Details of DPV and References
DPV NO: 146 October 1975
Family: Potyviridae
Genus: Potyvirus
Species: Pea seed-borne mosaic virus | Acronym: PSbMV
Pea seed-borne mosaic virus
R. O. Hampton Agriculture Research Service, US Department of Agriculture, Department of Botany and Plant Pathology, Oregon State University, Corvallis, Oregon 97331, USA
G. I. Mink Washington State University, Irrigated Agriculture Research and Extension Center, Prosser, Washington 99350, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Disease described by Musil (1966) and Inouye (1967); virus partially
characterized by Knesek et al. (1974).
- Selected synonyms
- Pea leaf-rolling virus (Rev. appl. Mycol. 45: 2293)
- Pea fizzletop virus (Rev. Pl. Path. 49: 5m)
- Pea leafroll mosaic virus (Rev. Pl. Path. 49: 2012)
- Pea leaf-rolling mosaic virus (Rev. Pl. Path. 50: 372)
- Pea fizzletop virus (Rev. Pl. Path. 49: 5m)
- An RNA-containing virus with filamentous particles c. 770 x 12 nm. Transmissible by mechanical inoculation of sap, in the non-persistent manner by aphids, and through seed. Disseminated primarily through infected pea seed but transmissible to several other plant species. A typical, apparently distinct member of the potyvirus group. Reported from the USA, Japan, Holland, and Czechoslovakia.
Main Diseases
Causes various degrees of stunting, downward rolling of leaflets, and a transient clearing and swelling of leaf veins of most cultivars of Pisum sativum. Infected plants may produce distorted flowers, which often give rise to small distorted pods. Ovule development in affected pods may be uneven, with only one or two seeds produced. Seed coats may split as the seeds mature (Fig. 1, Fig. 2, Fig. 4).
Geographical Distribution
Reported from the USA (Mink et al., 1969; Hampton, 1969; Stevenson & Hagedorn, 1969), Japan (Inouye, 1967), Czechoslovakia (Musil, 1966), and the Netherlands (Bos, 1970). Possibly distributed world-wide, because seeds of many cultivars known to be infected have been exchanged internationally.
Host Range and Symptomatology
Pisum cultivars are the main natural hosts, but the virus can infect 47 species in 12 dicotyledonous families (Aapola et al., 1974). Most non-leguminous hosts are infected without producing symptoms.
- Diagnostic species
- Pisum sativum
(pea). Transient clearing and swelling of veins on the first leaflets produced after infection, followed by downward rolling of leaflets and tight curling of tendrils. Progressive shortening of internodes, often producing a terminal rosette. These symptoms can be mistaken for physiological or genetic disorders. Plants infected early or arising from infected seed produce no flowers or a few distorted flowers and pods (Hampton, 1972) (Fig. 1, Fig. 2, Fig. 4). - Vicia faba (broad bean). Systemic dark and light-green zonal leaf
mottle, slight to moderate downward rolling of leaf margins. Distortions of leaf
shape associated with mottle patterns (Hampton & Baggett, 1970) (Fig. 3).
- Chenopodium amaranticolor and C. quinoa. Necrotic local lesions (Fig. 5) and chlorotic local lesions, respectively. No systemic symptoms or infection.
- Chenopodium amaranticolor and C. quinoa. Necrotic local lesions (Fig. 5) and chlorotic local lesions, respectively. No systemic symptoms or infection.
- Propagation species
- Most Perfection-type pea cultivars are good sources of inoculum and of virus for purification.
- Assay species
- C. amaranticolor.
Strains
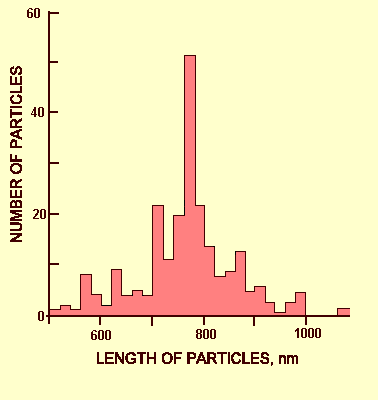
None described, but differences in particle length measurements (Musil, 1970; Hampton et al., 1974) and in ultrastructural cytology (Hampton et al., 1973) suggest that strains exist.
