectivity. The labile form (probably RNase-sensitive) is destroyed when the pH of the extract is adjusted to 7. Comparison of phenol-treated and buffer extracts showed that the labile form of infectivity predominates early in systemic infection (5-7 days after inoculation) and then declines, while the amount of stable infectivity increases, reaching a maximum after 8-9 days (Murant et al., 1973).
Purification
Partial purification of the virus was achieved by clarification with bentonite followed by chromatography on columns of calcium phosphate (brushite), and sucrose density gradient centrifugation (Murant et al., 1969).
Properties of Particles
When preparations obtained by the above procedure were centrifuged in sucrose density gradients, light-scattering material was distributed throughout the gradients and no specific virus-containing zone was observed. However, the sedimentation behaviour of the infective particles suggested that they had an average sedimentation coefficient of about 270 S and an average buoyant density in CsCl of about 1.15. Their low buoyant density, together with the sensitivity of the infectivity in leaf extracts to ether, chloroform and sodium deoxycholate, suggests that the particles contain lipid (Murant et al., 1969).
Particle Structure
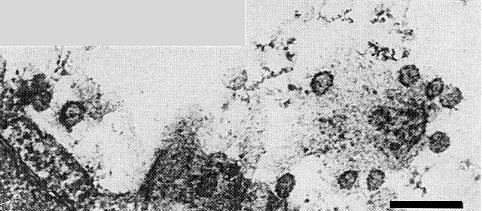
Preparations obtained by the purification procedure described above appeared to consist predominantly of cell membrane components when examined in 2% phosphotungstate, but a few roughly spherical particles c. 52 nm in diameter (Fig. 6), some partially disrupted, were seen in preparations mounted in 2% uranyl acetate. In ultrathin sections of infected Nicotiana clevelandii leaves, particles of about the same size occur in the cell vacuoles associated with the tonoplast (Fig. 7). They are bounded by a unit membrane and some have densely staining central spots. The nature of the nucleocapsid is unknown.
Watson et al. (1964) found particles c. 30 nm in diameter in preparations made from motley-dwarf diseased carrots. Possibly these were particles of carrot red leaf virus; some may even have contained carrot mottle virus RNA if the theory of heterologous encapsidation (Rochow, 1972) is correct.
Particle Composition
Nucleic acid: RNA, single-stranded.
Protein: No information.
Lipid: Probably present in the envelope.
Relations with Cells and Tissues
No virus-like particles or ultrastructural changes were seen in coriander or chervil plants containing both carrot mottle and carrot red leaf viruses (A. F. Murant & I. M. Roberts, unpublished data). However, extensive cytopathological changes occurred in Nicotiana clevelandii leaf tissue systemically infected with carrot mottle virus alone. No virus-like particles were found in the cytoplasm but those found in the cell vacuoles reached their maximum number 8 to 9 days after inoculation and this coincided with the time of maximum infectivity of leaf extracts in pH 7 buffer. Beginning at 6 days after inoculation, tubules appeared in the cytoplasm, associated with the plasmodesmata. Later the tubules, some of which became sheathed by cell wall material forming plasmodesmatal outgrowths, extended towards the vacuole, others towards the nucleus, causing invaginations in it. The outgrowths can be seen by light microscopy (Fig. 5). (Murant et al., 1973).
Notes
Reddening of carrots can have a variety of causes. The presence of carrot mottle virus is detected by aphid transmission to coriander and by manual inoculation to Nicotiana clevelandii and Chenopodium quinoa. Carrot mottle virus is the only virus found in carrots that is persistent in its aphid vector and also transmissible by manual inoculation of sap.
Acknowledgements
Photographs: Scottish Horticultural Research Institute.
Figures

Effect of motley dwarf disease (carrot mottle virus + carrot red leaf virus) on carrots. Upper two plants are healthy.

Plasmodesmatal outgrowths seen by light microscopy in a palisade cell from a systemically infected leaf of N. clevelandii, 9 days after the plant was inoculated.
References list for DPV: Carrot mottle virus (137)
- Anon., Rep. Dep. scient. ind. Res. N.Z. 1959, 112 pp., 1959.
- El Nagar & Murant, Rep. Scott. hort. Res. Inst., 1972: 66, 1973.
- El Nagar & Murant, Rep. Scott. hort. Res. Inst., 1973: 66, 1974.
- Frowd & Tomlinson, Ann. appl. Biol. 72: 177, 1972.
- Heinze, Z. PflKrankh. PflPath. PflSchutz 75: 513, 1968.
- Hull & Adams, Ann. appl. Biol. 62: 139, 1968.
- Kassanis & Govier, J. gen. Virol. 10: 99, 1971a.
- Kassanis & Govier, J. gen. Virol. 13: 221, 1971b.
- Komuro & Yamashita, Ann. phytopath. Soc. Japan 20: 155, 1956.
- Murant & Goold, Ann. appl. Biol. 62: 123, 1968,.
- Murant & Goold, Rep. Scott. hort. Res. Inst., 1968: 52, 1969.
- Murant & Goold, Rep. Scott. hort. Res. Inst., 1970: 53, 1971.
- Murant, Goold, Roberts & Cathro, J. gen. Virol. 4: 329, 1969.
- Murant, Roberts & Goold, J. gen. Virol. 21: 269, 1973.
- Okusanya & Watson, Ann. appl. Biol. 58: 377, 1966.
- Rochow, A. Rev. Phytopath. 10: 101, 1972.
- Smith, Parasitology 37: 21, 1946a.
- Smith, Parasitology 37: 131, 1946b.
- Stubbs, Aust. J. scient. Res. B1: 303, 1948.
- Stubbs, Aust. J. biol. Sci. 5: 399, 1952.
- Stubbs, J. Aust. Inst. agric. Sci. 21: 267, 1955.
- Stubbs, Pl. Dis. Reptr 40: 763, 1956.
- Watson, Pl. Path. 9: 133, 1960.
- Watson & Okusanya, Ann. appl. Biol. 60: 199, 1967.
- Watson & Serjeant, Ann. appl. Biol. 53: 77, 1964.
- Watson, Serjeant & Lennon, Ann. appl. Biol. 54: 153, 1964.
- Wolf, Acta phytopath. Acad. sci. Hung. 5: 95, 1970.
- Wolf & Schmelzer, Zentbl. Bakt. ParasitKde 127: 665, 1972.