Details of DPV and References
DPV NO: 158 September 1976
Family: Potyviridae
Genus: Potyvirus
Species: Onion yellow dwarf virus | Acronym: OYDV
Onion yellow dwarf virus
L. Bos Institute of Phytopathological Research, Wageningen, The Netherlands
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Melhus et al. (1929).
- Selected synonyms
- Allium
virus 1 (Rev. appl. Mycol. 17: 52) - Selected synonyms
- Marmor cepae (Rev. appl. Mycol. 28: 512)
- A virus with flexuous filamentous particles c. 775 nm long. Readily transmitted by several aphid species in the non-persistent manner and by inoculation of sap, mainly to certain Allium species. It is probably distributed world-wide.
Main Diseases
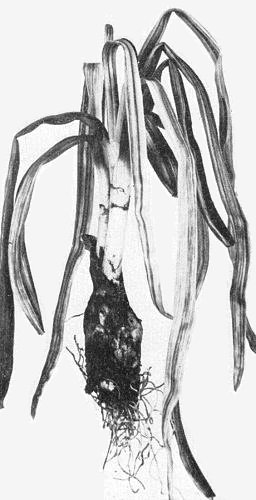
Causes onion yellow dwarf: stunting of first-year onion (Allium cepa) plants, with the leaves showing irregular yellow striping to almost complete yellowing, downward curling, flattening, crinkling and flaccidity (Fig. 2) (Melhus et al., 1929; Henderson, 1935). Also causes deterioration during storage and premature sprouting of onion bulbs (Bremer, 1937). In onion seed plants, the virus causes striping, curling and distortion of flower stems (Fig. 3), reduction in the number of flowers and seeds, and impairment of seed quality (Härdtl, 1965). In shallot (A. ascalonicum) leaf symptoms are similar, but leaf curling and plant stunting is often more severe (Fig. 1) (Brierley & Smith, 1946; Henderson, 1953). Recurrent reports of natural infection of garlic (A. sativum) and leek (A. porrum) were mainly based on visual observation. In these crops other viruses may play a major role (see Relationships and Notes).
Geographical Distribution
Reported from most countries where onions and shallots are cultivated.
Host Range and Symptomatology
Besides the species mentioned above, the virus has been isolated from naturally infected perennial tree, top or topset onion (A. cepa var. viviparum) and from garlic (A. sativum) (Brierley & Smith, 1944b). It was also said to occur in multiplier onion (A. cepa var. solaninum) (Brierley & Smith, 1944a) and in Welsh onion (A. fistulosum) (Costa et al., 1971), although others found this species to be immune (e.g. Brierley & Smith, 1946). It has also been detected in some ornamental Allium spp. (D. H. M. van Slogteren, personal communication) and some other Allium spp. (Havránek, 1973). Narcissus pseudonarcissus was found naturally infected (Brierley & Smith, 1946), and N. tazetta orientalis and the true jonquil N. odorus regulosus showed symptoms after inoculation (Henderson, 1935), and the virus could be recovered from them (Anon., 1932). Recently, local lesions were obtained with some isolates in Chenopodium amaranticolor and C. quinoa. With common isolates from onion, no symptoms or only slight symptoms were produced in Allium porrum and the virus could only rarely be recovered (L. Bos and N. Huijberts, unpublished data).
- Diagnostic species
- Allium cepa
(onion). Yellow striping and leaf curling, plant stunting; most cultivars (except some Spanish-type varieties) are susceptible. - Allium ascalonicum (shallot). Striping and curling of leaves, severe plant stunting.
- Propagation species
- Allium cepa
is easily infected. Roots, fleshy leaves, floral parts and pollen contain the virus, but basal portions of green leaves from plants 10-20 days after inoculation proved to be the best source of virus for mechanical transmission (Louie & Lorbeer, 1966).- Assay species
- No suitable quantitative assay host is known. Chenopodium species have not yet been thoroughly tested.
Strains
There seems to be some variation among isolates. In the USA a shallot isolate was readily transmitted to onion and infected all 27 onion cultivars tested, including Spanish-type cultivars immune to isolates from onion, garlic and narcissus (Brierley & Smith, 1946); the garlic and narcissus isolates were less infectious to onion and produced milder symptoms.
Transmission by Vectors
Several aphid species have transmitted the virus experimentally (Drake, Tate & Harris, 1933; Tate, 1940; Heinze, 1952), and may do so in the field when briefly probing in passing, because none is prevalent on Allium crops (Tate, 1940). Acquisition and inoculation is in short feeding times and aphid infectivity is rapidly lost (Drake et al., 1933). Myzus ascalonicus may spread the virus among onion bulbs and sprouts during storage (Szirmai, 1958). No experimental transmission by various other insects (Tate, 1940).
