Details of DPV and References
DPV NO: 192 August 1978
Family: Geminiviridae
Genus: Begomovirus
Species: Bean golden mosaic virus | Acronym: BGMV
Isolates previously assigned to this virus are now classified as 2 separate viruses: Bean golden mosaic virus-Brazil (BGMV-BR) and Bean golden mosaic virus-Puerto Rico (BGMV-PR).
Bean golden mosaic virus
R. M. Goodman Department of Plant Pathology, University of Illinois, Urbana, Illinois, USA
J. Bird Agricultural Experiment Station, University of Puerto Rico, Mayaguez, Puerto Rico, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Disease first described by Costa (1965). Virus first purified by Galvez &
Castano (1976).
- Selected synonyms
- Mosaico dorado (Rev. Pl. Path. 51: 867)
- Golden-yellow mosaic virus (Rev. Pl. Path. 51: 4506)
- Golden-yellow mosaic virus (Rev. Pl. Path. 51: 4506)
- A DNA-containing virus with isometric particles about 19 nm in diameter occurring predominantly in pairs. The host range is apparently restricted to certain wild and cultivated species of the Leguminosae. Transmitted by the whitefly, Bemisia tabaci, and by inoculation with sap from young infected leaves.
Main Diseases
Causes bright yellow mosaic diseases of legumes, including Phaseolus vulgaris (French bean), P. lunatus (lima bean), and closely related wild species.
Geographical Distribution
Widely distributed in tropical and subtropical America (Bird & Maramorosch, 1975). Similar diseases reported in tropical Africa and Asia.
Host Range and Symptomatology
Known host range includes only certain species in the legume genera Phaseolus, Macroptilium, Vigna and Calopogonium (Bird et al., 1972; Bird, Sanchez & Vakili, 1973; Meiners et al., 1973.) The virus did not infect 21 species in 8 other families, or 6 species in the legume genera Cassia, Cajanus, Glycine, Rhynchosia and Phaseolus (J. Bird, unpublished data). The virus has been transmitted by inoculation with sap to Phaseolus vulgaris from P. vulgaris and Calopogonium mucunoides (Meiners et al., 1973; Galvez & Castano, 1976; Goodman, Bird & Thongmeearkom, 1977) and to P. lunatus and P. vulgaris from P. lunatus (H. Lot & J. Bird, unpublished data).
- Diagnostic species
- Phaseolus lunatus
(lima bean). Bright yellow systemic vein chlorosis developing into golden mosaic. - Phaseolus vulgaris (French bean). Irregularly shaped chlorotic spots
develop in young systemically infected leaflets followed by vein chlorosis and,
in advanced infections, by golden mosaic (Fig. 2). Leaves are usually deformed
and curled downward at the edges when infected plants are grown in growth
chambers but this symptom is not always found in the field. Plants are usually
stunted and the number of pods reduced.
- Propagation species
- Phaseolus vulgaris
cv. Top Crop grown at 31°C with 14 h day length (10,000 lux) is a good source for virus purification. Systemically infected leaves are harvested 9-12 days after manual inoculation of expanding primary leaves (Goodman et al., 1977; Goodman, 1977a). Yields average 1 mg per 100 g tissue.- Assay species
- Phaseolus vulgaris
cv. Top Crop sometimes shows chlorotic local lesions on inoculated primary leaves (Fig. 1). P. vulgaris cv. Top Crop and Red Kidney have been used for assaying transmission by vectors.
Strains
No information.
Transmission by Vectors
Transmissible by the whitefly Bemisia tabaci (Costa, 1965). Some authors distinguish races of this species (Bird et al., 1972); race sidae is the one reported to transmit this virus (Bird et al., 1972). Males and females were equally effective as vectors (Bird et al., 1973). Acquisition and inoculation by adults can be effected in a total time of less than 6 min (J. Bird, unpublished data), but efficient transmission requires longer feeding periods (Gamez, 1971; Bird et al., 1973). No tests have been reported to determine if a latent period exists or if larval forms can transmit. Individual adult whiteflies transmit intermittently for up to 16 days after acquisition (Gamez, 1971).
Transmission through Seed
Not seed-transmitted (Costa, 1965).
Serology
An antiserum has been prepared with a titre of 1/320 in Ouchterlony double diffusion tests against sap from infected plants (Goodman, unpublished data). The presence in infected beans of copious amounts of the highly immunogenic phytoferritin (Fig. 4, Fig. 6), which is present in negligible amounts in healthy plants, may complicate antiserum production and interpretation of serological tests unless the virus is scrupulously purified.
