Details of DPV and References
DPV NO: 181 September 1977
Family: Tombusviridae
Genus: Dianthovirus
Species: Red clover necrotic mosaic virus | Acronym: RCNMV
Red clover necrotic mosaic virus
M. Hollings Glasshouse Crops Research Institute, Littlehampton, Sussex, BN16 3PU, England
Olwen M. Stone Glasshouse Crops Research Institute, Littlehampton, Sussex, BN16 3PU, England
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Musil & Matisova (1967) and
Musil (1969a).
An RNA-containing virus with isometric particles c. 27 nm diameter, found in red clover (Trifolium pratense) in Europe. The virus is readily sap- transmissible to a fairly wide range of herbaceous plant species. No vector is known, but the virus can be experimentally transmitted in soil.
Main Diseases
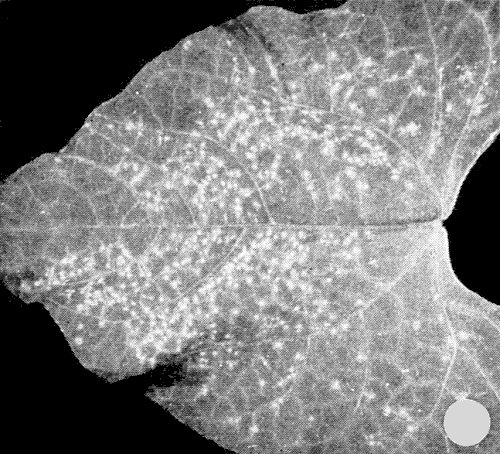
In naturally infected and in inoculated red clover, severe leaf mottle, distortion and necrotic areas (Fig. 4), with moderate to severe stunting, occur in the winter; in summer, infection is often masked. Mosaic symptoms develop in naturally infected Melilotus officinalis (Musil, 1969a), and severe chlorotic mosaic, stunting and necrotic flecks in experimentally infected Trifolium incarnatum (Fig. 1) (M. Hollings & O. M. Stone, unpublished information).
Geographical Distribution
Czechoslovakia (Musil, 1969a, 1969b); Poland (Kowalska, 1974); Sweden (Gerhardson & Lindsten, 1973); Britain (Hollings & Stone, 1974).
Host Range and Symptomatology
Different strains of the virus infected at least 47 of 106 plant species in 15 of 30 plant families (M. Hollings & O. M. Stone, unpublished information).
-
Diagnostic species
- Chenopodium quinoa.
Partially or completely necrotic local lesions in 3-6 days, subsequently enlarging slightly (Fig. 3). No systemic infection. - C. capitatum. Pin-point necrotic local lesions in 10-14 days; systemic
chlorosis, crinkle-mottle, rosetting, stunting and leaf distortion
(Fig. 8).
- Cucumis sativus (cucumber) cv. Butcher’s Disease Resister. Necrotic local lesions in cotyledons, fewer lesions in true leaves, after c. 7 days; no systemic infection.
- Gomphrena globosa. Whitish necrotic local lesions in 3 days, often becoming necrotic rings 4 mm in diameter. No systemic infection.
- Nicotiana clevelandii. Partially or completely necrotic local lesions and rings in 4-7 days. Systemic chlorotic rings, mottle, severe puckering and some leaf distortion and stunting (Fig. 2).
- N. glutinosa. Faint, chlorotic or necrotic local lesions, or symptomless infection of inoculated leaves, according to virus strain. No systemic infection.
- N. tabacum (tobacco) White Burley and Havana 425. Few partially necrotic local lesions in 3-5 days with the Swedish strain; chlorotic ring local lesions with the Czechoslovakian strains TpM-34 and TpM-48; almost symptomless infection of inoculated leaves with English H and Scottish strains. No systemic infection.
- Ocimum basilicum (basil). Necrotic brown local lesions in c. 6 days; no systemic infection.
