Details of DPV and References
DPV NO: 15 June 1970
Family: ssRNA satellite viruses
Genus: TNsatV-like satellite viruses
Species: Tobacco necrosis satellite virus | Acronym:
Satellite virus
B. Kassanis Rothamsted Experimental Station, Harpenden, Hertfordshire, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Kassanis & Nixon (1960, 1961) and Kassanis (1962).
The smallest RNA-containing virus known. It has isometric particles about 17 nm in diameter, and multiplies only in plants also infected with tobacco necrosis virus. Both viruses are transmitted by the zoospores of the chytrid fungus Olpidium brassicae.
Main Diseases
The virus is found in bean with stipple streak, and tulip with tulip necrosis (Augusta disease), but seems to play no part in causing these diseases.
Geographical Distribution
Found in Europe and North America in association with tobacco necrosis virus. Less common than tobacco necrosis virus.
Host Range and Symptomatology
Often assumed to multiply in all hosts and tissues infected by tobacco necrosis virus, but this might not necessarily be so, because, for example, the stipple streak strain of tobacco necrosis virus infects uninoculated leaves of Phaseolus vulgaris cv. The Prince much more readily than does satellite virus.
-
Diagnostic species
- Phaseolus vulgaris
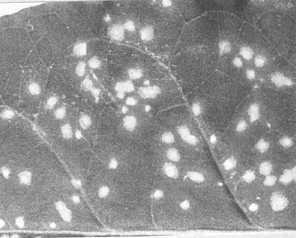
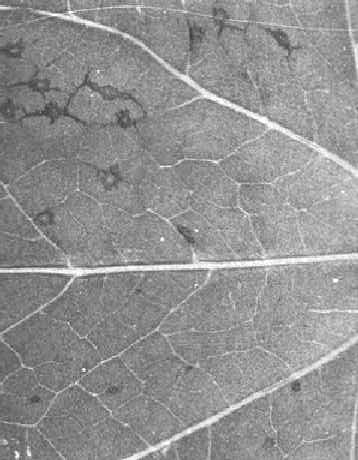
cv. The Prince (French bean) and Nicotiana tabacum cv. White Burley-Judy’s Pride (tobacco). In these, the necrotic local lesions produced by an inoculum containing both satellite and tobacco necrosis viruses are smaller and develop more slowly than do those produced by tobacco necrosis virus alone (Fig. 1, Fig. 2, Fig. 3). However, this difference is not always easy to detect, and for accurate diagnosis the sap of infected plants should be examined in the electron microscope or by serological tests.Propagation species
- Nicotiana tabacum, N. clevelandii
or Phaseolus vulgaris, depending on the strain of tobacco necrosis virus used as activator. For example P. vulgaris can be used for strains SV1 or SV2 in the presence of the stipple streak strain of tobacco necrosis virus.Assay species
- Phaseolus vulgaris
cv. The Prince. The concentration of satellite virus in an inoculum may be estimated from the proportion of small lesions produced (Kassanis, 1962). The most useful bait plant in transmission experiments with Olpidium zoospores is tobacco.
Strains
Three strains described from England are named SV1, SV2 and SV3 (Kassanis & Macfarlane, 1968). Three described from North America are named SVA, SVB, and SVC (Uyemoto, Grogan & Wakeman, 1968).
Transmission by Vectors
Transmitted by zoospores of the chytrid fungus Olpidium brassicae. Only strains SV2 and SV3 have been transmitted experimentally, success depending on a suitable combination of tobacco necrosis virus strain, satellite virus strain, fungus race and host plant species (Kassanis & Macfarlane, 1968).
Transmission through Seed
None reported.
Transmission by Dodder
None reported.
Serology
Moderately immunogenic. Reacts in tube precipitation tests in which it gives granular precipitates, and in gel-diffusion tests in which it gives a single band of precipitate.
Relationships
The four strains, SV1, SV2, SV3 and SVC differ antigenically and may be regarded as serotypes. The amino acid compositions of SV1, SV2 and SVC particles also differ considerably. SVA is serologically indistinguishable from SV1, and SVB from SV2 (Uyemoto & Grogan, 1969; Rees, Short & Kassanis, 1970).
The strains also differ in their ability to be activated by different strains of tobacco necrosis virus, and this difference seems unrelated to the serological grouping of either tobacco necrosis or satellite virus strains, but is correlated with the ability of tobacco necrosis virus to infect particular hosts. For example, the A, B, S, E, AC43, and citrus strains of tobacco necrosis virus infect tobacco and French bean readily and will all activate satellite virus strains SV1 and SV2 but not SVC, whereas the D, AC36 and grapevine strains of tobacco necrosis virus infect tobacco or French bean with difficulty or not at all and will activate satellite virus strain SVC but not strains SV1 or SV2 (Kassanis & Phillips, 1970).
No serological relationship has been found between satellite virus and any other virus, including the tobacco necrosis viruses used as satellite virus activators.
