Details of DPV and References
DPV NO: 144 October 1975
Family: Unallocated ssRNA+ viruses
Genus: Benyvirus
Species: Beet necrotic yellow vein virus | Acronym: BNYVV
There is a more recent description of this virus: DPV 391
Beet necrotic yellow vein virus
T. Tamada Hokkaido Central Agricultural Experiment Station, Naganuma, Hokkaido, Japan
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Tamada et al. (1970) and
Tamada & Baba (1973).
A virus with straight tubular particles of two or three lengths. It has a narrow host range and is transmitted by the fungus, Polymyxa betae, and by inoculation of sap.
Main Diseases
Associated with ‘rhizomania’ disease in sugar beet (Beta vulgaris) (Kanzawa & Ui, 1972). Diseased sugar beet plants show variable amounts of chlorosis, yellowing, necrotic vein-yellowing, crinkling, wilting and stunting. Most of these symptoms seem to be caused by root damage induced by the virus infection, and depend on the environmental conditions. Also causes yellow mottle and stunting in spinach (Spinacia oleracea).
Geographical Distribution
Found in Japan (Hokkaido); possibly occurs in Italy and France.
Host Range and Symptomatology
The virus was transmitted by inoculation of sap to 15 species in the Chenopodiaceae, to Tetragonia expansa (Aizoaceae) and to Gomphrena globosa (Amaranthaceae); 67 species in 16 families were not infected (Tamada & Baba, 1973). The virus tends to be restricted to inoculated leaves of host plants; it often becomes systemic in Beta macrocarpa but rarely in spinach and sugar beet.
-
Diagnostic species
- Beta vulgaris
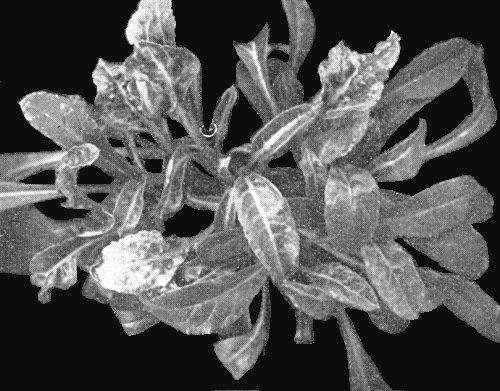
(sugar beet). Inoculated leaves develop chlorotic or yellow lesions 6-8 days after inoculation. The lesions then enlarge and tend to coalesce, spreading along the veins. Few plants are systemically infected; they show chlorotic or yellow spotting, yellow vein-banding, vein necrosis, leaf distortion, wilting and stunting (Fig. 1, Fig. 5). - Beta macrocarpa. Inoculated leaves usually develop chlorotic local lesions,
followed by systemic yellow mottle or yellow flecks with leaf distortion
(Fig. 2).
- Chenopodium amaranticolor, C. quinoa. Chlorotic or necrotic lesions appear in inoculated leaves 5-7 days after inoculation (Fig. 4). Not systemically infected.
- Tetragonia expansa. Chlorotic, yellowish or necrotic spots and rings, or concentric rings develop in inoculated leaves, depending on the virus isolate (Fig. 3). Sometimes infected systemically by YS isolates. This species can be used for distinguishing strains.
- Chenopodium amaranticolor, C. quinoa. Chlorotic or necrotic lesions appear in inoculated leaves 5-7 days after inoculation (Fig. 4). Not systemically infected.
-
Propagation species
- Sugar beet (Beta vulgaris) and Tetragonia expansa (inoculated leaves) can be used for maintaining cultures and as sources of virus.
Assay species
- Chenopodium amaranticolor
is a good local lesion host (Fig. 4).- Sugar beet (Beta vulgaris) seedlings are useful for testing transmission by the vector.
Strains
Isolates have been placed in four groups on the basis of the type of lesions produced in leaves of Tetragonia expansa (Tamada et al., 1975): CR (concentric rings), YS (yellow spots, Fig. 3, right), CS (pale chlorotic spots or rings), and NS (necrotic spots or rings, Fig. 3, left). In a few cases YS isolates infect systemically, causing yellow vein-banding, whereas other isolates always remain localized in the inoculated leaves. Typical yellow vein or necrotic yellow vein symptoms in sugar beet are produced only by YS isolates (Fig. 1, Fig. 5).
Transmission by Vectors
The virus is transmitted by the plasmodiophoromycete fungus, Polymyxa betae (Fig. 8). In naturally affected plants, infection with the virus is correlated with the occurrence of P. betae in the rootlets. The virus is transmitted when suspensions of resting spores or zoospores of the fungus from infected roots are added to the roots of healthy sugar beet seedlings, but not when the virus alone is added. A virus-free isolate of P. betae acquired the virus when grown in plants which were infected with the virus by manual inoculation of sap. The virus seems to be carried internally in the resting spores, because its infectivity is retained for at least 4 years in air-dried soil (Tamada et al., 1975).
Transmission through Seed
Not tested.
Transmission by Dodder
Not tested.
Serology
The virus seems moderately immunogenic. An antiserum against the virus, made using preparations partially purified by organic solvent clarification and polyethylene glycol precipitation, had a titre of 1/1024 in ring precipitin tests. Reactions have also been obtained in agar-gel diffusion tests (Tamada & Baba, 1973).
