Details of DPV and References
DPV NO: 163 September 1976
Family: Rhabdoviridae
Genus: Cytorhabdovirus
Species: Strawberry crinkle virus | Acronym: SCV
Strawberry crinkle virus
E. S. Sylvester Division of Entomology and Parasitology, University of California, Berkeley, California 94720, USA
N. W. Frazier Division of Entomology and Parasitology, University of California, Berkeley, California 94720, USA
Jean Richardson Division of Entomology and Parasitology, University of California, Berkeley, California 94720, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Disease described by Zeller & Vaughan (1932),
virus particles by Richardson, Frazier & Sylvester (1972).
Selected synonyms
-
Strawberry latent virus, strains A and B (Rev. appl. Mycol. 33: 306)
- Strawberry lesion-A and lesion-B viruses (Rev. appl. Mycol. 38: 217)
- Strawberry virus 3 (Rev. appl. Mycol. 28: 341)
- Strawberry virus 4 (Rev. appl. Mycol. 32: 573)
- Vein chlorosis virus (Rev. appl. Mycol. 32: 573)
- Fragaria virus 2 (Rev. appl. Mycol. 17: 52)
- Marmor fragariae (Holmes, 1939)
- Strawberry lesion-A and lesion-B viruses (Rev. appl. Mycol. 38: 217)
-
A large bacilliform virus with enveloped particles c. 190-380 nm long and 69 ± 6 nm wide. Transmitted in a persistent manner by two Chaetosiphon aphid species to Fragaria spp. Not transmissible by inoculation of sap. Widespread and can be very damaging, especially when in complexes with other viruses (Frazier & Mellor, 1970).
Main Diseases
When alone in strawberry it causes crinkle; in complexes with other viruses, variations of crinkle, yellows (xanthosis, yellow-edge) and leaf curl can result (Plakidas, 1964).
Geographical Distribution
World-wide (North and South America, Europe, British Isles, South Africa, New Zealand, Australia) except in regions where Chaetosiphon species of aphids are not found on strawberry.
Host Range and Symptomatology
Crinkle virus has a narrow host range among species of the dicotyledonous genus Fragaria; principally on the wild species F. vesca, F. virginiana, and F. chiloensis, as well as on the cultivated strawberry, F. chiloensis var. ananassa. The virus is transmissible among plants with difficulty by aphid vectors (Mellor & Fitzpatrick, 1961), readily by grafting (Frazier, 1974b), but not by inoculation of sap. It can be moved from aphid to aphid by needle inoculation (Sylvester, Richardson & Frazier, 1974).
-
Diagnostic species
- Fragaria vesca
, indicator clones (Frazier, 1974a): chlorotic to necrotic irregular spots on veins (Fig. 1); epinasty, crinkling, distortion and uneven expansion of leaflets (Fig. 1, Fig. 3); lesions on petioles and stolons producing angularity (Fig. 2); streaking and deformation of petals (Fig. 4). Lesions and/or petal streaks are diagnostic on F. vesca, but this symptom does not develop with enough consistency to be diagnostically useful on F. virginiana, F. chiloensis, or F. c. var. ananassa.Propagation species
- Fragaria chiloensis
and F. c. var. ananassa, usually symptomless carriers, are suitable plants for maintaining cultures.Assay species
- Alpine strawberry, Fragaria vesca var. semperflorens, is an excellent host for assaying transmission by vectors (Frazier, 1968).
Strains
Several strains have been distinguished by the severity of symptoms. The existence of an infection with a mild strain may be diagnosed only by the symptoms produced when the plants are inoculated with another virus such as strawberry mottle (Frazier, 1968). A variant of the C10 group of Frazier (1968) used in the serial passage of the virus in Chaetosiphon jacobi (Sylvester et al., 1974) is the best known. It is of intermediate virulence, but produces distinct chlorotic spotting, lesions and petal streaks on seedlings of the assay plant F. vesca var. semperflorens. Source, USA.
Transmission by Vectors
Transmitted by the strawberry aphids Chaetosiphon fragaefolii and C. jacobi (Frazier & Mellor, 1970). Prentice & Woollcombe (1951) using C. fragaefolii under unspecified conditions reported acquisition within 24 h, a latent period in the vector of 10-19 days, and the persistence of inoculativity for at least 2 weeks. The incubation period in the plant was 4 to 8 weeks. Using C. jacobi, again with uncontrolled conditions, Frazier (1968) reported a latent period of 14-59 (average of 32) days, and the persistence of inoculativity for nearly the lifetime of the aphids. The plant incubation period varied from 5 to 39 days, with an average of 12 days. Sylvester et al. (1974) obtained evidence that the virus multiplied in C. jacobi. The virus was transferred from aphid to aphid by needle injection for 6 consecutive serial passages, with an estimated dilution of the original dose of inoculum of more than 10-20. A stable median incubation period of c. 6 days at 25° was maintained throughout the passages. When kept at 25°, the proportion of injected insects transmitting the virus declined from a peak of 90% at 10 days after injection to about 10% at 22-24 days, although two-thirds of the injected insects were still alive at 30 days. There was no evidence of transmission through the egg.
Transmission through Seed
None reported.
Transmission by Dodder
None reported.
Serology
No information available.
Relationships
In plant protection tests, milder isolates gave partial protection against more virulent isolates introduced by grafting (Frazier & Posnette, 1958).
Particle morphology places strawberry crinkle virus in the rhabdovirus group. Among the better characterized viruses in this group, it resembles lettuce necrotic yellows (Francki & Randles, 1970), sowthistle yellow vein (Peters, 1971), broccoli necrotic yellows (Campbell & Lin, 1972) and raspberry vein chlorosis (Stace-Smith & Lo, 1973; Jones, Roberts & Murant, 1974) in being aphid-transmitted; it resembles the last two of these in reported particle width (c. 70 nm) in thin sections of infected tissue. In contrast, the particle width of lettuce necrotic yellows virus in thin sections is c. 50 nm (Chambers, Crowley & Francki, 1965) and that of sowthistle yellow vein virus is c. 80 nm (Richardson & Sylvester, 1968).
Stability in Sap
Little information available. The virus remains infectious for at least an hour in refrigerated extracts made by triturating the head of an infective aphid in 5 µl of distilled water. Infectious virus can be recovered from aphids frozen at -54°C for 2 years (Sylvester & Richardson, unpublished).
Purification
No information.
Properties of Particles
No information.
Particle Structure
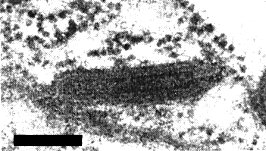
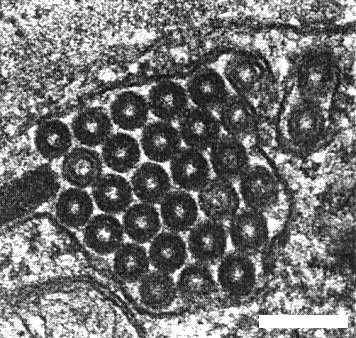
Particles are bacilliform, c. 190-380 nm long and 69 ± 6 nm wide. In situ, either in the aphid Chaetosiphon jacobi, or in Fragaria vesca var. semperflorens, particles occur in the cytoplasm in either the coated (Fig. 7, Fig. 8, Fig. 9, Fig. 11), or the uncoated form (Fig. 6, Fig. 10) (Richardson et al., 1972). An internal component that gives a striated appearance to some particles seen in thin sections may be a coiled helical structure. In negatively stained preparations bacilliform particles have a tendency to break and produce bullet-shaped particles (Fig. 5).
Particle Composition
No information.
Relations with Cells and Tissues
Both coated and uncoated particles have been found in cytoplasm of epidermal cells and parenchyma cells near vascular bundles of infected F. vesca var. semperflorens. Particles occur singly or in groups (Fig. 9), and at times they are encysted within an electron dense matrix in distorted cells (Richardson et al., 1972). Limited collections of particles may be found enclosed in membrane-bound cisternae (Fig. 11). In aphid tissues, both coated and uncoated particles have been found in salivary and accessory glands, in tissues of the oesophagus, muscles and central nervous system, as well as in the tonofilaments of the cuticular areas of muscle attachment (Sylvester & Richardson, unpublished). Particles have not been found in the nuclei of either plant or aphid cells.
Notes
Crinkle can be diagnosed by graft transmission to F. vesca with the production of petal streak and lesions on petioles and stolons. The extremely long incubation period in aphid vectors is also diagnostic, but usually so few insects acquire and transmit the virus the test normally would not be made. Control has been achieved by heat therapy (Posnette & Cropley, 1958), meristem culture (Miller & Belkengren, 1962) combined with annual planting of stock certified to be free of crinkle virus.
Figures

