Details of DPV and References
DPV NO: 194 August 1978
Family: Secoviridae
Genus: Waikavirus
Species: Maize chlorotic dwarf virus | Acronym: MCDV
Maize chlorotic dwarf virus
R. E. Gingery USDA-SEA-AR, Ohio Agricultural Research and Development Center, Wooster, Ohio 44691, USA
O. E. Bradfute Dept. of Plant Pathology, Ohio Agricultural Research and Development Center, Wooster, Ohio 44691, USA
D. T. Gordon Dept. of Plant Pathology, Ohio Agricultural Research and Development Center, Wooster, Ohio 44691, USA
L. R. Nault Dept. of Entomology, Ohio Agricultural Research and Development Center, Wooster, Ohio 44691, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Rosenkranz (1969) and
Bradfute et al. (1972).
Synonym
- Ohio corn stunt agent (GSA-OH) (Rosenkranz, 1969)
-
An RNA-containing virus with isometric particles about 30 nm in diameter. It has a narrow host range within the Gramineae, and is transmitted in a semi-persistent manner by leafhoppers, but not by inoculation with sap.
Main Diseases
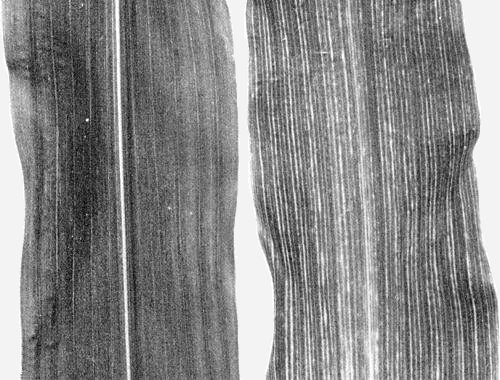
Causes plant stunting and chlorotic striping of tertiary leaf veins in maize (Zea mays) (Fig. 1, Fig. 2).
Geographical Distribution
Occurs in the southern USA. The distribution is limited to the overlapping areas of abundant Johnsongrass (the overwintering host) and the leafhopper vector, Graminella nigrifrons (Gordon & Nault, 1977).
Host Range and Symptomatology
The virus is not manually transmissible. In tests using leafhopper transmissions, only members of the Gramineae have been reported to be susceptible. Cultivated susceptible species include sorghum (Sorghum bicolor, S. vulgare var. sudanense), millet (Panicum miliaceum, Pennisetum glaucum) and wheat (Triticum aestivum). Symptoms on these hosts are very mild. Important weed hosts are Johnsongrass (Sorghum halepense), crabgrass (Digitaria sanguinalis) and the foxtails (Setaria spp.) (Nault et al., 1976).
-
Diagnostic species
- Zea mays
(maize). Chlorosis in the leaf whorl is evident from 5 to 10 days after inoculation. Later symptoms are chlorotic striping of tertiary veins and plant stunting. In some genetic lines there is reddening or yellowing and tearing of the leaves. Chlorotic striping of tertiary veins (Fig. 2) is diagnostic of maize chlorotic dwarf virus infection (Gordon & Nault, 1977).Propagation and assay species
- Zea mays
.
Strains
The type strain was collected in Ohio, USA (Nault et al., 1973). Several isolates from Johnsongrass differ from the type strain in the amount of stunting, discoloration and leaf tearing caused (Nault & Bradfute, 1979) (Fig. 3). An isolate causing more severe tertiary vein striping, but less stunting than the type strain, has been reported (Nault et al., 1976). No serological differences among strains and isolates have been found.
Transmission by Vectors
The virus is transmitted by the leafhopper, Graminella nigrifrons, in a semi-persistent manner with a retention period of several days. Transmission occurs after as little as 2 h acquisition access time and 2 h inoculation access time with no incubation period. Males and females, nymphs and adults can transmit. The virus is not passed transtadially or transovarially. Deltocephalus sonorus is a less efficient vector (Nault et al., 1973).
Transmission through Seed
None reported.
Serology
Antisera prepared in rabbits have titres greater than 1/100,000 in serological neutralization tests in which the amount of virus remaining was analysed by density gradient centrifugation (Gordon & Nault, 1977). Double diffusion tests in agar gels (D. T. Gordon & J. A. Foster, unpublished data), serum specific electron microscopic assays (O. E. Bradfute, unpublished data) and immunofluorescence tests (R. E. Gingery, unpublished data) have been done.
Relationships
There are no known relationships between maize chlorotic dwarf virus and other plant viruses. Although it resembles rice tungro virus in several properties including vector relationship, sedimentation rate and morphology (Galvez, 1968; Ling, 1966), the viruses are unrelated serologically (R. E. Gingery & D. T. Gordon, unpublished data).
