Details of DPV and References
DPV NO: 162 September 1976
Family: Potyviridae
Genus: Ipomovirus
Species: Sweet potato mild mottle virus | Acronym: SPMMV
Sweet potato mild mottle virus
M. Hollings Glasshouse Crops Research Institute, Littlehampton, Sussex, BN16 3PU, England
Olwen M. Stone Glasshouse Crops Research Institute, Littlehampton, Sussex, BN16 3PU, England
K. R. Bock Ministry of Overseas Development, E.A.A.F.R.O., P.O. Box 30148, Nairobi, Kenya
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by Hollings, Stone & Bock (1976).
Synonyms
- Sweet potato virus T (Hollings, Bock & Ngugi, 1971; Hollings, Stone & Bock, 1971)
An RNA-containing virus with filamentous particles c. 950 nm long, found in sweet potatoes in East Africa. The virus is readily sap-transmissible to a fairly wide range of herbaceous plant species, and by the whitefly Bemisia tabaci permitted long feeds.
Main Diseases
In sweet potato (Ipomoea batatas), the virus can cause leaf mottling, stunting and loss of yield (Fig. 1, Fig. 2). Different sweet potato genotypes differ greatly in susceptibility and reaction to the virus, some being symptomlessly infected and others apparently immune. The virus reaches only low concentrations in sweet potato.
In tomato (Lycopersicon esculentum), systemic vein-mottling and slight loss of vigour are caused, but in tobacco (Nicotiana tabacum), a very severe systemic blister-mottle, distortion and stunting occur (Fig. 3, Fig. 5).
Geographical Distribution
Kenya, Tanzania and Uganda.
Host Range and Symptomatology
Found in naturally infected sweet potato, but apparently not in tomato or tobacco crops. The virus infected 45 of 119 plant species in 14 of 36 plant families (Hollings et al., 1976). Sweet potato sap contains powerful inhibitors of infection, and sap transmission from this to test plants is sometimes difficult.
-
Diagnostic species
- Nicotiana tabacum
(tobacco), White Burley types. Chlorotic or grey necrotic local lesions after 7 days; severe systemic chlorotic vein-clearing and netting in 10-16 days, later developing into vein-banding, severe puckering, blistering, mottling and distortion (Fig. 3, Fig. 5). A characteristic epinasty often follows after 4-5 weeks. - N. glutinosa. Few, grey necrotic local lesions, at first not unlike those
of
tobacco mosaic virus, after 5-7 days; followed by systemic vein-clearing, leaf
mottling and crinkling, stunting and rosetting
(Fig. 7). Severe leaf distortion,
including tendril leaves, often develops after several weeks.
- Ipomoea setosa and I. nil cv. Scarlett O’Hara. Very conspicuous systemic vein-chlorosis and netting in 10-18 days, followed by a rugose mottle, leaf crinkle and distortion (Fig. 4). After 3-4 weeks, new growth is almost symptomless.
-
Propagation species
- N. tabacum
and N. clevelandii are useful for virus propagation, and N. glutinosa for maintaining cultures.Assay species
- Chenopodium quinoa:
small, chlorotic local lesions after 8-11 days (Fig. 6); not systemic. C. quinoa is 3-5 times more sensitive than C. amaranticolor.
Strains
Ten isolates from different parts of Kenya, Tanzania and Uganda gave very similar reactions in several host plants and were serologically very closely related.
Transmission by Vectors
One culture of the whitefly Bemisia tabaci transmitted the virus between sweet potato, N. glutinosa and N. clevelandii plants; a minimum inoculation period of 5 days was required (Hollings et al., 1976). An isolate from Tigoni (Nairobi Highland area) was apparently more readily transmitted than one from Embu (Mt. Kenya).
No transmission was obtained with the aphids Myzus persicae, Aphis craccivora or A. gossypii which are effective vectors of certain other sweet potato viruses (see Notes).
Transmission through Seed
No seed transmission was detected in Ipomoea nil cv. Scarlett O’Hara, or N. clevelandii.
Transmission by Dodder
Cuscuta campestris transmitted the virus from infected to healthy Calystegia sepium; the Cuscuta was not itself infected.
Serology
The virus is a fairly good immunogen, but it reacts only slowly with antiserum in precipitin tube tests. Antisera prepared in rabbits by one intravenous, and two intramuscular injections (with Freund's complete adjuvant) over 3 weeks had titres against purified virus preparations in tube precipitin tests of 1/8192 and 1/16,384, the precipitates being flocculent (flagellar). Microprecipitin drop tests under paraffin oil were also satisfactory, but ring tests gave poor reactions. Clarified sap of infected N. tabacum reacted poorly or not at all in tube and microprecipitin tests. The virus did not react in gel immunodiffusion tests, even when virus-disrupting agents were used (Hollings et al., 1976).