Transmission by Vectors
Transmissible in a non-persistent manner by the aphids Acyrthosiphon pisum, Dactynotus escalanti, Macrosiphum euphorbiae, M. rosae, Myzus persicae, Ovatus crataegarius, Rhopalosiphum padi, (Aapola & Mink, 1973), Aphis craccivora and A. fabae (Kvicala & Musil, 1967), but not by Periphyllus lyropictus (Aapola & Mink, 1973). Aphids acquire and inoculate in 10-90 sec feeding periods; no latent period is required (Gonzales & Hagedorn, 1970).
Transmission through Seed
Transmitted through up to 30% of the seed from Pisum sativum plants infected prior to flowering but not transmitted through seed from plants infected after flowering begins (J. E. Knesek & G. I. Mink, unpublished data). Commercial seed lots containing up to 90% infected seed were found (Knesek & Mink, 1970). Transmission was 8 times more frequent through seeds with split seed-coats than through normal seeds (Stevenson & Hagedorn, 1970). Seed transmission through seeds with split seed-coats, among 38 pea cultivars, varied from 0 to 100% (Stevenson & Hagedorn, 1973). Less than 1% of the seeds became infected as the result of pollination with pollen from an infected parent plant (Stevenson & Hagedorn, 1973). Transmitted through a low percentage of seeds of Vicia articulata, V. narbonensis and V. pannonica.
Serology
The virus is a moderately good immunogen: antisera obtained from rabbits given five intravenous injections of c. 0.2 mg virus had homologous titres of 1/1024 in microprecipitin tests (Knesek et al., 1974). Purified virus reacts with antisera in ring interface tests and in gel-diffusion tests. Chloroplast agglutination reactions have been obtained between sap of infected pea tissue and some, but not all, antisera.
Relationships
Isolates from the USA and Japan were found to be serologically related (Mink et al., 1974). Some antiserum preparations reacted to purified bean yellow mosaic virus in controlled microprecipitin tests, but not in microslide gel-diffusion tests. Relationships to other legume-infecting potyviruses not determined.
Stability in Sap
Juice from pea leaf or root tissue is highly infective when initially prepared. Extracts from roots remain infective for more than 96 h, whereas undiluted leaf extracts become non-infective within 24 h at room temp. Leaf extracts diluted 10-1 or more remain infective for more than 96 h (Knesek et al., 1974). The dilution end-point is between 10-3 and 10-4. Only traces of infectivity remain after 10 min at 55°C. Infectivity is lost on freezing.
Purification
Knesek et al. (1974): Homogenize 18 g freshly harvested leaf or root tissue in 180 ml freshly prepared buffer containing 0.01 M sodium diethyldithiocarbamate (NaDIECA), 0.01 M cysteine-HCl, and (for leaf tissue only) 0.01 M ethylenediamine-tetraacetic acid (EDTA). Express juice through cheesecloth, incubate for 1 h at 30°C, then emulsify for 30 min at 30°C with 0.5 vol chloroform. Centrifuge the emulsion for 30 min at 3000 g, retain the upper phase and centrifuge for 1.5 h at 90,000 g. Resuspend each pellet overnight in 4 ml cold distilled water adjusted to pH 7.0 with 0.005 M NaOH. Clarify suspension by centrifuging for 15 min at 10,000 g. Layer 20 ml of the supernatant fluid in Beckman SW25 rotor tubes over 10 ml 30%. sucrose containing 4% polyethylene glycol (PEG, M. Wt 6000) and 0.12 M NaCl. After 2 h at 24,000 rpm, resuspend the pellets overnight in 2.5 ml 2% sucrose containing 0.1% Igepon T73 at pH 7.0, and subject the virus to a second cycle of centrifugation through sucrose/PEG. When centrifuged in rate zonal density gradients the virus forms a single highly infective visible band. This procedure yields 1 to 1.5 mg virus from 18 g fresh tissue.
