Details of DPV and References
DPV NO: 14 June 1970
Family: Tombusviridae
Genus: Unassigned Tombusviridae
Species: Tobacco necrosis virus | Acronym: TNV-?
Isolates previously assigned to this virus are now classified as 2 separate viruses: TNV-A and TNV-D.
Tobacco necrosis virus
B. Kassanis Rothamsted Experimental Station, Harpenden, Hertfordshire, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Smith & Bald (1935),
Bawden (1941),
and
Babos & Kassanis (1963a).
Reviewed by
Kassanis (1964).
Synonym
- Tulip Augusta disease virus (Rev. appl. Mycol. 29: 215)
-
An RNA-containing virus with isometric particles about 26 nm in diameter. Readily transmitted by mechanical inoculation to a wide range of plants, but usually does not infect them systemically. Soil-borne, transmitted by zoospores of the chytrid fungus Olpidium brassicae. Distribution world-wide.
Main Diseases
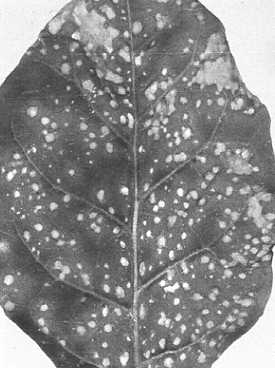
Tulip necrosis (Augusta disease) (Fig. 5) (Kassanis, 1949), bean stipple streak (Fig. 6) (van der Want, 1948), ABC disease of potato tubers (Noordam, 1957) and one form of cucumber necrosis (van Koot & van Dorst, 1955). Also obtained from pear trees showing ring pattern and bark split symptoms (Kegler et al., 1969), from grapevines (Cesati & van Regenmortel, 1969) and from citrus (Bové, personal communication).
Geographical Distribution
World-wide, common in irrigated fields, also in unsterilized soil in glasshouses.
Host Range and Symptomatology
Transmitted experimentally to at least 88 species in 37 dicotyledonous and monocotyledonous families (Price, 1940), usually causing necrotic lesions and rarely infecting systemically. Has been obtained from the naturally infected roots of many species of plants, but infects few species systemically.
-
Diagnostic species
- Phaseolus vulgaris
cv. The Prince (French bean). Symptoms depend on virus strain. Strains of group D (see Relationships) give discrete local lesions (Fig. 2, Fig. 3) whereas those of group A give local lesions that spread along the minor veins (Fig. 1). Some strains do not infect. Only the stipple streak strain infects systemically. - When satellite viruses are present the concentration of tobacco necrosis virus and the size of the lesions are both decreased.
-
Propagation species
- Nicotiana tabacum
(tobacco) (Fig. 4), Phaseolus vulgaris and, for some strains, Nicotiana clevelandii. As the virus causes only local lesions every leaf must be inoculated, with added abrasive, to obtain good yields of virus.Assay species
- Phaseolus vulgaris
or, for some strains, Chenopodium amaranticolor, are good hosts for local lesion assays. In tests for transmission of virus to roots by vectors, Mung bean (Phaseolus aureus), which gives local lesions in roots, lettuce (Lactuca sativa) and tobacco are useful.
Strains
Many strains have been distinguished by their symptomatology and host range, serological specificity and ability to activate different satellite virus strains (see Relationships). They include strains A, B, C, D, E and S (Babos & Kassanis, 1963a); strain C = cucumber necrosis strain of van Koot & van Dorst (1955); strain S = stipple streak strain of van der Want (1948); strains AC36, AC38, AC39 and AC43 (Uyemoto, Grogan & Wakeman, 1968), and the Urbana strain (Lesnaw & Reichmann, 1969).
Variants that seem to exist as free nucleic acid and are transmissible only when phenol or bentonite are used to prepare inocula, have been found (Kassanis & Welkie, 1963).
Transmission by Vectors
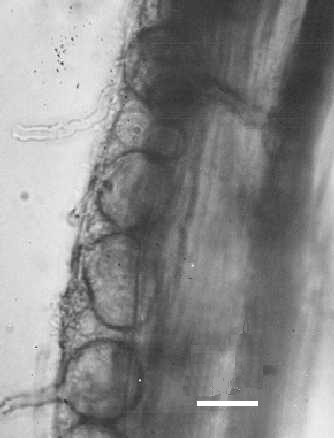
The virus is transmitted by zoospores of the root-infecting chytrid fungus Olpidium brassicae (Fig. 8, Fig. 9) (Teakle, 1962; Teakle & Gold, 1963; Kassanis & Macfarlane, 1964). The virus is quickly acquired by zoospores added to virus suspensions and enters the root at the same time as the fungus. Transmission depends on having a suitable combination of virus strain, fungus race and host species (Kassanis & Macfarlane, 1965). The virus does not survive in the resting spore of the fungus.
Transmission through Seed
None reported.
Transmission by Dodder
None reported.
Serology
Moderately immunogenic. Antisera collected 10 days after a single intravenous injection of virus (2 mg) are more specific than sera collected after a long immunization procedure that includes an intramuscular injection. The virus reacts well in tube tests in which it gives a granular precipitate, and in gel-diffusion tests in which it gives a single band of precipitate.
Relationships
Tobacco necrosis virus is serologically unrelated to satellite virus, which multiplies only in plants that are also infected with tobacco necrosis virus (Kassanis & Nixon, 1961). Strains of tobacco necrosis virus fall into two serological groups or serotypes (Babos & Kassanis, 1963a). Group A contains strains A, B, C and S; Group D contains strains D, E, AC43, AC36, and the strains obtained from pear, grapevine and citrus. The strains are most easily grouped using the specific antisera obtained after a single injection of immunogen (Kassanis & Phillips, 1970).