Relationships
The virus is serologically unrelated to any of 14 potyviruses, including several aphid-transmitted viruses from sweet potato from E. and S. Africa, USA, Puerto Rico, Australia and New Zealand (Hollings et al., 1976; M. Hollings & O. M. Stone, unpublished data). These viruses did not protect in Ipomoea setosa against subsequent challenge by sweet potato mild mottle virus.
Stability in Sap
The virus is rather labile in sap of N. clevelandii or N. tabacum. It was still infective in N. tabacum sap at dilutions of 10-3 to 2 x 10-4, but only at 1/25 in sweet potato sap. Infectivity withstood 10 min at 55 but not 60°C (although 95% of infectivity was lost at 50°C), and survived 3 but not 7 days at 18°C, and 42 but not 49 days at 2°C.
Infected tobacco leaves dried over anhydrous CaCl2 at 2°C were still infective after 3 years; lyophilised sap (with 7% each of peptone and dextrose added) retained some infectivity for 1 but not 2 yr under high vacuum at c. 18°C (Hollings et al., 1976).
Purification
The virus is easily purified from systemically infected N. tabacum. In
Europe, much better yields are obtained between September and March than between May
and August. Purification methods include:
1. (Hollings et al., 1976). Harvest tobacco plants 12-18 days after infection, blend in 0.05 M phosphate buffer (pH 7.6) (2 ml/g tissue) containing 0.1 % (v/v) thioglycollic acid, and filter through cloth. Add n-butanol drop-wise (9.3 ml/100 ml juice) and shake the mixture 1-2 h at laboratory temperature. Centrifuge at 10,000 g for 30 min. Precipitate the virus from the supernatant fluid by dissolving 2% (w/v) NaCl, then 4% (w/v) polyethylene glycol (PEG M.Wt 6000), stand the mixture 1-2 h at c. 4°C and centrifuge for 10 min at 10,000 g. Re-suspend the precipitate in 0.03 M phosphate buffer (pH 7.6), 1 ml/50 g of original leaf tissue and, after c. 1 h, remove insoluble material by brief centrifugation.
Further purification can be effected by one cycle of differential centrifugation,
or by sucrose density-gradient centrifugation, although the latter results in up to
90% loss of virus.
2. (R. J. Barton & M. Hollings, unpublished information). About 1.5 ml of the re-suspended PEG precipitate from procedure (1) above is applied to an 80 x 1.5 cm column of controlled-pore glass beads (70 nm pore size; Sigma London Chemical Co. Ltd.). To prevent non-specific adsorption, the column is pre-treated with polyethylene glycol M.Wt 20,000 at 1% (w/v) in 0.04 M phosphate buffer pH 7.0. The virus is eluted with 0.04 M phosphate buffer pH 7.0 and appears in the void volume, well separated from smaller host material; it is further concentrated by ultracentrifugation.
Properties of Particles
Purified preparations produce a single light-scattering band in density-gradient centrifugation, and a single component in analytical ultracentrifugation with an s°20, w of 155 S.
A260/A280: 1.33; Amax(259)/Amin(237): 1.23 (values corrected for light-scattering) (Hollings et al., 1976).
Particle Structure
Infective particles are rather stiff, filamentous rods c. 950 nm long in leaf-dip preparations and in 0.05 M MgCl2 (Fig. 8); in 0.04 M EDTA (pH 7.0) they are slightly flexuous and 800-900 nm long.
Particle Composition
Nucleic acid: Probably RNA, single-stranded.
Protein: Particles contain one protein of M. Wt 37,700 (Hollings et al., 1976).
Relations with Cells and Tissues
The virus is present in leaves, stems, flowers and roots. Pinwheel inclusion bodies are seen in thin sections of the mesophyll, often associated with the plasmalemma (P. T. Atkey & M. Hollings, unpublished information).
Notes
The symptoms in sweet potato are not diagnostic and, as with most sweet potato viruses, differ greatly according to host genotype.
Virus-free clones of sweet potato can be established by thermotherapy (4-5 weeks at 35°C) immediately followed by rooting mini-cuttings or stem nodes (remove the leaves and petioles), or by meristem-tip culture (Hollings et al., 1976). Ipomoea nil was not freed from the virus after 8 weeks at 35-37°C.
Most of the previously reported sweet potato viruses have not been characterised in vitro, but some have recently been shown to be typical aphid-transmitted potyviruses- e.g. russet crack (Lawson et al., 1971; Campbell et al., 1974); vein mosaic (Nome, 1973); feathery mottle (Alconero 1973; Campbell et al., 1974; Nome et al., 1974) and ‘hardcore’ (Hammond et al., 1974). These and other aphid-borne presumed viruses of sweet potato are not readily sap transmissible and apparently are restricted in host range to the Convolvulaceae, e.g. the viruses of internal cork (Hildebrand, 1960) and ringspot (Loebenstein & Harpaz, 1960). The claimed whitefly transmission of feathery mottle virus (Hildebrand, 1959) has not been confirmed; this name has been re-defined by Campbell et al. (1974).