Transmission through Seed
Not detected in onion by several authors, although the virus was found in pollen of infected onion plants (Louie & Lorbeer, 1966). Seed transmission seems improbable because the disease was completely controlled in New Zealand by introducing an Allium-free period (Chamberlain & Baylis, 1948); however, Härdtl (1962; 1972) reported 6-29 % seed transmission in onion cv. ‘Stuttgarter Riesen’ on the basis of field observations.
Transmission by Dodder
Not reported.
Serology
Jermoljev et al. (1962) prepared specific antisera (titres 1/4 and 1/8) which they used to test onion cultivars for infection. After partial purification, D. H. M. van Slogteren (personal communication) prepared an antiserum with a titre of 1/320. D. Z. Maat (unpublished data) recently prepared an antiserum to a well-studied isolate, with a titre of 1/1024.
Relationships
The onion virus belongs to the potyvirus group, and de Wijs (1973), using van Slogteren’s antiserum, detected a serological relationship to pepper veinal mottle virus. The virus causing yellow stripe of leek (Bos, 1972; Verhoyen & Horvat, 1973; Verhoyen, 1973) is now known to differ from onion yellow dwarf virus. The onion virus is not, or poorly, infectious to leek and the leek virus behaves similarly in onion. The leek virus more consistently produces local lesions in Chenopodium amaranticolor and C. quinoa, and the two viruses are only distantly related serologically (antisera to the leek and onion viruses with titres of 1/4096 and 1/1024, respectively, reacted with the heterologous antigens only at dilutions up to 1/4 and 1/16, respectively) (D. Z. Maat, unpublished data).
Stability in Sap
The thermal inactivation point is between 60 and 65°C. The dilution end-point is 10-3 to 10-4 and longevity in vitro is 2-3 days. The virus withstands drying and storage in leaves over CaCl2 at 4°C (L. Bos, unpublished data).
Purification
Presence of mucilage in sap from Allium spp. impeded early attempts to isolate the virus. A method developed by Huttinga (1975) for purification of the leek virus also proved successful for the onion yellow dwarf virus from onion (it should be noted that the chromatography is of special importance):
Grind 100 g tissue in 500 ml 0.1 M Tris buffer (adjusted to pH 9 with thioglycollic acid) together with 20 ml chloroform, 20 ml carbon tetrachloride and 10 ml diethyl ether. Centrifuge for 10 min at 4000 g. Centrifuge the supernatant fluid for 1.5 h at 26,500 g and resuspend pellets in 50 ml 0.1 M Tris-HCl buffer pH 9. After 2 h at 4°C filter the suspension through filter paper in Buchner funnel; then pass the filtrate through the Sephadex G-200 column with the above buffer containing 4 x 10-4 M NaN3, using a peristaltic pump allowing a flow rate of 4.6 ml/h/cm2. Pool the virus-containing ultraviolet-absorbing fractions and concentrate by centrifuging for 1.75 h at 47,000 g. Resuspend the pellets, and centrifuge through a 10-40% sucrose density-gradient for 2 h at 25,000 rev/min.
Properties of Particles
No information.
Particle Structure
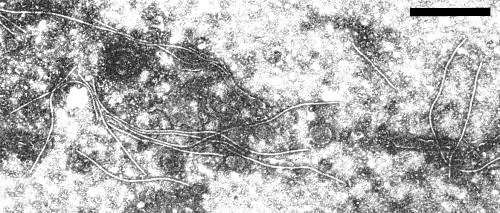
Particles are flexuous filaments, 722 nm long and 16 nm in diameter (Schmidt & Schmelzer, 1964), or over 800 nm long (L. Bos & N. Huijberts, unpublished data). They can easily be detected in extracts of diseased onion leaves chopped in phosphotungstic acid, and often occur in aggregates (Fig. 5).
Particle Composition
No information.
Relations with Cells and Tissues
Cells in epidermal strips from onion may contain one, two
or sometimes more round or slightly elongate inclusions visible by light microscopy (Fig. 4); these
inclusions can be easily confused with the nuclei (Tate, 1935) but consist of numerous rod-like
structures (McWhorter, 1937). In ultrathin sections of diseased onion leaves pinwheels, scrolls
and virus particles associated with vesicles can be observed with the electron microscope
(Edwardson, 1974).