- Phaseolus vulgaris (French bean) cv. The Prince. Numerous, small pale chlorotic to partially necrotic local lesions, often forming ringspots, in 3-5 days (Fig. 5), enlarging to necrotic areas and often killing the inoculated leaves. Systemic yellow and brown flecks, stunting, leaf distortion and necrosis often follow.
- Vicia faba (broad bean). Faint chlorotic local lesions (English H strain), or symptomless infection of inoculated leaves (Swedish strain). Not systemic.
- Vigna sinensis (cowpea) cv. Blackeye. Reddish-brown necrotic local lesions 1 mm diameter in 5-6 days with English H and Scottish strains; no systemic infection. Similar local lesions occur with the Swedish strain, often followed by systemic mottle and apical necrosis. With the Czechoslovakian strain TpM-34, numerous conspicuous chlorotic local lesions, 1-2 mm diameter, develop in 4-5 days, each lesion developing a red-brown necrotic margin and expanding to become confluent with adjacent lesions; there is systemic dwarfing and chlorosis. With strain TpM-48, few, rather faint chlorotic local lesions c. 1 mm diameter in 5-7 days, are sometimes followed by systemic dwarfing and necrotic veinal flecks.
- Tetragonia expansa. Very small, whitish necrotic local lesions in 5-6 days; systemic infection only with strain TpM-34, visible as faint mottle and leaf buckling.
- Cucumis sativus (cucumber) cv. Butcher’s Disease Resister. Necrotic local lesions in cotyledons, fewer lesions in true leaves, after c. 7 days; no systemic infection.
-
Propagation species
- Nicotiana clevelandii
and Phaseolus vulgaris are useful for virus propagation, and N. clevelandii for maintaining virus cultures.Assay species
- Chenopodium quinoa
and Phaseolus vulgaris give satisfactory local lesions.
Strains
Five Czechoslovakian isolates were divided into two serological types, designated A and B (Musil, 1969b). Most Swedish isolates tested were related to both Czechoslovakian A and B serotypes (Gerhardson & Lindsten, 1973), but antiserum to one Swedish isolate did not react with preparations of several other Swedish strains of the virus.
Five isolates from England, Scotland, Sweden and Czechoslovakia differed only slightly in host range and reactions, but showed considerable serological differences (Hollings & Stone, 1974, and unpublished information). English isolate H was serologically very closely related to Czechoslovakian isolate TpM-48 (serotype B), but much more distant serological relationships were found among isolates S (Scotland), Sw (Sweden), TpM-34 (Czechoslovakia, serotype A) and H (England) (Fig. 7) (Hollings et al., 1977b, and unpublished information).
Transmission by Vectors
No transmission by the aphids Myzus persicae, Aphis fabae, Aphis craccivora or Acyrthosiphon pisum (Gerhardson & Lindsten, 1973; Hollings & Stone, 1974).
The virus is exuded into soil from roots of infected Nicotiana clevelandii and Trifolium pratense, and can infect healthy N. clevelandii (but apparently not T. repens) bait seedlings planted in such soil, possibly in a manner similar to that shown by cymbidium ringspot virus (Hollings et al., 1977a).
Transmission through Seed
No seed transmission was detected in Trifolium pratense (Gerhardson & Lindsten, 1973), or in T. incarnatum, Nicotiana clevelandii, Phaseolus vulgaris or Trigonella foenum-graecum (Hollings & Stone, 1974).
Transmission by Dodder
No information.
Serology
The virus is a good immunogen and antigen in vitro. Antisera prepared in rabbits by one intravenous, plus two intramuscular injections with Freund’s complete adjuvant (0.5-1.5 mg virus/injection) had specific titres of 1/1024 to 1/4096 in tube precipitin tests, producing somatic (granular) precipitates. In immunodiffusion, a single line of precipitation is formed (Fig. 7), and serum titres ranged from 1/512 to 1/2048 (Hollings & Stone, 1974, and unpublished information).