The reported whitefly-transmitted viruses of sweet potato include mosaic (Girardeau, 1958); vein clearing (Loebenstein & Harpaz, 1960); yellow-dwarf (Hildebrand, 1961) and virus B (Sheffield, 1957, 1958). No particles have been seen for any of these, and little or no information exists on their in vitro properties. Only Sheffield's virus B, however, is reported to be sap-transmissible, and able to infect hosts outside the Convolvulaceae. Campbell et al. (1974) suggest that mosaic, vein-clearing and yellow dwarf may be the same or similar entities. We suggest that mild mottle virus is probably the same as virus B, and distinct from all other sweet potato viruses so far described.
Although pinwheel inclusions are produced, the particle morphology and whitefly vector, the lack of serological relationship to a range of potyviruses and failure to protect against those from sweet potato, distinguish sweet potato mild mottle virus from the potyviruses.
Figures
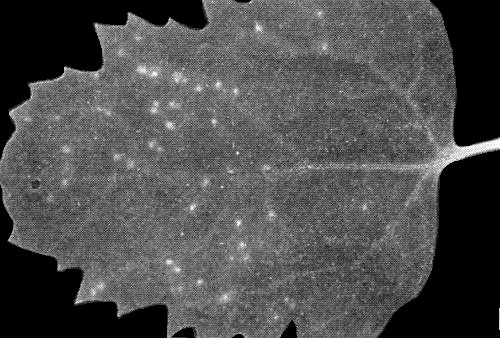
Transient systemic chlorotic vein netting in leaf of sweet potato (local Kenyan cv.) 4 weeks after experimental infection.
Systemic chlorotic vein flecks and slight mottle in leaf of sweet potato (seedling line) 8 weeks after experimental infection.

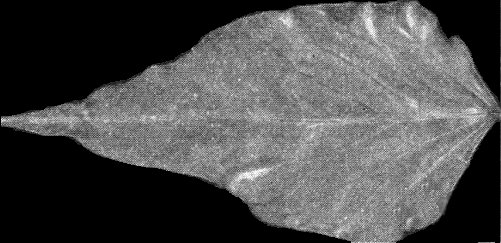
Characteristic early symptoms in Burley tobacco (selection Dutch A) 10 days after infection: necrotic local lesions enlarging to small rings; systemic intense chlorotic vein netting, which persists only for a few days.

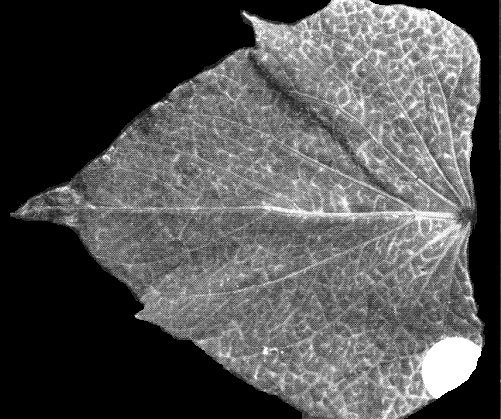
Transient systemic vein clearing in leaf of Ipomoea nil cv. Scarlett O’Hara, 14 days after infection.

Later symptoms in Burley tobacco (Dutch A) 4 weeks after infection: severe systemic dwarfing, crumpling, epinasty and distortion of younger leaves.
References list for DPV: Sweet potato mild mottle virus (162)
- Alconero, Phytopathology 63: 377, 1973.
- Campbell, Hall & Mielinis, Phytopathology 64: 210, 1974.
- Girardeau, Pl. Dis. Reptr 42: 819, 1958.
- Hammond, Dames & Corbett, Pl. Dis. Reptr 58: 17, 1974.
- Hildebrand, Pl. Dis. Reptr 43: 712, 1959.
- Hildebrand, Phytopathology 50: 751, 1960.
- Hildebrand, Science, N.Y. 133: 282, 1961.
- Hollings, Bock & Ngugi, Rep. E. Afr. Agric. Forest. Res. Org. for 1970: 78, 1971.
- Hollings, Stone & Bock, Rep. Glasshouse Crops Res. Inst. for 1970: 155, 1971.
- Hollings, Stone & Bock, Ann. appl. Biol. 82: 511, 1976.
- Lawson, Hearon & Smith, Virology 46: 453, 1971.
- Loebenstein & Harpaz, Phytopathology 50: 100, 1960.
- Nome, Phytopath. Z. 77: 44, 1973.
- Nome, Shalla & Petersen, Phytopath Z. 79: 169, 1974.
- Sheffield, Phytopathology 47: 582, 1957.
- Sheffield, Phytopathology 48: 1, 1958.