Relationships
Relationships among the whitefly-transmitted pathogens causing similar diseases of various species and in various countries are not understood. Disease symptoms and particle morphology suggest that bean golden mosaic viruses from Puerto Rico, El Salvador, Colombia, Guatamala, and probably Brazil are the same virus, although possible strain relationships are not worked out. Causal agents of similar diseases in other tropical areas have not been characterized.
Stability in Sap
In Phaseolus vulgaris sap the thermal inactivation point (10 min) is between 50° and 55°C, dilution end-point about 10-1 to 10-2, and longevity in vitro about 72 h at 23°C (Galvez & Castano, 1975; J. Bird, unpublished data).
Purification
Highest yields have been obtained by the following method (Goodman et al., 1977). Freeze systemically infected leaves (9-12 days after inoculation of plants) in dry ice. Crush frozen leaves to a coarse powder and grind in cold 0.1 M sodium phosphate, 10 mM disodium ethylenediamine-tetra acetate, 1 mM cysteine (pH 7.8). Strain through cheesecloth and clarify by centrifugation (12,000 g, 20 min, 4°C). Add NaCl to 0.2 M and polyethylene glycol (PEG) to 4% (w/v). Discard supernatant fluid containing phytoferritin. Resuspend virus from PEG pellet with 0.1 M sodium phosphate, pH 7.8. Clarify by centrifugation (12,000 g, 20 min) and subject to one or two cycles of high- and low-speed centrifugation, resuspending the high-speed pellets in the sodium phosphate buffer. Final purification is by sucrose density gradient centrifugation (Fig. 6). Work at 4°C throughout.
Properties of Particles
Sedimentation coefficient (s20,w): 69 S (Fig. 7).
M. Wt: about 2.6 x 106 daltons.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 7.7.
A260/A280: 1.4.
Particle Structure
Particles are isometric, about 19 nm diameter, and in aldehyde-fixed negatively stained preparations are predominantly in pairs (‘geminate’) (Fig. 3) (Galvez & Castano, 1976; Goodman et al., 1977). It is not certain whether the pairing of the particles is a fixation/staining artefact or real and, if real, whether it is of biological significance. The best electron micrographs available suggest icosahedral (T = 1) symmetry (Fig. 5). The particles disintegrate when mounted in phosphotungstate, uranyl acetate, or ammonium molybdate unless aldehyde-fixed (Galvez & Castano, 1976; Goodman et al., 1977).
Particle Composition
Nucleic acid: DNA, M. Wt about 0.75 x 106 daltons (Goodman, 1977b), comprising about 29% of particle mass. DNA is single-stranded (Goodman, 1977b) and resistant to exonucleases (R. M. Goodman, unpublished data). Infective DNA is easily prepared from purified virus by treatment with 1% sodium dodecyl sulphate at pH 9.0 or 0.3 M NaOH (Goodman, 1977a). DNA sediments as a single component under neutral or alkaline conditions with a sedimentation coefficient (s20,w) at pH 7.0, i = 0.1, of 16.0 S. Two components are resolved when analysis is by polyacrylamide gel electrophoresis in 8 M urea (R. P. Ricciardi & R. M. Goodman, unpublished data). Buoyant density in CsCl is 1.717 g/ml.
Protein: Subunits have M. Wt of about 3.1 x 104 daltons (T. L. Shock & R. M. Goodman, unpublished data).
Relations with Cells and Tissues
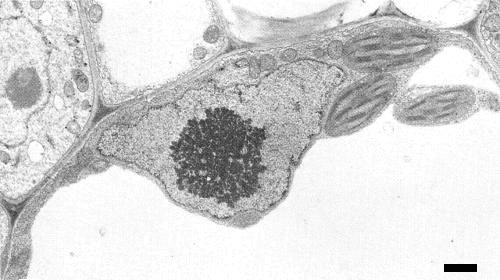
Infection of Phaseolus vulgaris cv. Top Crop is confined to phloem and adjacent parenchyma cells. Virus-like particles occur in hexagonally-packed crystalline array or in loose aggregates in the nuclei of infected cells (Fig. 8, Fig. 9). Distinctive alterations occur in nucleoli, including segregation of granular and fibrillar components and formation of electron-dense rings. Electron-dense areas shown by cytochemical tests with the electron microscope to contain deoxyribonucleoprotein may be regions where viral DNA synthesis occurs (Kim, Shock & Goodman, 1977). Virus-like particles have been seen in disintegrating nuclei of immature phloem and in mature phloem sieve elements.
Notes
Virus purification has been reported only for isolates from Puerto Rico, Colombia and El Salvador (Galvez & Castano, 1976; Goodman et al., 1977). Geminate particles are associated with a similar disease in Brazil but the virus has not been transmitted by inoculation of sap and the reported diameter of individual particles from electron microscopy observations is 12-13 nm (Matyis et al., 1976). Bean golden mosaic virus can be readily distinguished from other viruses infecting Phaseolus vulgaris by the distinctive symptoms and cytopathological alterations it causes in infected plants, by its narrow host range, and by its transmission by whiteflies.
