Details of DPV and References
DPV NO: 182 September 1977
Family: Caulimoviridae
Genus: Caulimovirus
Species: Carnation etched ring virus | Acronym: CERV
Carnation etched ring virus
R. H. Lawson US Department of Agriculture, Agricultural Research Service, Beltsville, Maryland 20705, USA
Suzanne S. Hearon US Department of Agriculture, Agricultural Research Service, Beltsville, Maryland 20705, USA
F. L. Civerolo US Department of Agriculture, Agricultural Research Service, Beltsville, Maryland 20705, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Hollings & Stone (1961).
A DNA-containing virus with isometric particles about 45 nm in diameter occurring in cytoplasmic inclusion bodies. Transmissible by mechanical inoculation and by Myzus persicae. It is found naturally only in Dianthus caryophyllus. Distribution world-wide.
Main Diseases
Causes etched ring disease, characterized by necrotic flecks, rings and line patterns on the leaves of carnation, Dianthus caryophyllus. These necrotic leaf patterns may enlarge to form blotches (Fig. 1) without conspicuous chlorosis. More severe symptoms including leaf chlorosis, necrotic spots and rings, and stem streaks and flecks are associated with mixed infection by carnation etched ring and carnation mottle viruses (Hakkaart, 1968). Expression of carnation etched ring symptoms may also be intensified in mixed infections with unidentified viruses having 25 nm and 29 nm particles (Hollings, Stone & Bouttell, 1968). Symptom development in carnation is temperature-dependent. Plants of cv. Peace River grown with 15,000 lux fluorescent illumination show necrotic rings, spots and patches at 21°C, but are symptomless at 26°C (R. H. Lawson & S. S. Hearon, unpublished data). Symptoms are most conspicuous on D. caryophyllus from March-June in young vegetative growth in the greenhouse (Paludan, 1970).
Geographical Distribution
World-wide.
Host Range and Symptomatology
Only members of the Caryophyllaceae are infected. The virus is transmissible by
mechanical inoculation with crude sap and by Myzus persicae.
Diagnostic species
cv. Pink Beauty. Mechanically inoculated plants develop
several different types of symptom separately, or in combination. The symptoms may
be (1) concentric red rings about 2 to 5 mm in diameter appearing after 10 to 30 days
on inoculated leaves (Fig. 2, Fig. 3);
(2) necrotic flecks and patches often appearing
on the midvein at the base of the leaf and causing collapse of systemically invaded
leaves; necrotic patches may also be on inoculated leaves; (3) yellow spots, lines and
rings on the inoculated leaves or one or two leaf pairs above the inoculated leaves
(Fig. 4); (4) shortening of the internodes mainly in the lower to mid portion of the
plant (Hakkaart, 1974).
Leaf curl and red lines develop on one or two leaf pairs above the inoculated leaves on some plants.
Propagation and assay species
Strains
None reported.
Transmission by Vectors
Transmitted by the aphid Myzus persicae to Silene armeria (Hollings & Stone, 1973). The virus was also transmitted from Saponaria vaccaria to S. vaccaria by Myzus persicae after starving for 2 h, then an acquisition feeding period of 10 min followed by an inoculation access period of 24 h (F. F. Smith & R. H. Lawson, unpublished data).
Transmission through Seed
None reported.
Transmission by Dodder
None reported.
Serology
The virus is moderately immunogenic. Injection of a rabbit with a total of 100 µg of partially purified virus in three intravenous injections gave antiserum, collected 20 days after the first injection, that had a titre of 1/256 in microprecipitin tests (Lawson & Civerolo, 1976). Antiserum reacted with the homologous antigen in agar gel double diffusion tests (Hollings & Stone, 1969).
Relationships
Concentrated preparations of the virus reacted with antisera to dahlia mosaic virus or cauliflower mosaic virus (Hollings & Stone, 1969; Brunt, 1971). The relationship between carnation etched ring and cauliflower mosaic viruses was confirmed in microprecipitin tests with partially purified antigens (Lawson & Civerolo, 1976).
Stability in Sap
In S. vaccaria sap the thermal inactivation point (10 min) is between 80 and 85°C; dilution end-point is between 10-3 and 10-4. The virus retains infectivity in frozen Saponaria leaf tissue for 4 months and isolated inclusion bodies retain infectivity when stored in water at 4°C for at least 7 months.