Stability in Sap
The thermal inactivation point is about 95°C. The virus is very stable at 20°C, and remains infective for many years at -4°C.
Purification
Frozen leaves or leaf pulp are thawed and triturated in a blender with water. After low speed centrifugation satellite virus is separated from tobacco necrosis virus and host plant constituents by repeated precipitation with ammonium sulphate (0.25 g/ml), each time resuspending the pellets in a smaller volume of water or 0.2 M sodium acetate buffer pH 4.5. Yields can be 50-100 mg of virus per litre of sap. The ammonium sulphate, or buffer, ensures that the pH remains low, and in these conditions some strains of tobacco necrosis virus (isoelectric point c. pH 4.5) are less soluble than satellite virus (isoelectric point c. pH 7.0) so that the tobacco necrosis virus remains insoluble when the satellite virus resuspends. The two viruses are finally separated by electrophoresis or by centrifuging in sucrose density gradients. Satellite virus crystallizes readily from concentrated preparations (even impure ones) and when sedimented in the ultracentrifuge such crystals are difficult to resuspend; this is one of the reasons for using the salt method instead of ultracentrifugation to purify the virus.
Properties of Particles
Purified preparations contain one type of particle.
Sedimentation coefficient (s20,w) at infinite dilution: c. 50 S; strain SV1 forms aggregates of 169, 231 and 332 S (Fig. 6, Fig. 7) (Kassanis & Woods, 1968).
Molecular weight (daltons): 1.97 x 106 (Reichmann, 1964).
Diffusion coefficient (D20,w x 10-7 cm2 sec-1): 2.04.
Isoelectric point: c. pH 7.0.
Partial specific volume (calculated from composition): 0.71.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 6.5.
A260/A280: 1.6-1.7.
Electrophoretic mobility: + 1 x 10-5 cm2 sec-1 volt-1 in 0.066 M phosphate buffer, pH 5.3 (Kassanis & Kleczkowski, 1965).
Particles crystallize in different forms - rhombic, rectangular or hexagonal plates or laths, depending on the virus strain and crystallization conditions.
Particle Structure
Particles are isometric (Fig. 4, Fig. 5, Fig. 6) about 17 nm in diameter and contain 60 protein subunits (Rees et al., 1970).
Particle Composition
RNA: Single-stranded, molecular weight about 0.4 x 106, constituting about 20% of the particle weight. Estimates of the nucleotide composition differ: G22-24; A28-29; C20-22; U25-29 (Uyemoto & Grogan, 1969; Reichmann, 1964). s20,w: 13.5-14.5 S in 0.1 ionic strength phosphate buffer pH 7.0.
Protein: 80% of the particle by weight. Probably only one kind of protein in the particle. Subunit molecular weight about 2.3 x 104, consisting of 208 or 209 amino acid residues (Uyemoto & Grogan, 1969; Rees et al., 1970). Earlier reports of a larger size for the subunit have not been confirmed (Roy et al., 1969; Lesnaw & Reichmann, 1969a, 1969b). The amino acid composition has been reported by the authors listed above and by Reichmann (1964).
Relations with Cells and Tissues
Satellite virus particles occur in the cytoplasm in close proximity to the tobacco necrosis virus particles (Kassanis, Vince & Woods, 1970).
Notes
The reason for the dependency of satellite virus on tobacco necrosis virus for its multiplication is presumably because its RNA is too small to specify all the proteins needed for replication. Of its 1200 nucleotides about half are needed to code for the protein in its particles, and therefore this virus may have only two cistrons.
Figures
References list for DPV: Satellite virus (15)
- Kassanis, J. gen. Microbiol. 27: 477, 1962.
- Kassanis, Adv. Virus Res. 13: 147,1968.
- Kassanis & Kleczkowski, Nature, Lond. 205: 310, 1965.
- Kassanis & Macfarlane, J. gen. Virol. 3: 277, 1968.
- Kassanis & Nixon, Nature, Lond. 187: 713, 1960.
- Kassanis & Nixon, J. gen. Microbiol. 25: 459, 1961.
- Kassanis & Phillips, J. gen. Virol. 9: 119, 1970.
- Kassanis, Vince & Woods, J. gen. Virol. 7: 143, 1970.
- Kassanis & Woods, J. gen. Virol. 2: 395, 1968.
- Lesnaw & Reichmann, Virology 38: 368, 1969a.
- Lesnaw & Reichmann, Virology 39: 729, 1969b.
- Reichmann, Proc. natn. Acad. Sci. U.S.A. 52: 1009, 1964.
- Rees, Short & Kassanis, Virology 40: 448, 1970.
- Roy, Fraenkel-Conrat, Lesnaw & Reichmann, Virology 38: 368, 1969.
- Uyemoto & Grogan, Virology 39: 79, 1969.
- Uyemoto, Grogan & Wakeman, Virology 34: 410, 1968.