Details of DPV and References
DPV NO: 167 September 1976
Family: Potyviridae
Genus: Bymovirus
Species: Wheat spindle streak mosaic virus | Acronym: WSSMV
Wheat spindle streak mosaic virus
J. T. Slykhuis Agriculture Canada Research Station, Summerland, B.C. V0H IZ0, Canada
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by Slykhuis (1960, 1970).
A soil-borne virus with flexuous, filamentous particles frequently exceeding 2000 nm in length. It is sap-transmissible and transmitted in soil by the fungus, Polymyxa graminis. Wheat (Triticum spp.) is the only known host. It affects wheat in Ontario, Canada, eastern USA and probably southern France.
Main Diseases
Causes a mosaic disease of autumn- or winter-sown wheat (Triticum spp.) characterized by chlorotic to necrotic spindle-shaped streaks in the leaves (Fig. 2), slight stunting, reduced tillering and reduced seed yield. Yields of wheat growing in infective soil may be reduced 25 to 50% (Slykhuis, 1970) but generally much smaller losses are evident (Gates, 1969). In Ontario the disease has been found only in fields in which winter wheat has been grown at least three times at intervals of five years or less. In the first year that the disease appears in a field it affects only scattered plants. In the next crop of wheat it occurs in patches through the field and may affect most plants in the patches. In subsequent wheat crops it may affect most plants in the field. In years favouring the disease, virtually all the plants are diseased in most fields in which wheat has been grown many times before. It did not develop in fields that regularly received heavy applications of animal or poultry manures between wheat crops (Slykhuis, 1973a).
Temperature is the major climatic factor affecting disease development. Wheat sown in infective soil in the autumn becomes infected after emergence when the soil temperature is around 15°C (Slykhuis, 1975b). Symptoms are not generally evident until growth resumes in the spring at temperatures between 5°C and 15°C. Wiese & Hooper (1971) suggested that vernalization environments increased symptom severity and the percentage of plants affected. Losses are greatest when cool conditions persist through May and June in Ontario, but symptom development is curtailed and recovery pronounced if temperatures are predominantly above 20°C in the later tillering and through the jointing stage of crop development.
Geographical Distribution
Ontario in Canada; Indiana (Jackson et al., 1975), Kentucky (Williams et al., 1976), Maryland, Michigan (Wiese et al., 1970), New York and Pennsylvania in the USA. A similar virus causes a similar disease of winter-grown durum wheat in southern France (P. A. Signoret, personal communication).
Host Range and Symptomatology
The virus affects winter wheat (Triticum aestivum) in Canada and the USA, and durum wheat (T. durum) grown during winter in southern France. Most wheat cultivars can be infected by growing in infective soil or by inoculating leaves with sap from diseased plants. Yamhill (Wiese, Ravenscroft & Everson, 1974) and Skorospilka appear to be immune. Other species of Gramineae tested that did not develop symptoms include Agropyron repens, Avena sativa, Bromus inermis, Dactylis glomerata, Hordeum vulgare, Lolium multiflorum, Phleum pratense, Poa pratensis, Secale cereale, Trifolium pratense and Zea mays. Neither symptoms nor infection were detected in Amaranthus retroflexus, Chenopodium album, Trifolium pratense, or any of a number of other dicotyledonous plants grown in infective soil.
-
Diagnostic species
- T. aestivum
and T. durum (wheat). Light green to yellow spindle-shaped streaks (Fig. 2) develop in 21 to 42 days after leaf inoculations; after sowing wheat in infective soil the symptoms appear after 28 to 120 days but usually in 60 to 90 days if the conditions for testing are favourable (Slykhuis, 1974, 1975a, 1976).For mechanical transmission, wheat plants are grown in a greenhouse at about 20°C. Inoculum is prepared from leaves with severe chlorotic streaks from diseased plants grown at 10°C and not exposed to temperatures much above 15°C for long periods. The leaves are ground in 0.1 M phosphate buffer, pH 7 or 0.1 M Na2SO3 (1 g leaves: 3 ml solution). Inoculation by the air-brush method is more effective than by leaf rubbing. After inoculation the plants are grown at 10°C with light at 10,000 to 15,000 lux for 12 h per day.
For soil transmission, wheat is grown in small pots of infective soil at 15°C with light at 15,000 lux for 12 h per day for 2 to 4 weeks, then moved or transplanted into larger pots of soil and grown at 10°C.