Figures
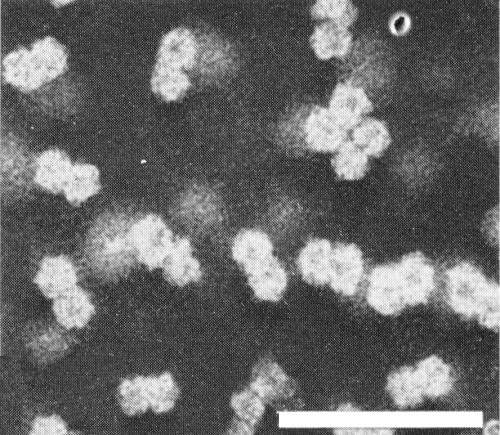
Electron micrograph of purified particles of bean golden mosaic virus. Preparation fixed with glutaraldehyde and stained with sodium phosphotungstate, pH 7.0. Bar represents 100 nm.
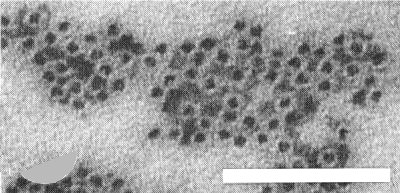
Electron micrograph of purified phytoferritin particles from infected plants. Preparation fixed with glutaraldehyde and stained with sodium phosphotungstate, pH 7.0. Bar represents 100 nm.
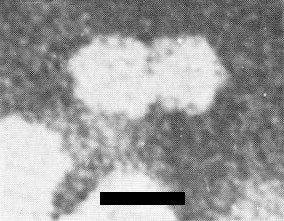
Electron micrograph of a bean golden mosaic virus ‘particle-pair’ at high magnification. Preparation fixed with glutaraldehyde and stained with sodium phosphotungstate, pH 7.0. Bar represents 20 nm.
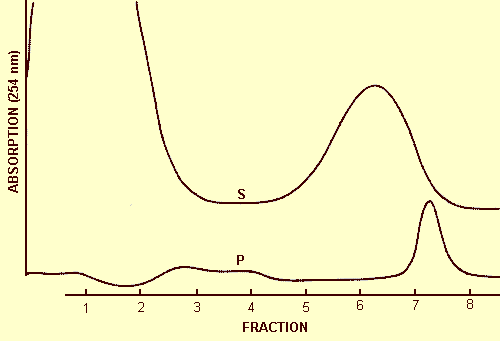
Sucrose density gradient profile of (S) supernatant and (P) pellet fractions following treatment of clarified extracts from infected plants with PEG. The broad peak in (S) is phytoferritin (see Fig. 4); the peak in (P) is bean golden mosaic virus. Sedimentation from the left.
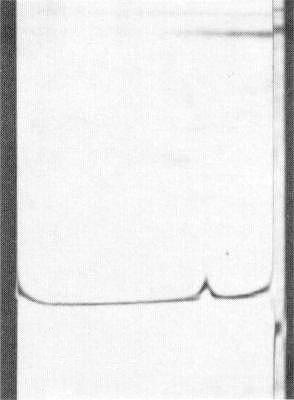
Schlieren diagram of a sedimentation velocity experiment after 8 min at 35,660 rev./min. Sedimentation from the right.
References list for DPV: Bean golden mosaic virus (192)
- Bird, Perez, Alconero, Vakili & Melendez, J. agric. Univ. Puerto Rico 56: 64, 1972.
- Bird, Sanchez & Vakili, Phytopathology 63:1435, 1973.
- Bird & Maramorosch, eds., Tropical Diseases of Legumes, Academic Press, New York, 1975.
- Costa, Pl. Prot. Bull. F.A.O. 13:123, 1965.
- Costa, A. Rev. Phytopath. 14: 429, 1976.
- Galvez & Castano, Proc. Am. Phytopath. Soc. 2: 75, 1975.
- Galvez & Castano, Turrialba 26: 205, 1976.
- Gamez, Turrialba, 21: 22, 1971.
- Goodman, Nature, Lond. 266: 54, 1977a.
- Goodman, Virology 83: 171, 1977b.
- Goodman, Bird & Thongmeearkom, Phytopathology 67: 37, 1977.
- Kim, Shock & Goodman, Proc. Am. Phytopath. Soc. 4:130, 1977.
- Matyis, Silva, Oliveira & Costa, IX Congresso Brasiliero Fitopatologia, Abstract 064, 1976.
- Meiners, Lawson, Smith & Diaz, Phytopathology 63: 803, 1973.