Details of DPV and References
DPV NO: 145 October 1975
Family: Potyviridae
Genus: Bymovirus
Species: Oat mosaic virus | Acronym: OMV
Oat mosaic virus
T. T. Hebert Department of Plant Pathology, N. Carolina State University, Raleigh, N. C. 27607, USA
C. H. Panizo Department of Plant Pathology, N. Carolina State University, Raleigh, N. C. 27607, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by Atkinson (1945) and McKinney (1946).
Selected synonyms
- Soil-borne oat mosaic virus (Rev. appl. Mycol. 25: 554)
- Marmor terrestre (Rev. appl. Mycol. 25: 554)
-
A soil-borne virus with flexuous, rod-shaped particles about 600-750 nm long and 12-14 nm wide. It is mechanically transmissible but with difficulty, Host range is restricted to Avena species. It is transmitted by the fungus Polymyxa graminis, and occurs in the USA, UK, and possibly New Zealand.
Main Diseases
Oat mosaic virus causes a disease of autumn-sown oats. The field symptoms vary with the oat cultivar, virus strain, level of infestation of the soil and with environmental conditions. The mottling symptoms are most evident in the first flush of growth in the spring and tend to be masked with further growth and warmer weather. Stunting is the principal symptom seen late in the season. On heavily infested soil, yields may be reduced 25 to 50% in tolerant cultivars and most plants of very susceptible cultivars produce no grain.
Geographical Distribution
Occurs in south-eastern USA from Maryland to Florida and Alabama (McKinney, 1946; Toler & Hebert, 1964). It has been reported from north-western USA (Bruehl & Damsteegt, 1961), England (Macfarlane et al.,1968) and Wales (Catherall & Hayes, 1970). A mosaic of oats in New Zealand may be caused by the same virus (Slykhuis, 1962).
Host Range and Symptomatology
Hosts are apparently confined to the genus Avena. Toler & Hebert (1963) reported that all of 21 oat cultivars and 8 other Avena species were infected following mechanical inoculation but wheat, barley, rye, maize, rice, and 73 species in 19 other plant families (4 monocotyledonous and 15 dicotyledonous) were not infected. McKinney (1946), however, reported that 2/50 plants of Michigan Amber wheat developed mosaic symptoms. Bruehl & Damsteegt (1961) and Catherall & Hayes (1970) failed to infect cereals (other than oats), several pasture grasses, and several dicotyledonous species.
-
Diagnostic species
- Avena sativa
(oat). All cultivars are susceptible to infection by mechanical inoculation and when grown in infested soil. However, some are more easily infected than others by mechanical inoculation and the percentage of plants thus infected and the intensity of the resulting symptoms are not good indications of the field tolerance of the cultivar (Toler & Hebert, 1963). The air-brush or spray-gun methods of inoculation are more effective than leaf rubbing (Toler & Hebert, 1965; Catherall & Hayes, 1970). Symptom expression is best at 15-18°C, with few or no symptoms above 24°C. Symptoms usually appear about 2 weeks after inoculation but may appear after only 10 days or not until 2 months. Growing test plants at 25-30°C or in darkness for 1-2 days prior to inoculation increases the proportion of plants infected.The earliest symptoms in mechanically inoculated plants appear in the youngest leaf as it emerges from the whorl. At this stage the young leaf shows light green streaks or dashes, largely confined to the interveinal areas. As the leaf emerges fully and expands, the light green areas become longer and some expand laterally across 2 or 3 veins to produce a mosaic pattern of light and dark green areas (Fig. 5). Further development varies with the virus strain, cultivar, and environmental conditions (see Strains).
Propagation species
- Avena sativa
(oat).Assay species
- Avena sativa
(oat). No local lesion hosts known. Assays are based on the proportion of plants becoming systemically infected.Tests for soil transmission may be conducted outdoors by planting oats in autumn in soils suspected of carrying the virus, and in appropriate control soils, and observing the symptoms in the spring. Alternatively, about two months after sowing, the plants may be moved to a glasshouse at 15 to 18°C where symptoms usually develop within a week or two. In controlled environments, optimum conditions for transmission by Polymyxa graminis are 16 hour periods of darkness at 13°C alternating with 8 hour periods of light (10,000 lux) at 17°C (Panizo, 1975).