Relationships
This virus resembles several other small, stable isometric viruses in particle morphology and in vitro properties, and in having one capsid polypeptide and one genome segment of ss-RNA. However, none of the five strains mentioned above reacted with antisera to 60 strains of 45 isometric viruses, nor did antisera to the five strains react with purified preparations of 33 of these other viruses (M. Hollings & O. M. Stone, unpublished information).
Stability in Sap
The virus is very stable in sap of Nicotiana clevelandii. It was still infective at dilutions of 5 x 10-4, and most strains withstood 10 min at 85°C, occasionally 90°C. However, 80% of infectivity of strain TpM-48 was lost at 70°C. Infectivity survived 9-10 weeks at 20°C, and at least 43 weeks at 2°C. Ampoules of lyophilized sap of N. clevelandii were still infective after 4 years at 20°C under high vacuum (M. Hollings & O. M. Stone, unpublished information).
Purification
The virus is readily purified from systemically infected Nicotiana clevelandii or from inoculated leaves of Phaseolus vulgaris (Hollings & Stone, 1974; Hollings et al., 1977b): harvest plants 2½-3 weeks after infection (7-10 days with P. vulgaris), homogenise leaves in 0.05 M phosphate buffer pH 7.6 containing 0.01 M EDTA and 0.14% (v/v) 2-mercaptoethanol (4 ml buffer/g tissue), and filter through cloth. Add n-butanol dropwise (9.3 ml/100 ml juice), and stir for 1 h. Concentrate the virus by one or more cycles of differential centrifugation (25 min at 12,000 g; 90 min at 78,000 g). Re-suspend the final pellets in 0.03 M phosphate buffer (or distilled water) (1 ml/30 g original leaf) and, after 1-2 h, remove insoluble material by brief centrifugation (10 min at 10,000 g). Yields up to 30-70 mg virus/kg leaf tissue can be obtained. Under high glasshouse temperatures during summer, Nicotiana spp. often give low yields of the virus; plants kept in growth chambers at c. 20°C and 16 h/day illumination (c. 5000 lux) consistently give good virus yields.
Further purification can be obtained by centrifugation in discontinuous density gradients of sucrose, and re-pelleting the virus from the specific band (Marcinka et al., 1969). Good purification is also obtained by applying 1.0-1.5 ml of partially purified preparations to columns (85 x 1.5 cm) of controlled pore glass beads (70 nm pore size) and eluting the virus in the void volume by adding 0.04 M phosphate buffer (Barton, 1977).
Properties of Particles
Purified preparations usually produce a single light-scattering band in density-gradient centrifugation, but the Swedish isolate sometimes gives 2 bands. In analytical ultracentrifugation, a single component is obtained, with s°20,w=130-135 S (M. Hollings & O. M. Stone, unpublished information).
A260/A280: 1.63-1.69; Amax(260)/Amin(242): 1.31 (values corrected for light-scattering).
Buoyant density: in CsCl, one major component at 1.363 g/ml and one minor one at 1.356 g/ml (Swedish strain) (M. Hollings & A. A. Brunt, unpublished information).
Electrophoretic behaviour: in immunoelectrophoresis in 0.8% Ionagar no. 2 in 0.03 M phosphate buffer pH 7.6, differences occurred between strains of the virus: all five strains contained one antigenic component moving towards the cathode; the Swedish and TpM-34 strains moved extremely slowly, strain TpM-48 moved more rapidly, and the English H and Scottish strains very much faster (M. Hollings & O. M. Stone, unpublished information).
Particle Structure
In 2% phosphotungstate (pH 6.5) particles are isometric, c. 27 ± 1.1 nm diameter (M. Hollings & O. M. Stone, unpublished information) or c. 30 nm diameter (Marcinka et al., 1969) (Fig. 6).
Particle Composition
Nucleic acid: RNA, single-stranded, of M. Wt c. 1.55 x 106, comprising about 20% of particle weight estimated spectophotometrically (M. Hollings & R. J. Barton, unpublished information).
Protein: In polyacrylamide gel electrophoresis, a single capsid polypeptide of M. Wt 39,500 (H isolate), or 40,800 (Swedish isolate) (M. Hollings & R. J. Barton, unpublished information).