Groups based on serological tests do not coincide with groups based on symptoms, nor with those based on the ability of strains to support the growth of satellite virus strains, for some strains of group D will only support satellite virus strains SV1 and SV2, whereas others will only support strain SVc; all strains of group A support SV1 and SV2 but not SVc (Kassanis & Phillips, 1970).
The cucumber necrosis strain is not to be confused with the cucumber necrosis virus of McKeen (1959); this and other similar viruses seem unrelated to tobacco necrosis virus (Dias & Douane, 1968). Moreover, cucumber necrosis virus is transmitted by the zoospores of Olpidium cucurbitacearum, but not by those of O. brassicae (Dias, 1969).
Stability in Sap
In sap from infected tobacco the thermal inactivation point is between 85°C and 95°C depending on the virus strain. At 20°C the sap remains infective for many days, and at -20°C for several years. The kinetics of heat inactivation were studied by Babos & Kassanis (1963b).
Purification
The following procedure is useful with most strains of the virus. Frozen leaves or leaf pulp are thawed and triturated in a blender with water. After low speed centrifugation, virus is precipitated from clarified sap by adding ammonium sulphate (0.25 g/ml), resuspending the precipitate in a sufficient volume of water, sedimenting the virus in the ultracentrifuge and then resuspending the pellet in water. Precipitation and sedimentation are repeated until the preparation is clear (usually three cycles), taking care to ensure that most of the virus has been recovered from the pellets. Preparations can be further fractionated by centrifuging in sucrose density gradients.
Properties of Particles
Purified preparations contain one type of particle.
Sedimentation coefficient (s20, w) at infinite dilution: c. 118 S, though smaller and larger values have been reported.
Molecular weight (daltons): c. 7 x 106 (calculated from the RNA content and size).
Isoelectric point: c. pH 4.5; strain B precipitates at pH 4.5 in the presence of salts.
Electrophoretic mobility: -7.4 x 10-5 and -3 x 10-5 cm2 sec-1 volt-1 respectively for strains B and A in 0.066 M phosphate buffer, pH 7.0.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 5.0-5.5.
A260/A280: 1.7 (Cesati & van Regenmortel, 1969).
Buoyant density in CsCl: 1.399 (Cesati & van Regenmortel, 1969).
All strains of the virus, except strain B, readily crystallize.
Particle Structure
Particles are isometric, about 26 nm in diameter (Fig. 7). They may be negatively stained with acid phosphotungstate except for strain B, which is degraded in the stain unless previously treated with 2% formaldehyde for 30 min. Details of structure not known.
Particle Composition
RNA: Probably single-stranded; molecular weight 1.3-1.6 x 106; constitutes about 19% of the particle weight. The reported nucleotide compositions differ (Uyemoto & Grogan, 1969; Lesnaw & Reichmann, 1969): G24; A26-29; C22-23; U24-27 (moles %). s20, w in 0.1 M NaCl: 27 S.
Protein: About 80% of particle weight. Probably only one kind of protein in particle. Molecular weight variously reported as 22,600 (197 residues; Uyemoto & Grogan, 1969) and 33,500 (312 residues; Lesnaw & Reichmann, 1969). The amino acid compositions reported by these authors also differ greatly.
Relations with Cells and Tissues
Virus particles occur in cytoplasm (Kassanis, Vince & Woods, 1970).
Notes
This virus is of academic interest because of its mode of transmission by Olpidium brassicae and its relations with satellite virus, which depends on it for multiplication.
Figures
References list for DPV: Tobacco necrosis virus (14)
- Babos & Kassanis, J. gen. Microbiol. 32: 135, 1963a.
- Babos & Kassanis, Virology 20: 490, 1963b.
- Bawden, Brit. J. exp. Path. 22: 59, 1941.
- Cesati & van Regenmortel, Phytopath. Z. 64: 363, 1969.
- Dias, Proc. Can. phytopath. Soc. 36: 13, 1969.
- Dias & Douane, Can. J. Bot. 46: 47, 1968.
- Kassanis, Ann. appl. Biol. 36: 14, 1949.
- Kassanis, Annls Inst. phytopath. Benaki 6: 7, 1964.
- Kassanis & Macfarlane, J. gen. Microbiol. 36: 79, 1964.
- Kassanis & Macfarlane, Virology 26: 603, 1965.
- Kassanis & Nixon, J. gen. Microbiol. 25: 459, 1961.
- Kassanis & Phillips, J. gen. Virol. 9: 119, 1970.
- Kassanis, Vince & Woods, J. gen. Virol. 7: 143, 1970.
- Kassanis & Welkie, Virology 21: 540, 1963.
- Kegler, Prohl, Schmidt & Opel, Phytopath. Z. 65: 21, 1969.
- Lesnaw & Reichmann,Virology 39: 729, 1969.
- McKeen, Can. J. Bot. 37: 913, 1959.
- Noordam, Tijdschr. PlZiekt. 63: 237, 1957.
- Price, Am. J. Bot. 27: 530, 1940.
- Smith & Bald, Parasitology 27: 231, 1935.
- Teakle, Virology 18: 224, 1962.
- Teakle & Gold, Virology 19: 310, 1963.
- Uyemoto & Grogan, Virology 39: 79, 1969.
- Uyemoto, Grogan & Wakeman, Virology 34: 410, 1968.
- van der Want, Tijdschr. PlZiekt. 54: 85, 1948.
- van Koot & van Dorst, Tijdschr. PlZiekt. 61: 163, 1955.