Propagation species
- Triticum aestivum
(wheat). Winter wheat cultivars Kent, Genesee, Talbot, or any others that show symptoms in the field, are suitable.Assay species
- No local lesion host is known. The virus may be assayed by determining the percentage of wheat plants that develop symptoms after inoculation with different dilutions of plant sap.
Strains
No strain differences have been detected. The type strain is lodged with the American Type Culture Collection as No. PV 116.
Transmission by Vectors
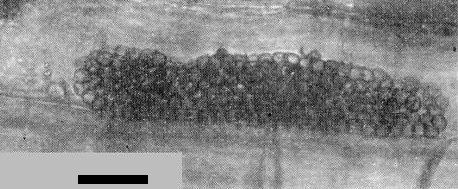
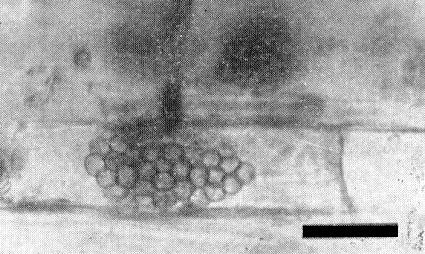
Transmission by a soil-borne organism with long-lived resting spores was indicated by retention of infectivity in soil after passage through a 44 µm screen, and while in storage for more than 8 years. Infectivity of soil was destroyed by heating to 52°C for 30 min or treating with captan or some other fungicidal chemicals (Slykhuis, 1970). Several zoosporic fungi are usually present on the roots of plants infected from soil (Barr & Slykhuis, 1969), but unpublished results of tests with cultures containing single fungus species show that transmission is associated only with Polymyxa graminis (Fig. 5, Fig. 6).
Transmission through Seed
None found in Triticum aestivum.
Transmission by Dodder
Not reported.
Serology
No antiserum has been prepared.
Relationships
The virus differs morphologically from soil-borne wheat mosaic virus which has tubular particles (Brakke, 1971b). It appears more closely related to other soil-borne viruses with filamentous particles including oat mosaic virus (Hebert & Panizo, 1975), and the viruses causing barley yellow mosaic, rice necrosis mosaic and wheat yellow mosaic in Japan (Inouye & Saito, 1975; Inouye, 1968, 1969). All are associated with P. graminis. Wheat spindle streak mosaic virus has longer particles than the other soil-borne viruses, frequently exceeding 2000 nm. It has the lowest temperature range for symptom development of any known plant virus, 5° to 17°C with the optimum about 10°C (Slykhuis, 1974).
Stability in Sap
The virus is very unstable in sap undiluted or diluted in water. When leaves are ground in 0.1 M Na2SO3 or 0.1 M phosphate buffer at pH 7, infectivity is retained 3-4 days at 5°C or 1 h at 20°C. The virus is inactivated in 10 min at 50°C. Leaves that were freeze-dried or stored in liquid nitrogen for the American Type Culture Collection retained a low level of infectivity after one year.
Purification
Particles were concentrated from juice extracted from wheat leaves in 0.5 M sodium borate buffer at pH 9.0, then emulsified in 1/3 volume of a 1:1 mixture of n-butanol and chloroform and the final pellet resuspended in phosphate buffer, but none of these preparations was infective. Examination with the electron microscope indicated that all particles were fragmented. It appears that the particles, being exceedingly long filaments, may be difficult to concentrate without fragmentation (Slykhuis & Polak, 1971).
Properties of Particles
Not reported.
Particle Structure
Particles are filamentous (Fig. 1), with measurements ranging from 190-1975 x 12.8 nm in leaf dip preparations (Slykhuis & Polak, 1971). In thin-sectioned wheat leaves the particles measured 18-20 nm in diameter, and had lengths exceeding 3000 nm (Hooper & Wiese, 1972; Langenberg & Schroeder, 1973).
Particle Composition
Unknown.