Properties of Particles
Preparations usually contain one sedimenting component with a sedimentation
coefficient (s°20, w) of 154 S and buoyant density
in CsCl of 1.329 g/cm3 (Huttinga, 1975).
A260/A280: 1.14-1.18.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 2.5.
Particle Structure
Particles (Fig. 6) are slightly flexuous filaments c. 770 x 12 nm. Some isolates appear highly susceptible to breakage unless fixed with 3.5% glutaraldehyde (Hampton et al., 1974).
Particle Composition
Nucleic acid: 5.3 ± 1% of the particle weight. RNA, presumably single-stranded. Molar percentage of nucleotides, G 22.8; A 44.0; C 17.6; U 15.6 (Knesek et al., 1974).
Protein: About 94% of the particle weight. Subunit M.Wt 34,000 (Huttinga, 1975). Relative amino acid molar ratio: Ala 3.3, Arg 2.5, Asx 4.7, Glx 4.9, Gly 2.7, His 1.0, Iso 1.8, Leu 2.2, Lys 1.5, Met 1.9, Phe 1.1, Pro 1.4, Ser 2.2, Thr 2.0, Tyr 1.2, Val 2.4, Cys and Try not determined (Knesek et al., 1974).
Relations with Cells and Tissues
Most isolates induce pinwheel inclusions (Inouye, 1971) in the cytoplasm of mesophyll cells of Pisum sativum and Vicia faba (Hampton et al., 1973). Less common isolates induce tonoplast aggregates or dense bodies and laminated aggregates. One isolate characterized by induction of tonoplast aggregates also induced formation of convoluted endoplasmic reticulum (Hampton et al., 1973) (Fig. 7, Fig. 8). Masses of virus-like particles occur in pea root parenchyma cytoplasm.
Notes
The following features distinguish other pea viruses from pea seed-borne mosaic virus. Pea mosaic virus (Doolittle & Jones, 1925), although a potyvirus, typically induces striking mosaic patterns in leaves of infected plants, with little stunting, and is rarely seed-borne. Pea early browning virus (Bos & Van der Want, 1962) is a tobravirus, induces stem, petiole, leaf and pod necrosis, and is transmitted in nature both by seed and by nematodes (Trichodorus spp.). False pea leaf roll virus (Thottappilly & Schmutterer, 1968) (no report on particle morphology) induces leaf rolling as well as chlorotic and necrotic spots and vein necrosis of leaves and is reportedly seed-borne, soil-borne and aphid-borne (Myzus persicae). Bean leaf roll virus (Quantz & Volk, 1954) (pea leaf roll virus, pea tip-yellowing virus, pea top yellows virus, pea yellows virus) (no report on particle morphology) induces various degrees of plant stunting, erect plant habit, sieve-tube necrosis, and chlorosis of apical leaves, is transmissible by aphids (Myzus persicae), but not mechanically or through the seeds of pea. The disease it induces in pea resembles that caused in Australia by subterranean clover stunt virus (Smith, 1966).
Pea seed-borne mosaic virus has appeared abruptly in pea breeding programs in the USA and elsewhere. This appearance is attributable to the international exchange of infected seed-lots, particularly for breeding. Symptoms induced by this virus have been mistaken by breeders as plant variants or mild physiological disorders. Symptoms may be mild, are unlike classical virus symptoms, and tend to fade as plants approach flowering (Hampton, 1972). The disease seriously threatens pea production, jeopardizes international shipments of pea seed, and may exist unrecognised in breeding lines from which it may be spread to healthy lines by aphid vectors.
Virus-infected seeds may or may not have split seed-coats. A low incidence of growth-cracking is normal for the seeds of many pea cultivars. Drought during seed development and other factors may affect the proportion of split seed-coats produced by virus-free or infected plants.
The virus may be present without inducing symptoms in 5 to 10% of the plants from infected seed-lots (Hampton, 1972). Moreover, the virus may not be detectable in symptomless plants by assay on C. amaranticolor until 8 to 10 weeks after emergence. Most infected seed, however, gives rise to seedlings showing some stunting, leaf-roll and vein-swelling.