Purification
The virus has been purified from Dianthus caryophyllus (Fujisawa, Rubio-Huertos & Matsui, 1971) and Saponaria vaccaria (Lawson & Civerolo, 1976). We estimate that the yield of virus from S. vaccaria (50-400 µg/100 g tissue) is 5-6 times greater than the yield from Silene armeria (Fujisawa, Rubio-Huertos & Matsui, 1972). The S. vaccaria plants are inoculated when they have four true leaves with the upper two leaves about half expanded. Harvest inoculated leaves, together with one or two pairs of systemically infected leaves immediately above the inoculated leaves, 18-20 days after inoculation. Homogenize fresh or frozen tissue in 4-5 volumes of 0.1 M phosphate buffer, pH 7.2, add n-butanol to 8% (v/v) and stir for 2 h. Add urea to 6% (w/v) and Triton X-100 to 2.5% (v/v) and stir the extract for an additional 16-17 h. Centrifuge the extract at 5,000 g for 15 mm and pour the supernatant fluid through cheesecloth. Concentrate the virus by differential centrifugation and purify further by centrifugation in sucrose density gradients (Lawson & Civerolo, 1976).
Properties of Particles
A sedimentation coefficient (s20,w) of 206 S is reported (Fujisawa et al., 1971), similar to that of cauliflower mosaic virus.
Particle Structure
Virus particles from the major UV-absorbing zone in sucrose density gradients in phosphate buffer when mounted in 2% potassium phosphotungstate, pH 7.0, are 42-44 nm in diameter (85 particles measured) (Fig. 5). Virus particles separated from a sucrose density gradient in phosphate buffer were 47 nm in diameter (Fujisawa et al., 1971). Virus particles are also associated with a second small peak below the major UV-absorbing zone (R. H. Lawson & E. L. Civerolo, unpublished data). They occur in clusters with amorphous material apparently binding them together. The particle clusters may originate from fragments of inclusion bodies. There was no evidence of regular aggregates of dimers or trimers. In leaf-dip preparations stained with neutral 2% KPTA, 85% of 64 particles measured 44-47 nm and the remaining particles measured 47-50 nm. Results were similar when virus particles in leaf-dip preparations were stained with neutral 3% ammonium molybdate.
Particle Composition
Nucleic acid: DNA: The nucleic acid isolated from purified virus is completely digested by deoxyribonuclease (DNase) but not by ribonuclease (Fujisawa et al., 1972). Infectivity is destroyed by DNase (Lawson & Civerolo, 1976). The DNA occurs as short linear forms 0.38-0.62 µm long, with some molecules measuring 1.8-2.1 µm, and circular molecules of 2.15-2.25 µm (Fig. 6). Some preparations contain many highly twisted forms that lack free ends and are apparently circular. The M.Wt of the circular molecules is c. 4.21-4.31 x 106 daltons. Virus DNA preparations contain three electrophoretic components labelled A, B and C in order of migration toward the anode in 1.8-2.0% polyacrylamide-0.5% agarose gels in 0.05 M Tris, 0.02 M Na-acetate, 0.002 M EDTA, 0.018 M NaCl (pH 8.0) and electrophoresed at 2.5 to 3.3 V/cm for 12 h at room temperature (Fig. 7). Gel slices including the A and B components contained some highly twisted, apparently circular forms. The C component contained only linear forms of various lengths.
Protein: No information.
Relations with Cells and Tissues
Virus particles occur in the cytoplasm in association with electron-dense inclusion bodies of irregular shape, varying in size up to about 10 µm and enlarging with increasing age of infection. The most elementary forms of the inclusion in Saponaria vaccaria leaves are small electron-dense areas that apparently fuse to form larger dense matrices with associated ribosomes and virus particles (Fig. 8). The fully formed inclusion bodies have a defined margin (Fig. 9). Inclusions occur in palisade and mesophyll cells and can be readily observed in epidermal cells stained with 1% phloxine B, or when extracted and concentrated (Fig. 10). Inclusions are abundant in the inoculated leaves of S. vaccaria but are more difficult to detect in D. caryophyllus. The dense staining matrix material in the inclusion body is mostly protein and can be digested with protease before the virus particles are affected (Hearon & Lawson, 1975). Virus particles are present in the nuclei of Dianthus barbatus (Rubio-Huertos et al., 1972) and S. vaccaria (Lawson & Hearon, 1974) (Fig. 11), but not D. caryophyllus. Some nuclei in infected S. vaccaria contain virus particles and membranes (Fig. 11). Virus particles are present in modified plasmodesmata (Lawson & Hearon, 1974) and are associated with enlarged channels and wall thickenings similar to those found in plants infected with dahlia mosaic (Kitajima & Lauritis, 1969), cauliflower mosaic (Conti et al., 1972) and mirabilis mosaic viruses (Brunt & Kitajima, 1973).
Notes
The virus was eliminated from D. caryophyllus by meristem tip culture and by a combination of heat treatment and meristem tip culture (Paludan, 1970).
S. vaccaria is also susceptible to carnation mottle, carnation ringspot, and carnation vein mottle viruses, an uncharacterized 29 nm isometric virus (Hakkaart, 1974) and tobacco mosaic virus. The severe symptoms caused by carnation mottle virus may mask the milder symptoms caused by other carnation viruses. Carnation streak virus (Poupet et al., 1975) also infects S. vaccaria. This virus may be the same as carnation necrotic fleck virus (Inouye & Mitsuhata, 1973) and carnation yellow fleck virus (Smookler & Loebenstein, 1974).
