Details of DPV and References
DPV NO: 189 August 1978
Family: Virgaviridae
Genus: Unassigned Virgaviridae
Species: Nicotiana velutina mosaic virus | Acronym: NVMV
Nicotiana velutina mosaic virus
J. W. Randles Department of Plant Pathology, Waite Agricultural Research Institute, The University of Adelaide, Glen Osmond, South Australia, 5064
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Randles, Harrison & Roberts (1976).
An RNA-containing virus with straight rod-shaped particles 18 nm in diameter, and a large range of lengths, the commonest being 125-150 nm. Transmissible through seed, and by mechanical inoculation of sap to plants in three families. Has no known economic importance.
Main Diseases
Isolated from a Nicotiana velutina plant showing bright yellow mosaic.
Geographical Distribution
Only known occurrence is adjacent to a dam 8 km from Lake Frome Station Homestead, in the semi-arid region of South Australia.
Host Range and Symptomatology
Fairly readily transmissible by inoculation of sap. Infects species in the Solanaceae (including many Nicotiana species) and Chenopodiaceae; Gomphrena globosa (Amaranthaceae) is the only known host outside these families. Symptom intensity and speed of development vary with light and temperature conditions, and are favoured by continuous light of approximately 5000 lux at 25°C.
-
Diagnostic species
- Nicotiana glutinosa.
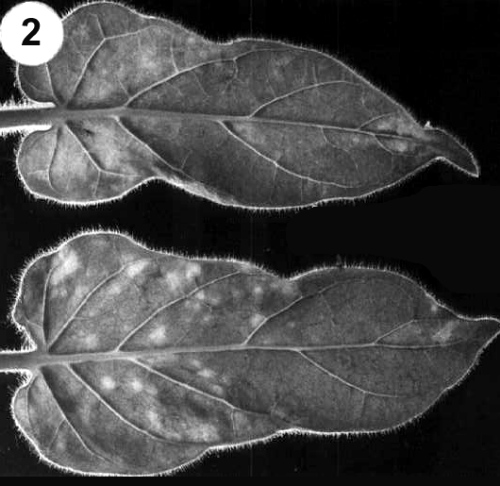
Inoculated leaves are usually symptomless but may develop chlorotic rings. Systemic vein chlorosis and mosaic persist throughout the life of the plant (Fig. 2). -
Nicotiana tabacum (tobacco) cv. Xanthi-nc. Inoculated leaves develop
delicate necrotic rings and lines. A systemic mild mosaic is produced.
- Nicotiana debneyi. Large spreading chlorotic blotches develop in inoculated leaves, sometimes accompanied by delicate necrotic rings. The first leaf with systemic symptoms becomes yellow, often with necrosis. Leaves produced later show a mild mosaic, rugosity, and narrowing of the lamina (Fig. 5).
- Chenopodium amaranticolor and C. quinoa. Chlorotic local lesions, becoming necrotic. Not infected systemically (Fig. 4).
- Beta macrocarpa. Chlorotic local lesions and systemic mosaic (Fig. 3).
- Gomphrena globosa. Enlarging local necrotic ringspots. Systemic chlorosis with crinkling and epinasty of leaves (Fig. 1).
- Tetragonia expansa is not a host.
- Nicotiana debneyi. Large spreading chlorotic blotches develop in inoculated leaves, sometimes accompanied by delicate necrotic rings. The first leaf with systemic symptoms becomes yellow, often with necrosis. Leaves produced later show a mild mosaic, rugosity, and narrowing of the lamina (Fig. 5).
-
Propagation species
- Nicotiana glutinosa
plants and seed are suitable for maintaining inoculum; N. debneyi is favoured as a source for virus purification.Assay species
- Chenopodium amaranticolor
and C. quinoa are useful local lesion hosts.
Strains
None found.
Transmission by Vectors
None found.
Transmission through Seed
Frequently transmitted through the seed of Nicotiana glutinosa (up to 72%), N. clevelandii, N. debneyi and N. rustica.
Serology
The virus is moderately to poorly immunogenic. Tube precipitin tests, and double diffusion in gels containing agarose at 0.5% and below are satisfactory. Observation of antigen-antibody agglutination by electron microscopy is favoured for studying cross-reactions because specific attachment of antibody to antigen can be observed, sensitivity is high, and requirements for reactants are small.
Relationships
Host range and structure of the virus suggest relatedness to the rod-shaped fungus-transmitted viruses such as potato mop-top virus, which are now regarded as tentative members of the tobamovirus group. However, the size of the coat protein subunits, and the frequent seed transmission suggest that it may also be allied to barley stripe mosaic virus.
No serological relationship has been found between Nicotiana velutina mosaic virus and potato mop-top, soil-borne wheat mosaic, tobacco mosaic, beet necrotic yellow vein, barley stripe mosaic or tobacco rattle viruses.