Strains
Apical mosaic strain: Faint light green to yellow streaks, spots and patches tend to occur in the distal portions of the upper one to three leaves of the plant (Fig. 1). Symptoms later tend to disappear.
Eyespot strain. Produces fusiform spots with green centres and light green or yellowish to
ash-grey borders
(Fig. 2). The eyespot symptoms are more prominent in leaves that have completed growth
and are more persistent than the apical mosaic symptoms.
McKinney (1946) described the above two strains and noted that the apical mosaic strain predominated in south-eastern USA. In contrast, Catherall & Hayes (1970) found more plants with eyespot symptoms than with apical mosaic symptoms in Wales.
Transmission by Vectors
Apparently transmitted by Polymyxa graminis (Panizo, 1975). Infectivity of soils is not affected by drying. In infested soil that had been treated at different temperatures there was a good correlation between infection of the plants with P. graminis and infection with the virus. In other tests, oat seedlings inoculated with small pieces of root (about 5 mm long) from plants infected with oat mosaic virus and also containing resting spores of P. graminis (Fig. 3) developed mosaic symptoms. No virus symptoms developed in control plants inoculated with adjacent pieces of the same virus-infected roots, equal to or larger in size, but containing no resting spores of P. graminis.
Transmission through Seed
The virus is not transmitted through seed. A’Brook (1971) found virus particles in seed from plants grown in infested soil but plants grown from this seed were not infected by the virus.
Transmission by Dodder
No reports.
Serology
Attempts to produce an antiserum have not been successful and the virus seems to be not strongly immunogenic (T. T. Hebert, unpublished results).
Relationships
Not known to be related serologically to any other virus. Partially purified preparations of oat mosaic virus and crude juice from infected plants failed to react with antiserum to soil-borne wheat mosaic, maize dwarf mosaic, barley stripe mosaic, potato Y, tobacco etch, and peanut mottle viruses (T. T. Hebert, unpublished results). Edwardson (1974) placed oat mosaic virus in the potato virus Y group, but a soil-borne virus with a fungus vector seems out of place in this group. Its closest affinities should probably be sought among other soil-borne viruses with flexuous rod-shaped particles, e.g. wheat spindle streak mosaic virus (Slykhuis, 1970) and the viruses of rice necrosis mosaic, barley yellow mosaic, and wheat yellow mosaic found in Japan (Inouye, 1968a, 1968b).
Stability in Sap
In oat sap, thermal inactivation occurs after 10 min at 44-46°C, and longevity in vitro is between 24 and 48 h at 20°C; the dilution end-point is between 1/1000 and 1/5000 using the air-brush method of inoculation, or between 1/100 and 1/1000 using the leaf rubbing method (Toler & Hebert, 1964).
Purification
The virus has not been highly purified. It has a strong tendency to aggregate and the concentration in the plant appears to be low. Partially purified preparations were prepared by extracting leaves in 0.2 M citrate or phosphate buffer at pH 7.5, clarifying with chloroform, passing the aqueous phase through a column of 2% agar granules, sedimenting the eluted particles by centrifugation and resuspending the pellet in 0.01 M borate buffer at pH 8.3. Butanol inactivated much of the virus and varying the molarity of the extracting buffer from 0.01 M to 0.5 M did not seem greatly to affect the success of purification (T. T. Hebert, unpublished results).
Properties of Particles
No information.
Particle Structure
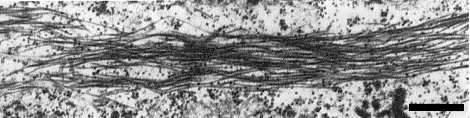
Flexuous rod-shaped particles about 12 to 14 nm wide and 600 to 750 nm long are found in infected plants (Fig. 6). The ‘normal’ or ‘modal’ length has not been determined. A wide range of particle lengths is commonly observed in leaf dips. Some of the reported dimensions (in nm) are: 660 (mean length) x 14 (Gold et al.,1959), 600-700 x 14 (Watson, 1968), 600-700 x 12 (Macfarlane et al.,1968), 700-750 x 12 (Catherall & Hayes, 1970), and 600-700 x 12 (A’Brook, 1971).
Particle Composition
No information.