Figures

Appearance of Perfection-type pea with severe leaf roll symptoms (left), moderate leaf roll (centre); healthy plant (right). Note apparent sparsity of foliage caused by reduced leaf size and leaf roll.

Close-up of severely affected pea plant, showing downward rolling of lateral leaf margins, an abortive phyllody-type flower (AF), and a young distorted pod (P). Inset: seeds from infected plants, showing (above) characteristic split seed-coats (growth cracking), (below) seeds from healthy plants.

Characteristic symptoms induced in broad bean (Vicia faba), including zonal-mottle, downward rolling and distortions of leaf margins. Healthy leaf at bottom.
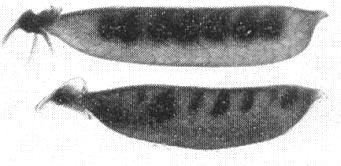
Incomplete and uneven ovule development in pod from an infected pea plant (bottom) and complete development in pod from healthy plant (top).

Characteristic lesions induced in leaves of Chenopodium amaranticolor, with necrotic centres and translucent haloes.

Electron micrograph of a thin section of infected pea mesophyll parenchyma cell, showing aggregates of virus-like particles inside tonoplast (TA), convoluted endoplasmic reticulum (ERc), and a tangential section through pinwheel configurations (PW). Bar represents 250 nm.
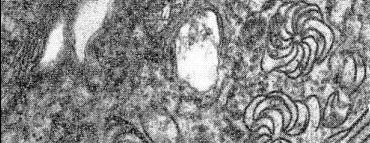
Electron micrograph of a thin section of the same infected pea mesophyll parenchyma cell as in Fig. 7, showing transverse section through a pinwheel.
References list for DPV: Pea seed-borne mosaic virus (146)
- Aapola & Mink, Pl. Dis. Reptr 57: 552, 1973.
- Aapola, Knesek & Mink, Phytopathology 64: 1003, 1974.
- Bos, Neth. J. Pl. Path. 76: 8, 1970.
- Bos & van der Want, Tijdschr. PlZiekt. 68: 368, 1962.
- Doolittle & Jones, Phytopathology 15: 763, 1925.
- Gonzales & Hagedorn, Phytopathology 60: 1293, 1970.
- Hampton, Phytopathology 59: 1029, 1969.
- Hampton, Phytopathology 62: 268, 1972.
- Hampton & Baggett, Pl. Dis. Reptr 54: 355, 1970.
- Hampton, Phillips, Knesek & Mink, Archiv. ges. Virusforsch. 42: 242, 1973.
- Hampton, Knesek & Mink, Phytopathology 64: 1358, 1974.
- Huttinga, Neth. J. Pl. Path. 81: 58, 1975.
- Inouye, Ann. phytopath. Soc. Japan 33: 38, 1967.
- Inouye, Nogaku Kenkyu 53: 189, 1971.
- Knesek & Mink, Pl. Dis. Reptr 54: 497, 1970.
- Knesek, Mink & Hampton, Phytopathology 64: 1076, 1974.
- Kvicala & Musil, Biológia Bratisl. 22: 10, 1967.
- Mink, Kraft, Knesek & Jafri, Phytopathology 59: 1342, 1969.
- Mink, Inouye, Hampton & Knesek, Phytopathology 64: 569, 1974.
- Musil, Biológia Bratisl. 21: 133, 1966.
- Musil, Biológia Bratisl. 25: 379, 1970.
- Quantz & Volk, NachrBl. dt. PflSchutzdienst. Stuttg. 12: 177, 1954.
- Smith, Aust J. agric. Res. 17: 875, 1966.
- Stevenson & Hagedorn, Phytopathology 59: 1051, 1969.
- Stevenson & Hagedorn, Phytopathology 60: 1148, 1970.
- Stevenson & Hagedorn, Pl. Dis. Reptr 57: 248, 1973.
- Thottappilly & Schmutterer, Z. PflKrankh. PflPath. PflSchutz 75: 1, 1968.