Stability in Sap
No information.
Purification
Homogenize infected maize tissue in 0.1 M or 0.5 M potassium phosphate, pH 7, containing 0.5% 2-mercaptoethanol (2 ml buffer/g tissue). Squeeze the sap through cheesecloth and clarify by emulsifying with chloroform (1/3 to 1/2 volume). Recover the aqueous phase and proceed with either of the following methods:
1. Centrifuge the clarified extract at 100,000 g for 1 h. Resuspend the pellets overnight in 0.1 M or 0.5 M potassium phosphate, pH 7, and centrifuge in 10-40% sucrose gradients in 0.5 M potassium phosphate, pH 7. Recover the virus- containing zones and remove the sucrose by pelleting and resuspending the virus in 0.5 M potassium phosphate, pH 7 (D. T. Gordon, unpublished data; Louie, Knoke & Gordon, 1974).
2. Add polyethylene glycol (av. M. Wt 6000-7500) to 6% (w/v) and potassium chloride to 4% (w/v). Stir slowly for 1 h at room temperature. Centrifuge at 10,000 g for 20 min and resuspend the pellet in 1/50 to 1/30 volume (cf. original extract) of 0.5 M potassium phosphate, pH 7. Precipitate the virus by dialysis against distilled water and concentrate 2- to 3-fold by low-speed centrifugation and resuspension in 0.5 M potassium phosphate, pH 7.
The virus may be further purified by isopycnic centrifugation in caesium chloride at a density of 1.5 g/ml (Gingery, 1976; Gordon & Nault, 1977).
Properties of Particles
(Gingery, 1976;
D. T. Gordon, unpublished data).
Sedimentation coefficient (s20,w): 183 ± 6 S.
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 5.9.
Buoyant density in caesium chloride: 1.51 ± 0.01 g/ml in 0.5 M potassium
phosphate, pH 7.
A260/A280: 1.92 ± 0.04.
Molecular weight: 8.8 x 106.
Particle Structure
Particles are isometric with a diameter of about 30 nm in 2% potassium phosphotungstate (Fig. 4) (Bradfute et al., 1972). Empty protein shells may also be present (R. E. Gingery, unpublished data).
Particle Composition
Nucleic acid: About 36% of the particle weight is single-stranded RNA. The M. Wt is 3.2 ± 0.2 x 106 as determined by polyacrylamide gel electrophoresis and sedimentation rate. Molar percentages of nucleotides are G24, A30, C17, U29 (Gingery, 1976).
Protein: No information.
Relations with Cells and Tissues
Particles have been observed in the phloem, phloem parenchyma and bundle sheath, and less frequently in the mesophyll parenchyma. The particles are usually embedded in dense, granular cytoplasmic inclusions. Infected cells contain dense, curved, striated sheet inclusions randomly massed in the cytoplasm, and numerous cytoplasmic vesicles (Bradfute et al., 1972).
Notes
Prior to the discovery of maize chlorotic dwarf virus, most US maize plants with stunting and yellow or red leaf discoloration were assumed to have the corn stunt disease caused by a spiroplasma. However, the frequent association of maize chlorotic dwarf virus with such symptoms suggests that, in most cases, this disease is maize chlorotic dwarf and not corn stunt. The corn stunt spiroplasma has been identified in US maize in only a few samples from Louisiana and Texas (Bradfute & Robertson, 1974; Gordon & Nault, 1977). In contrast, the spiroplasma is frequently associated with similar symptoms in Mexico (Davis, 1973).
Only rice tungro and maize chlorotic dwarf viruses are known to have a semi-persistent relationship with their leafhopper vectors (Ling, 1966; Nault et al., 1973).
Figures
References list for DPV: Maize chlorotic dwarf virus (194)
- Bradfute, Gingery, Gordon & Nault, J. Cell Biol. 55: 25a, 1972.
- Bradfute & Robertson, Proc. Am. Phytopath. Soc. 1: 35, 1974.
- Davis, Pl. Dis. Reptr 57: 333, 1973.
- Galvez, Virology 35: 418, 1968.
- Gingery, Virology 73: 311, 1976.
- Gordon & Nault, Phytopathology 67: 27, 1977.
- Ling, Phytopathology 56: 1252, 1966.
- Louie, Knoke & Gordon, Phytopathology 64: 1455, 1974.
- Nault & Bradfute, In Leafhopper vectors and plant disease agents, ed. K. Maramorosch & K. F. Harris, NY: Academic Press, 1979.
- Nault, Styer, Knoke & Pitre, J. econ. Entomol. 66: 1271, 1973.
- Nault, Gordon, Robertson & Bradfute. Pl. Dis. Reptr 60: 374, 1976.
- Rosenkranz, Phytopathology 59: 1344, 1969.