Relations with Cells and Tissues
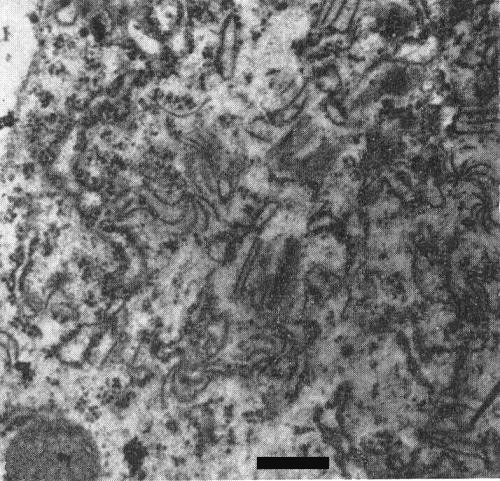
Virus-infected mesophyll cells contain cylindrical inclusions with pinwheel cross-sections (Fig. 4) and bundles or parallel aggregates of virus particles (Fig. 7). Few free virus particles are seen in infected cells. Microbodies, chloroplasts, nuclei, and mitochondria of infected cells deteriorate in that order and the cells become filled with the remnants of these organelles (Smith, 1973).
Notes
Antiserum to oat mosaic virus is not available and identification must be based on other properties. Mechanical transmissibility distinguishes this virus from barley yellow dwarf virus and from the leaf-hopper-transmitted viruses that infect oats. It is distinguished from ryegrass mosaic, oat necrotic mottle and cocksfoot streak viruses by its inability to infect Lolium multiflorum; from barley stripe mosaic, wheat streak mosaic and brome mosaic viruses by its inability to infect barley; and from tobacco rattle and tomato black ring viruses by its inability to infect tobacco or Chenopodium amaranticolor. In addition, its loss of infectivity after 10 min at 46°C distinguishes it from most if not all viruses mechanically transmissible to oats. In being soil-borne it resembles tobacco rattle and tomato black ring viruses, but it may be distinguished from these by its ability to survive (in the vector) for a long time in dry soil.
The properties most useful in identifying oat mosaic virus are symptoms in oats, transmission from dry soil, host range limited to Avena species, presence of characteristic virus particles in infected plants, and low thermal inactivation point. Additional evidence for the identity of this virus may be obtained by determining the reactions of oat cultivars in infested soil. Among the cultivars most susceptible to oat mosaic virus are Tech C.I. 947, Stanton C.I. 3855, Mustang C.I. 4660 and Fulwin Composite Sel. C.I. 6993 (Toler & Hebert, 1963). Most of the tolerant cultivars appear to derive their tolerance from a single source, Fulghum C.I. 699, and include Ballard C.I. 6980, Arlington C.I. 4657, Excel C.I. 7603, and Early Ballard C.I. 7622 (Coffman et al.,1963).
Figures
References list for DPV: Oat mosaic virus (145)
- A’Brook, Rep. Welsh Pl. Breed. Stn 1970: 42, 1971.
- Atkinson, Pl. Dis. Reptr 29: 86, 1945.
- Bruehl & Damsteegt, Pl. Dis. Reptr 45: 884, 1961.
- Catherall & Hayes, Pl. Path. 19: 78, 1970.
- Coffman, Hebert, Gore & Byrd, Pl. Dis. Reptr 47: 54,1963.
- Edwardson, Monogr. Florida agric. Exp. Stn 4, 398 pp., 1974.
- Gold, McKinney & Scott, Proc. 4th int. Cong. Crop Protection, Hamburg 1957 1: 351, 1959.
- Inouye, Nogaku Kenyu 52: 31, 1968a.
- Inouye, Ann. phytopath. Soc. Japan 34: 301, 1968b.
- MacFarlane, Jenkins & Melville, Pl. Path. 17: 167, 1968.
- McKinney, Phytopathology 36: 359, 1946.
- Panizo, Ph.D. Thesis, N. Carolina State Univ., 1975.
- Slykhuis, Pl. Prot. Bull. F.A.O. 10: 1, 1962.
- Slykhuis, Phytopathology 60: 319, 1970.
- Smith, Ph.D. Thesis, N. Carolina State Univ., 1973.
- Toler & Hebert, Pl. Dis. Reptr 47: 58, 1963.
- Toler & Hebert, Phytopathology 54: 428, 1964.
- Toler & Hebert, Pl. Dis. Reptr 49: 553, 1965.
- Watson, Rep. Rothamsted exp. Stn 1967: 126, 1968.