Details of DPV and References
DPV NO: 138 July 1974
Family: Virgaviridae
Genus: Pomovirus
Species: Potato mop-top virus | Acronym: PMTV
There is a more recent description of this virus: DPV 389
Potato mop-top virus
B. D. Harrison Scottish Horticultural Research Institute, Invergowrie, Dundee, Scotland
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Calvert & Harrison (1966) and
Harrison & Jones (1970).
A virus with straight tubular particles, occurring in small concentration in tissue extracts, and transmissible by inoculation of sap. The host range is narrow. The soil-borne fungus Spongospora subterranea is a vector. Found in western Europe and South America.
Main Diseases
In potato (Solanum tuberosum), causes a wide range of symptoms, which depend greatly on the cultivar (Calvert, 1968) and the environmental conditions. The three commonest shoot symptoms are yellow blotching or mottling (Fig. 1), particularly of the lower leaves, chlorotic V-shaped markings in the leaflets (Fig. 2) and extreme stunting of the shoots, known as ‘mop-top’ (Fig. 5). Shoot symptoms develop best in cool conditions (5-15°C). In the year of infection from soil the tubers of some cultivars, such as Arran Pilot, develop internal brown arcs (‘spraing’) seen as brown rings on the tuber surface (Fig. 3). The arcs develop at the boundary of the virus-infected tissue in response to a specific change of temperature before or after harvest but do not prevent the virus spreading through the tuber (Harrison & Jones, 1971a). The tubers of other cultivars may develop superficial raised rings without internal symptoms or may show little evidence of infection (Calvert, 1968). In the year following infection from soil, the tubers of some cultivars may be symptomless, or cracked and distorted (Fig. 6), or they may show brown arcs centred on the stolon. When infected tubers are planted, the virus is usually passed to only half, or fewer, of the resulting plants.
Geographical Distribution
Western Europe, Peru, and probably many other countries where potatoes are grown. In Scotland, the prevalence of infection increases with the annual rainfall (Cooper & Harrison, 1973).
Host Range and Symptomatology
Transmitted by inoculation of sap to 26 species in the Solanaceae or Chenopodiaceae and to Tetragonia expansa; species in 11 other families were not infected (Harrison & Jones, 1970). Symptoms are greatly affected by the environmental conditions (Harrison & Jones, 1971b).
-
Diagnostic species
- Chenopodium amaranticolor.
Concentric fine necrotic ringspot lesions develop in inoculated leaves after a week or more at 13-16°C (Fig. 4). A single lesion may spread to cover half a leaf. Not systemic. -
Nicotiana debneyi. Inoculated leaves develop necrotic spots, or necrotic or chlorotic
ringspots. The first systemically infected leaves show chlorotic or necrotic
‘thistle-leaf’ line
patterns
(Fig. 8).
- N. tabacum (tobacco) cv. Xanthi-nc or Samsun NN. Inoculated leaves develop necrotic or chlorotic ringspots below 20°C, but usually no symptom at higher temperatures. Lesion type can be altered by changing the environmental conditions (Harrison & Jones, 1971b). Systemic infection occurs predominantly in the winter months, causing necrotic or chlorotic ‘thistle-leaf’ line patterns, which appear first in a leaf immediately above an inoculated leaf and later in leaves nearer the shoot tip (Fig. 7).
-
Propagation species
- N. tabacum
cv. Samsun NN (inoculated leaves) and N. debneyi can be used for maintaining cultures and as sources of virus.- Assay species
- N. tabacum
cv. Xanthi-nc or Samsun NN is best for isolates that produce necrotic ringspot lesions, and C. amaranticolor for other isolates. - N. debneyi seedlings (systemic symptoms) are useful for testing transmission by vectors (Fig. 8).
Strains
The type strain (isolate T; Harrison & Jones, 1970) produces necrotic lesions in many Nicotiana spp. and is more virulent than most other isolates. Several isolates differing in virulence are reported (Harrison & Jones, 1970).
Transmission by Vectors
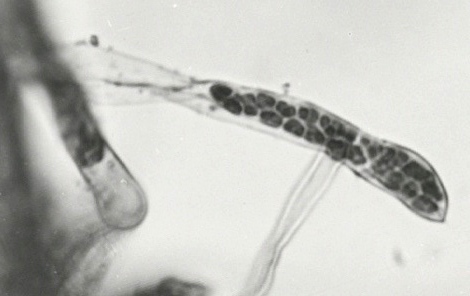
The only vector known is the plasmodiophoromycete fungus, Spongospora subterranea (Fig. 9). The virus is carried, apparently internally, in the resting spores, in which it persists for at least 2 years; transmission to roots is by zoospores released by virus-carrying cultures of S. subterranea. Virus-free S. subterranea zoospores fail to acquire transmissible virus when exposed to a virus suspension. Field soil is best tested for infectivity by air-drying the soil at 20°C for 2 weeks, then moistening it and planting N. debneyi bait seedlings (Jones & Harrison, 1969).