Relations with Cells and Tissues
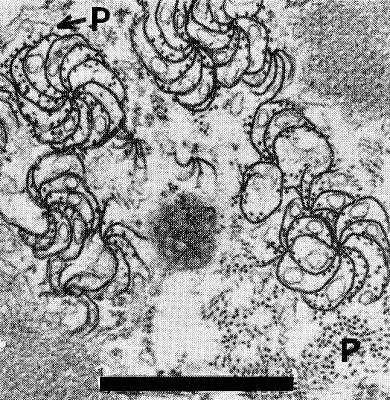
Complex membraneous bodies, cylindrical inclusions (pinwheels), virus-like rods, and associated crystalline materials occur in the cytoplasm of all leaf cell types except sieve elements and differentiated xylem cells (Fig. 3, Fig. 4). Similar inclusions were observed in cortical, endodermal and vascular parenchyma cells in roots of affected plants. All inclusions are apparent by the time the leaves develop visible chlorosis and necrosis. In such tissues the cellular constituents and the inclusions degenerate, filling the cells with their debris (Hooper & Wiese, 1972).
Notes
Wheat spindle streak mosaic virus is distinguished from other viruses that infect wheat by the extremely long, filamentous particles, frequently exceeding 2000 nm in length, and the lowest temperature range for symptom development known for a plant virus. It does not infect rye or barley, which are susceptible to soil-borne wheat mosaic virus (Brakke, 1971b) and barley stripe mosaic virus (Atabekov & Novikov, 1971); oats, which are susceptible to wheat streak mosaic virus (Brakke, 1971a); or Agropyron repens which is susceptible to agropyron mosaic virus (Slykhuis, 1973b).
Figures

Threadlike particles in a negatively stained leaf dip preparation from wheat infected with wheat spindle streak mosaic virus. Bar represents 1 µm.

Pairs of the four youngest leaves from two wheat plants with wheat spindle streak mosaic. Note that one of the youngest leaves (left) is symptomless, the other has light green spindle-shaped dashes near the tip. The successively older leaves have more pronounced streaks progressing to chlorotic and necrotic blotches.

Section through a mesophyll cell of a wheat leaf infected with wheat spindle streak mosaic virus showing longitudinal section of pinwheels (PW) an aggregate of rodlike particles (P) and a dense membranous inclusion (M) (Wiese & Hooper, 1971). Bar represents 1 µm.
References list for DPV: Wheat spindle streak mosaic virus (167)
- Atabekov & Novikov, CMI/AAB Descriptions of Plant Viruses 68, 4 pp., 1971.
- Barr & Slykhuis, Can. Pl. Dis. Surv. 49: 112, 1969.
- Brakke, CMI/AAB Descriptions of Plant Viruses 48, 4 pp., 1971a.
- Brakke, CMI/AAB Descriptions of Plant Viruses 77, 4 pp., 1971b.
- Gates, Can. Pl. Dis. Surv. 49: 58, 1969.
- Hebert & Panizo, CMI/AAB Descriptions of Plant Viruses 145, 4 pp., 1975.
- Hooper & Wiese, Virology 47: 664, 1972.
- Inouye, Ann. phytopath. Soc. Japan 34: 301, 1968.
- Inouye, Nogaku Kenkyu 53: 61, 1969.
- Inouye & Saito, CMI/AAB Descriptions of Plant Viruses 143, 3 pp., 1975.
- Jackson, Bracker, Huber, Scott & Shaner, Pl. Dis. Reptr 59: 790, 1975.
- Langenberg & Schroeder, Virology 55: 218, 1973.
- Slykhuis, Can. Pl. Dis. Surv. 40: 43, 1960.
- Slykhuis, Phytopathology 60: 319, 1970.
- Slykhuis, Can. J. Pl. Sci. 53: 477, 1973a.
- Slykhuis, CMI/AAB Descriptions of Plant Viruses 118, 4 pp., 1973b.
- Slykhuis, Phytopathology 64: 554, 1974.
- Slykhuis, Phytopathology 65: 582, 1975a.
- Slykhuis, Phytopathology 65: 1133, 1975b.
- Slykhuis, Phytopathology 66: 130, 1976.
- Slykhuis & Polak, Phytopathology 61: 569, 1971.
- Wiese & Hooper, Phytopathology 61: 331, 1971.
- Wiese, Ravenscroft & Everson, Pl. Dis. Reptr 58: 522, 1974.
- Wiese, Saari, Clayton & Ellingboe, Pl. Dis. Reptr 54: 635, 1970.
- Williams, Pirone, Slykhuis & Tutt, Pl. Dis. Reptr 59: 888, 1976.