Chlorotic spotting, crinkling, deformation and unequal development of Fragaria vesca var. semperflorens (Alpine strawberry) leaflets.
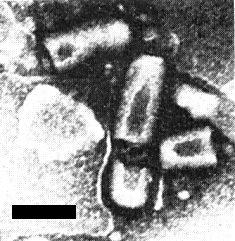
Bullet-shaped negatively stained particles from the head of an infected aphid, Chaetosiphon jacobi. Bar represents 100 nm.

Coated virus particles in cuticular tissue in the head of an infected C. jacobi. Bar represents 100 nm.

Coated virus particles in the cytoplasm of a mesophyll cell from a petal of infected F. vesca var. semperflorens. Bar represents 200 nm.
References list for DPV: Strawberry crinkle virus (163)
- Campbell & Lin, CMI/AAB Descriptions of Plant Viruses 85, 4 pp., 1972.
- Chambers, Crowley & Francki, Virology 27: 320, 1965.
- Francki & Randles, CMI/AAB Descriptions of Plant Viruses 26, 4 pp., 1970.
- Frazier, Phytopathology 58: 165, 1968.
- Frazier, Pl. Dis. Reptr 58: 28, 1974a.
- Frazier, Pl. Dis. Reptr 58: 203, 1974b.
- Frazier & Mellor, In Virus Diseases of Small Fruits and Grapevines, p. 18, Univ. of Calif. Div. of Agric. Sci., Berkeley, Calif. (Frazier, N. W., ed.), 1970.
- Frazier & Posnette, Hilgardia 27: 455, 1958.
- Holmes, Handbook of Phytopathogenic Viruses, p. 78, Minneapolis,Burgess, 1939.
- Jones, Roberts & Murant, Ann. appl. Biol. 77: 283, 1974.
- Mellor & Fitzpatrick, Can. Pl. Dis. Surv. 41: 218, 1961.
- Miller & Belkengren, Phytopathology 52: 743, 1962.
- Peters, CMI/AAB Descriptions of Plant Viruses 62, 3 pp., 1971;
- Plakidas, Strawberry Diseases, Louisiana State Univ. Press, Baton Rouge, 1964.
- Posnette & Cropley, J. hort. Sci. 33: 282, 1958.
- Prentice & Woollcombe, Ann. appl. Biol. 38: 389, 1951.
- Richardson & Sylvester, Virology 35: 347, 1968.
- Richardson, Frazier & Sylvester, Phytopathology 62: 491, 1972.
- Stace-Smith & Lo, Can. J. Bot. 51: 1343, 1973.
- Sylvester, Richardson & Frazier, Virology 59: 301, 1974.
- Zeller & Vaughan, Phytopathology 22: 709, 1932.