Stability in Sap
Sap of infected N. glutinosa or N. debneyi loses infectivity when stored from 1 to 4 days at room temperature. Infectivity declines in sap heated at 50°C for 10 min, and is lost after 10 min at between 60° and 70°. The dilution end-point of infective N. glutinosa sap, diluted in water and tested on N. glutinosa, is between 10-1 and 10-2.
Infectivity is enhanced and greatly stabilized by adding reducing agents (for example 0.002 M dithiothreitol or 0.05% thioglycerol), but not chelating agents (for example 0.01 M sodium diethyl dithiocarbamate), to preparations in 0.05 M Tris-HCl buffer, pH 8.1.
Purification
The purification method employs a high pH step to dissociate virus particles from rapidly sedimenting material. Clarification is achieved by sedimentation through 55% sucrose.
The following modification (D. Cartwright, unpublished data) of the original method of Randles et al. (1976) yields up to 20 mg of virus per kg of N. debneyi leaf. Blend tissue with 3 vol. 0.067 M phosphate buffer (pH 5.6; containing 0.01 M sodium diethyl dithiocarbamate and 0.05% thioglycerol), strain through muslin and centrifuge at 12,000 g for 20 min. Resuspend the pellet in 0.05 M glycine buffer (pH 9.0; containing 0.05% thioglycerol), stir 4-6 h. Clarify at 2500 g for 20 min, layer over 55% sucrose containing 0.05% thioglycerol, and centrifuge at 176,000 g for 90 min. Resuspend the pellet in 1-2 ml 0.05% thioglycerol for 16 h. Clarify and centrifuge again through 55% sucrose, resuspend for 2 h and centrifuge through a 10-40% linear sucrose gradient (buffered with 0.05 M glycine, pH 9.0) at 74,000 g for 25 min. Recover the very broad light-scattering zone and sediment the virus at 176,000 g for 40 min. Work at 4°C.
Properties of Particles
Ultraviolet absorption spectrum shows a minimum at 248-250 nm, maximum at 262-264 nm.
A260/A280: 1.15-1.22.
Particle Structure
Particles are straight rods, 18-19 nm in diameter (Fig. 6). In partially purified preparations most particles are 100-175 nm long. However, the length distribution is broad with the frequency of particles in the smaller size classes increasing during purification to give a peak in the 125-150 nm class. This suggests that particles are either fragile, or undergo progressive dissociation of end-to-end aggregates during purification. Particles have helical symmetry, with a pitch of 2.86 ± 0.03 nm when stained in ammonium molybdate or uranyl formate (Fig. 7).
Particle Composition
Nucleic acid: Nucleic acids extracted from infected leaf show a zone of infectivity between 15 S and 33 S when sedimented in sucrose density gradients. Infectivity of these extracts is lost upon treatment with ribonuclease A (1 µg/ml). Nucleic acid extracts from purified virus show a 2.3 x 106 M. Wt component and up to 8 smaller minor species when fractionated on polyacrylamide gels (D. Cartwright, unpublished data).
Protein: Subunits have M. Wt of 2.14 x 104.
Other components: None known.
Relations with Cells and Tissues
Particles were not seen in locally or systemically infected tissue, and no intracellular changes have been observed.
Notes
Nicotiana velutina mosaic virus resembles the seedborne rod-shaped barley stripe mosaic virus (Atabekov & Novikov, 1971), and also the fungus-transmitted tobamoviruses, such as potato mop-top virus (Harrison, 1974), soil-borne wheat mosaic virus (Brakke, 1971), beet necrotic yellow vein virus (Tamada, 1975; Putz, 1977), and possibly peanut clump virus (Thouvenel, Dollet & Fauquet, 1976). However, it is distinguishable from these by its host range, efficient seed transmissibility in some Nicotiana species, particle instability and serological uniqueness. A useful test which distinguishes it from all the above viruses is its ability to infect Nicotiana species and Gomphrena globosa, but not Tetragonia expansa.
Acknowledgements
Figs 1, 3, 6, and 7 by courtesy of Scottish Horticultural Research Institute.
Figures
References list for DPV: Nicotiana velutina mosaic virus (189)
- Atabekov & Novikov, CMI/AAB Descriptions of Plant Viruses 68, 4 pp., 1971.
- Brakke, CMI/AAB Descriptions of Plant Viruses 77, 4 pp., 1971.
- Harrison, CMI/AAB Descriptions of Plant Viruses 138, 4 pp., 1974.
- Putz, J. gen. Virol. 35: 397, 1977.
- Randles, Harrison & Roberts, Ann. appl. Biol. 84: 193, 1976.
- Tamada, CMI/AAB Descriptions of Plant Viruses 144, 4 pp., 1975.
- Thouvenel, Dollet & Fauquet, Ann. appl. Biol. 84: 311, 1976.