Notes
From recent partially published work it appears that several viruses, including some that are soil-borne, naturally infect Allium species. Onion yellow dwarf virus and leek yellow stripe virus are potyviruses that can be easily diagnosed by their particle morphology and characteristic inclusion bodies. They differ from each other in host range and antigenic specificity. Other Allium viruses inadequately or incompletely described so far, are garlic mosaic virus, tentatively ascribed to the carlavirus group, and a shallot latent virus belonging to the same group (Bos, 1972). Garlic mosaic virus (Messiaen & Arnoux, 1960; Marrou & Fauvel, 1963, 1964; Messiaen & Marrou, 1965) also produces local lesions in C. amaranticolor and C. quinoa, but is difficult to transmit to onion, in which it produces mild symptoms; similarly, onion yellow dwarf virus proved poorly infectious to garlic (Marrou & Fauvel, 1964). Potyvirus like particles (probably onion yellow dwarf virus) may also occur naturally in garlic plants (Cadilhac, Quiot & Leroux, 1975). The onion mosaic virus reported in Russia (Andrejev, 1937; Rischkov & Vovk, 1937) and prevalent there (Razvjazkina, 1971), has been associated with flexuous particles 675 nm long (Prozenko & Legunkova, 1961; Razvjazkina, 1971) and spherical ones, 200 nm in diameter (Prozenko & Legunkova, 1961) and with transmission by aphids and by mites.
Figures
References list for DPV: Onion yellow dwarf virus (158)
- Andrejev, Sb. nauchno-issled. Rab. azovo-chernomorsk. sel'. -khoz. Inst. 5: 125, 1937.
- Anonymous, Rep. Iowa agric. Exp. Stn 1932: 32, 1932.
- Bos, Gewasbescherming 3: 81, 1972.
- Bremer, Phytopath. Z. 10: 79, 1937.
- Brierley & Smith, Phytopathology 34: 506, 1944a.
- Brierley & Smith, Phytopathology 34: 990, 1944b.
- Brierley & Smith, Phytopathology 36: 292, 1946.
- Cadilhac, Quiot & Leroux, Abstr. 2nd int. Conf Progr. Probl. Vegetable Virus Res. Avignon-Montfavet, 1975: 29, 1975.
- Chamberlain & Baylis, N.Z. Jl Sci. Technol. A 29: 300, 1948.
- Costa, Costa, Nagai & Kitajima, Biológico 37: 157, 1971.
- De Wijs, Neth. J. Pl. Path. 79: 189, 1973.
- Drake, Tate & Harris, J. econ. Ent. 26: 841, 1933.
- Edwardson, Monogr. Ser. Fla agric. Exp. Stn 4: 398 pp., 1974.
- Härdtl, Z. PflKrankh. Pflpath. PflSchutz 69: 587, 1962.
- Härdtl, Gartenbauwissenshaft 30: 347, 1965.
- Härdtl, Z. PflKrankh. PflPath. PflSchutz 79: 694, 1972.
- Havránek, Proc. 7th Conf. Czechosl. Pl. Virol. High Tatras, 1971: 133, 1973.
- Heinze, Z. PflKrankh. PflPath. PflSchutz 59: 3, 1952.
- Henderson, Res. Bull. Iowa agric. Exp. Stn 188: 209, 1935.
- Henderson, Pl. Path. 2: 130, 1953.
- Huttinga, Neth. J. Pl. Path. 81: 81, 1975.
- Jermoljev, Cech, Pozdena & Chod, Sb. csl. Akad. zeméd. Ved 35: 551, 1962.
- Louie & Lorbeer, Phytopathology 56: 1020, 1966.
- Marrou & Fauvel, Rapp. Activ. Inst. natn. Rech. agron.: 32, 1963.
- Marrou & Fauvel, Rapp. Activ. Inst. natn. Rech. agron.: 62, 1964.
- McWhorter, Phytopathology 27: 1627, 1937.
- Melhus, Reddy, Henderson & Vestal, Phytopathology 19: 73, 1929.
- Messiaen & Arnoux, Étud. Virol. appl. INRA 1: 29, 1960.
- Messiaen & Marrou, C. r. 1e Journées Phytiat. Phytopharm, circum-médit., Marseille. 1965: 204, 1965.
- Prozenko & Legunkova, Mikrobiologiya 30: 165, 1961.
- Razvjazkina, TagBer. dt. Akad. LandwWiss. Berl. 115: 69, 1971.
- Rischkov & Vovk, Dokl. Akad. Nauk SSSR 16: 69, 1937.
- Schmidt & Schmelzer, Phytopath. Z. 50: 191, 1964.
- Szirmai, Novenytermelés 7: 63, 1958.
- Tate, Iowa St. Coll. J. Sci. 9: 677, 1935.
- Tate, Iowa St. Coll. J. Sci. 14: 267, 1940.
- Verhoyen, Parasitica 29: 3, 1973.
- Verhoyen & Horvat, Parasitica 29: 16, 1973.