Transmission through Seed
No reports.
Transmission by Dodder
No reports.
Serology
The virus seems moderately immunogenic but difficulty may be experienced with most isolates in obtaining enough virus for antiserum production. Precipitin tests in mixed liquids can be used but some virus preparations precipitate with normal serum (Kassanis, Woods & White, 1972).
Relationships
A distant serological relationship to tobacco mosaic virus is reported (Kassanis et al., 1972). Potato mop-top virus seems also to have affinities with soil-borne wheat mosaic virus (Brakke, 1971); the two viruses have similar particles of two predominant lengths and are transmitted in similar ways by plasmodiophoromycete vectors.
Stability in Sap
In tobacco sap, the thermal inactivation point (10 min) is 75 to 80°C and dilution end-point from 10-2 to 10-4. In sap at 20°C, the virus loses most of its infectivity in 1 day but retains a little for 10 weeks.
Purification
Kassanis et al. (1972). For partial purification, centrifuge sap for 20 min at 9000 g and resuspend pellets in 0.5-1 vol. 0.5 M borate buffer at pH 7.5. Clarify by centrifuging 3 min at 9000 g, emulsify supernatant fluid with 1 vol. diethyl ether, centrifuge at low speed and emulsify aqueous phase with 1 vol. carbon tetrachloride, then centrifuge at low speed. Concentrate virus from aqueous phase by centrifuging for 2 h at 100,000 g and resuspending in a small volume of water, keeping 24 h at 4°C before clarifying at 9000 g. Colour may be removed from preparations by emulsifying with ethylene dichloride.
Properties of Particles
Sedimentation coefficients (s20, w) of components in partially purified preparations were 126, 171 and 236 S. The 236 S component may consist of dimers of 171 S particles (Kassanis et al., 1973). Infectivity was associated with the longer (300 nm) particles.
Particle Structure
Particles are straight, helically constructed, with a pitch of 2.4-2.5 nm and a hollow core. Particle width is 18-20 nm and length usually 100-150 or 250-300 nm. The helix is loosely coiled at the ends of many particles in sap (Fig. 10) (Harrison & Jones, 1970; Kassanis et al., 1972).
Particle Composition
Nucleic acid: Probably single-stranded RNA.
Protein: Polyacrylamide gel electrophoresis of coat protein revealed one polypeptide of M. Wt 18,500-20,000.
Relations with Cells and Tissues
The virus particles aggregate in sheaves in the cytoplasm of tobacco cells (Fig. 11; White, Kassanis & James, 1972). The pattern of systemic invasion of test plants suggests that the virus may move from cell to cell in parenchyma tissue and not through sieve tubes.
Notes
Symptoms induced by potato mop-top virus in potato resemble those caused by tobacco rattle virus, although most potato cultivars react somewhat differently to the two viruses. Also, the two viruses have different effects on Chenopodium amaranticolor, and potato mop-top virus seems not to infect Phaseolus vulgaris. The vectors of tobacco rattle virus are nematodes (Trichodorus spp.), which are killed by air-drying infested soil.
Potato mop-top virus can be distinguished from other viruses occurring in potato by its particle size and shape, and by the reaction of C. amaranticolor, Nicotiana debneyi, N. tabacum and P. vulgaris. In Scotland, potato seems to be its only important host and the virus survives between potato crops in resting spores of S. subterranea (Jones & Harrison, 1972). The virus is gradually self-eliminating in potato stocks grown on virus-free soil.
Figures

External (above) and internal (below) symptoms of spraing in potato tubers of cv. Arran Pilot. Naturally infected plants.

Lesions consisting of concentric necrotic etched rings in inoculated leaf of Chenopodium amaranticolor.
References list for DPV: Potato mop-top virus (138)
- Brakke, CMI/AAB Descriptions of Plant Viruses 77, 4 pp., 1971.
- Calvert, Rec. agric. Res. Minist. Agric. N. Ire. 17: 31, 1968.
- Calvert & Harrison, Pl. Path. 15: 134, 1966.
- Cooper & Harrison, Pl. Path. 22: 73, 1973.
- Harrison & Jones, Ann. appl. Biol. 65: 393, 1970.
- Harrison & Jones, Ann. appl. Biol. 68: 281, 1971a.
- Harrison & Jones, Ann. appl. Biol. 67: 377, 1971b.
- Jones & Harrison, Ann. appl. Biol. 63: 1, 1969.
- Jones & Harrison, Ann. appl. Biol. 71: 47, 1972.
- Kassanis, Woods & White, J. gen. Virol. 14: 123, 1972.
- White, Kassanis & James, J. gen. Virol. 15: 175, 1972.