Figures

Mild necrotic etch (right) and large necrotic blotches (left) on leaves of naturally-infected Dianthus caryophyllus cv. Peace River.
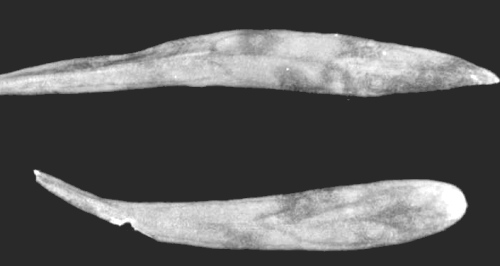
Concentric red rings in inoculated leaves of Saponaria vaccaria cv. Pink Beauty after 10-13 days on plants grown at 21-26°C with fluorescent and incandescent illumination at 10,000-12,000 lux for 12 h per day.
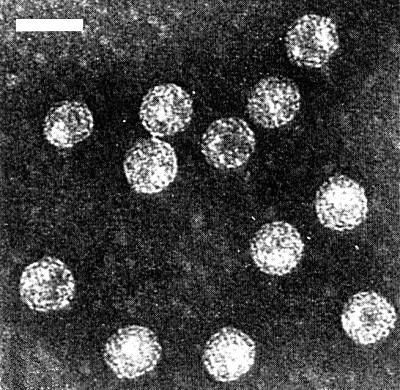
Particles in a partially purified preparation stained with potassium phosphotungstate. Bar represents 50 nm.

DNA showing a circular molecule (left) and a linear molecule (right). Nucleic acid was spread in 1 M ammonium acetate with 0.01% cytochrome C on a 0.15 M ammonium acetate hypophase. Bar represents 200 nm.

Electrophoresis of CERV nucleic acid in a composite 2.0% polyacrylamide-0.5% agarose gel. Note components A, B and C. Migration is from - to +

Aggregate of small electron-dense inclusions forming a larger body with an irregular profile in the cytoplasm of S. vaccaria. The dense matrix is surrounded by ribosomes; virus particles occur around the margin of the mature inclusions or embedded in the small bodies that form the larger inclusions. Bar represents 1 µm.
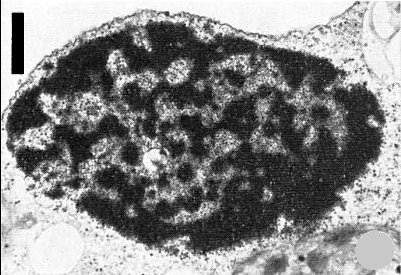
Large inclusion body with a more regular outline and at a more advanced stage of development than in Fig. 8. Bar represents 1 µm.
References list for DPV: Carnation etched ring virus (182)
- Brunt, CMI/AAB Descriptions of Plant Viruses 51, 4 pp., 1971.
- Brunt & Kitajima, Phytopath. Z. 76: 265, 1973.
- Conti, Vegetti, Bassi & Favali, Virology 47: 694, 1972.
- Fujisawa, Rubio-Huertos & Matsui, Phytopathology 61: 681, 1971.
- Fujisawa, Rubio-Huertos & Matsui, Phytopathology 62: 810, 1972.
- Hakkaart, Neth. J. Pl. Path. 74: 150, 1968.
- Hakkaart, Acta Hort. 36: 35, 1974.
- Hearon & Lawson, Proc. Am. Phytopath. Soc. 2: 52, 1975.
- Hollings & Stone, Rep. Glasshouse Crops Res. Inst., 1960: 94, 1961.
- Hollings, Stone & Bouttell, Rep. Glasshouse Crops Res. Inst., 1967: 94, 1968.
- Hollings & Stone, Rep. Glasshouse Crops Res. Inst., 1968: 102, 1969.
- Hollings & Stone, Rep. Glasshouse Crops Res. Inst., 1972: 102, 1973.
- Inouye & Mitsuhata, Ber. Ohara Inst. landw. Biol. 15: 195, 1973.
- Kitajima & Lauritis, Virology 37: 681, 1969.
- Lawson & Civerolo, Acta Hort. 59: 49, 1976.
- Lawson & Hearon, J. Ultrastruct. Res. 48: 201, 1974.
- Paludan, Tidsskr. PlAvl. 74: 75, 1970.
- Poupet, Cardin, Marais & Cadilhac, Annls Phytopath. 7: 277, 1975.
- Rubio-Huertos, Castro, Fujisawa & Matsui, J. gen. Virol. 15: 257, 1972.
- Smookler & Loebenstein, Phytopathology 64: 979, 1974.