Relationships
The virus is morphologically similar to other viruses with tubular particles, such as tobacco mosaic virus. In many respects, beet necrotic yellow vein virus also resembles soil-borne wheat mosaic virus (Brakke, 1971) and potato mop-top virus (Harrison, 1974); these three viruses have particles of two or three predominant lengths and are transmitted in similar ways by plasmodiophoromycete vectors. No serological relationship was detected between beet necrotic yellow vein virus and tobacco mosaic, tobacco rattle or soil-borne wheat mosaic viruses (Tamada & Baba, 1973).
Stability in Sap
In sugar beet sap, the thermal inactivation point (10 min) is 65 to 70°C and dilution end-point about 10-4. Infectivity is retained for 5 days at 20°C and for 8 days at 4°C. Infectivity of sap extracts is decreased greatly by freezing (Tamada & Baba, 1973).
Purification
No purification procedure has been completely successful, because of aggregation of the particles. Partially purified virus for serological tests was obtained using the following procedure (Tamada & Baba, 1973). Grind fresh leaves of sugar beet in 0.1 M phosphate buffer, pH 7.0, containing 1.0% 2-mercaptoethanol. Clarify the extracts with one-half volume of carbon tetrachloride and precipitate the virus by adding polyethylene glycol (M.Wt 6000) to 4%. After low-speed centrifugation, clarify the resuspended preparations with fluorocarbon (Diafron S-3) and then concentrate by further precipitation with polyethylene glycol, resuspending the virus in a small volume of 0.01 M phosphate buffer, pH 7.0, or water.
Properties of Particles
No information.
Particle Structure
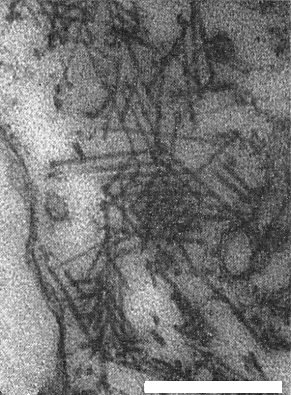
Particles are straight rods, helically constructed, and have a central canal. They are about 20 nm wide and of two or three predominant lengths, usually about 390 nm (long particles), about 270 nm (intermediate particles), and 65-105 nm (short particles) (Fig. 6). Isolate CR contains only long and intermediate particles. The short particles of isolate YS are about 105 nm long, those of isolate CS about 65 nm, and those of isolate NS about 65 nm and 90 nm long. Each isolate has more short particles than long and intermediate ones (Tamada et al., 1975).
Particle Composition
Not determined.
Relations with Cells and Tissues
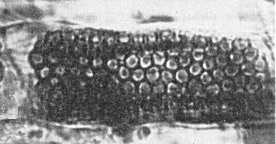
The virus is found in the cells of roots, stems and leaves of systemically infected sugar beet plants. Virus particles are found scattered in the cytoplasm or in angled-layer aggregates (Fig. 7). Virus-like particles are seen in sections of P. betae zoospores (Fig. 9).
Notes
A ‘rhizomania’ disease of sugar beet has been a serious problem in Italy since the mid 1950s, and is characterized by abnormal proliferation of rootlets (Bongiovanni & Lanzoni, 1964; Ui, 1973). The disease is quite similar to that found in Japan (Kanzawa & Ui, 1972; Ui, 1973). Canova (1966) suggested that the Italian disease is caused by mixed infection with Polymyxa betae and a virus, and Faccioli & Giunchedi (1974) recently isolated a tubular virus from diseased plants. Characteristics of the virus are very similar to those of beet necrotic yellow vein virus. A similar disease has recently been reported in France (Putz & Vuittenez, 1974). In Japan, rhizomania disease results from root damage induced by infection with beet necrotic yellow vein virus, and is not induced by P. betae, the vector of the virus (Tamada et al., 1975; H. Abe, unpublished data). The virus is usually restricted to the roots of sugar beet plants grown in infested soil. It is readily detected by examination of root-dip preparations by electron microscopy (Tamada & Baba, 1973).
Symptoms, host range and some properties of beet necrotic yellow vein virus are quite similar to those of beet yellow vein virus (Ruppel & Duffus, 1971). However, the mode of transmission and particle morphology of the latter virus are not known.
Figures
References list for DPV: Beet necrotic yellow vein virus (144)
- Bongiovanni & Lanzoni, Progr. agric., Bologna 10: 209, 1964.
- Brakke, CMI/AAB Descriptions of Plant Viruses 77, 4 pp., 1971.
- Canova, Inftore fitopatol. 16: 235, 1966.
- Faccioli & Giunchedi, Phytopath. medit. 13: 10, 1974.
- Harrison, CMI/AAB Descriptions of Plant Viruses 138, 4 pp., 1974.
- Kanzawa & Ui, Ann. phytopath. Soc. Japan 38: 434, 1972.
- Putz & Vuittenez, Annls Phytopath. 6: 129, 1974.
- Ruppel & Duffus, Phytopathology 61: 1418, 1971.
- Tamada & Baba, Ann. phytopath. Soc. Japan 39: 325, 1973.
- Tamada, Kanzawa & Ui, Ann. phytopath. Soc. Japan 36: 365 (abstr.), 1970.
- Tamada, Abe & Baba, Proceedings of the First Intersectional Congress of IAMS, Tokyo, 1974, 3: 313, 1975.
- Ui, Proceedings of Sugar Beet Research, Japan 15: 233, 1973.