Relations with Cells and Tissues
The virus is present in leaves, stems and roots of infected clovers. Amoeboid intracellular inclusion bodies occur sometimes in Nicotiana clevelandii, although usually in fewer than 5% of cells, but apparently do not occur in clovers (O. M. Stone & M. Hollings, unpublished information).
Notes
The symptoms in clovers are seldom entirely diagnostic, and differ in severity according to the strain of the virus and the growing conditions of the plant.
The brown necrotic flecks, usually produced in at least a few leaves by this virus, distinguish it from most other viruses found in clovers, such as alfalfa mosaic, clover yellow mosaic, clover yellow vein, red clover mottle and red clover vein mosaic. But some necrotic strains of white clover mosaic virus can induce symptoms very like those of red clover necrotic mosaic virus.
After 8 weeks thermotherapy at c. 36°C, all cuttings taken from Nicotiana clevelandii plants infected with strain H or S were virus-free (M. Hollings & O. M. Stone, unpublished information).
Figures
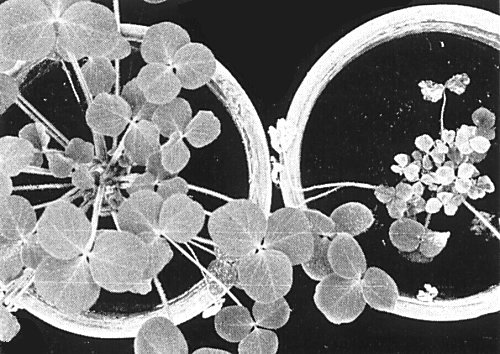
Severe stunting, chlorosis, mottle and necrotic flecks in Trifolium incarnatum (right), 6 weeks after experimental infection in winter with the English H strain of the virus. Healthy plant (left).

Systemic mottle, puckering and leaf malformation in Nicotiana clevelandii 2 weeks after infection with the Scottish strain.

Systemic mosaic, necrotic flecks and leaf distortion in Trifolium pratense, 6 weeks after infection with the Scottish strain.
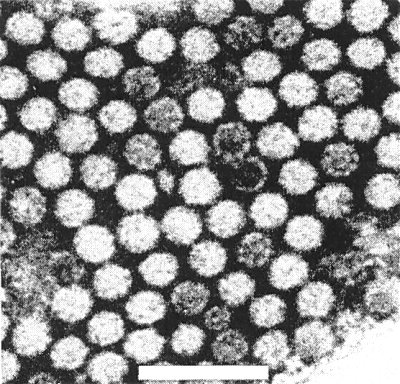
Partially purified preparation of the virus (strain H), stained in phosphotungstate. Bar represents 100 nm.

Immunodiffusion test, showing serological relationships between 5 strains of the virus. Central well = antiserum to strain H. Peripheral wells contain purified virus preparations of: English strain (H); Swedish strain (Sw); Scottish strain (Sc); Czechoslovak strain TpM-34 (4); and Czechoslovak strain TpM-48 (5).
References list for DPV: Red clover necrotic mosaic virus (181)
- Barton, J. gen. Virol. 35: 77, 1977.
- Gerhardson & Lindsten, Phytopath. Z. 76: 67, 1973.
- Hollings & Stone, Rep. Glasshouse Crops Res. Inst. for 1973: 122, 1974.
- Hollings, Stone & Barton, Ann. appl. Biol. 85: 233, 1977a.
- Hollings, Stone & Pawley, Rep. Glasshouse Crops Res. Inst. for 1976: 129, 1977b.
- Kowalska, Rocznik nauk rolniczych Series E4: 89, 1974.
- Marcinka, Musil & Mrena, Acta virol., Prague 13: 247, 1969.
- Musil, Biologia Bratisl. 24: 33, 1969a.
- Musil, Acta virol., Prague 13: 226, 1969b.
- Musil & Matisova, Ochr. Rost. 3: 225, 1967.