Details of DPV and References
DPV NO: 220 September 1980
Family: Tymoviridae
Genus: Marafivirus
Species: Maize rayado fino virus | Acronym: MRFV
Maize rayado fino virus
R. Gámez Centro de Investigación en Biología Celular y Molecular, Universidad de Costa Rica, Ciudad Universitaria, Costa Rica
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Ancalmo & Davies (1961),
Gámez (1969) and
Gámez, Kozuka & Fukuoka (1977).
A virus with small RNA-containing isometric particles c. 31 nm in diameter which sediment as two components. Transmitted by leafhoppers in a persistent manner, but not by inoculation of sap. Host range apparently restricted to Gramineae. The virus is widely distributed in tropical areas of the American Continent.
Main Diseases
In maize (Zea mays), the virus causes a fine chlorotic stipple-striping of the veins, of variable severity (Fig. 1, Fig. 2, Fig. 3). Stunting and chlorosis are observed in the most susceptible varieties. The disease may cause significant reductions in yield, depending on environmental conditions, variety and time of infection (Gámez, 1969, 1980). In the coastal plains of Central America, the virus is frequently found in mixed infections with spiroplasmas and mycoplasmas associated with the maize (corn) stunt complex (Gámez, 1977, 1980).
Geographical Distribution
The virus is prevalent in Central America, Mexico and several countries in S. America including Peru and Uruguay (Gámez, 1977; Gámez, Kitajima & Lin, 1979), Brazil (Kitajima, Yano & Costa, 1976) and Colombia (Martínez-López, 1977). It was recently found in southern USA (Bradfute et al., 1980), and probably exists in many other countries in the Caribbean islands and S. America.
Host Range and Symptomatology
Maize rayado fino virus appears to be restricted to the Gramineae; it is found in maize, the only important natural host, and teosinte (Z. mays mexicana), which is limited to parts of Mexico and northern Guatemala. Several wild and cultivated grasses have been experimentally inoculated by means of leafhoppers but with negative results (Gámez, 1973, 1980). The virus has not been transmitted by manual inoculation of sap.
-
Diagnostic species
- Zea mays.
First disease symptoms appear in seedlings 8-14 days after inoculation, as distinct small chlorotic spots at the bases and along the veins of young leaves (Fig. 1). The typical stipple-striping symptom is evident in subsequently produced leaves, which show increasing numbers of spots, some of which fuse forming short stripes (Fig. 2). Numerous dots and stripes, and severe chlorosis are frequently observed in more susceptible cultivars (Fig. 3) (Gámez, 1980), as well as formation of holes in the leaf blade, wilting, and death of the plant (Martínez-López, 1977). Roots and ears are reduced in size, and the latter may bear few or no grains, depending on the sensitivity of the cultivar.Propagation species
- Several local maize cultivars (Tico H-1, Tico H-4) have been used for maintaining the virus and as a source of virus for purification.
-
Assay species
- The maize cultivars used for propagation are useful as source and test plants in experiments with insect vectors.
Strains
Maize rayado colombiano virus (Martínez-López, 1977) and Brazilian corn streak virus (Kitajima et al., 1976) share the majority of their properties with maize rayado fino virus (Kitajima & Gámez, 1977), but are serologically distinguishable from each other by spur formation in gel double diffusion tests (C. Rivera & R. Gámez, unpublished data).
Transmission by Vectors
The only known natural vector in the American tropics is the cicadellid leafhopper Dalbulus maidis (Gámez, 1977, 1980). The leafhoppers D. elimatus, Baldulus tripsaci and Graminella nigrifrons were shown to transmit the virus under experimental conditions (Nault & Bradfute, 1977; Wolanski & Maramorosch, 1979). Insects may also acquire the virus by intrahaemocoelic injection, or by feeding through membranes on crude or purified preparations. Virus-vector relations have been studied for the Costa Rican strain of the virus. In D. maidis the minimum acquisition and inoculation feeding periods are 6 h and 8 h respectively. Insects transmit the virus after long incubation periods of 8-37 days at 20 or 25°C, and retain infectivity for an additional 1-20 days. Transmission is intermittent and ability of vectors to transmit decreases with time after acquisition. However, when extracts from insects that have lost their ability to transmit are injected into healthy nymphs, the recipient insects become viruliferous. Females are more efficient vectors than males. Nymphs and young adults are equally capable of acquiring and inoculating the virus. Abdominal punctures cause no significant increase in the ability of nymphs to transmit. Insects retain the virus through the moult, but there is no transmission through the egg (Gámez, 1973, 1980; González & Gámez, 1974; Paniagua & Gámez, 1976). Maize rayado fino virus is transmitted in a manner typical of viruses that multiply in their vectors, but direct evidence for multiplication is lacking. Simultaneous transmission of the virus and the maize stunt spiroplasma has been demonstrated (Gámez, 1973; Wolanski & Maramorosch, 1979).
Transmission through Seed
Not seed-transmitted in maize.
Serology
The virus is moderately immunogenic. Precipitin ring tests and gel double diffusion tests are useful. Antibody titres of 1/64 to 1/256 are easily obtained by the latter test, in which a single line of precipitate is produced (Gámez, 1980; Gámez et al., 1979). Serologically specific electron microscopy has been used for identification of the virus (Bradfute et al., 1980). Titres of 1/1000 for antiserum and virus in maize sap were obtained in preliminary tests with this technique (C. Rivera & R. Gámez, unpublished data).
Relationships
Isolates of the virus collected in different countries in Central and S. America, Mexico (Gámez et al., 1979) and the USA (Bradfute et al., 1980) are serologically related. Antisera to 52 different small isometric viruses including members of the nepovirus, tymovirus, comovirus, tombusvirus, bromovirus and southern bean mosaic virus groups were tested against maize rayado fino virus with negative results (R. Koenig, personal communication). Similar results were obtained with other leafhopper-borne maize viruses that have small isometric particles, including maize stripe and maize line viruses (Martínez-López, 1977) and maize chlorotic dwarf virus (Gámez, 1980). Maize rayado fino virus and oat blue dwarf virus have similar vector relations and their particles share several characteristics but they are not serologically related (E. Banttari & R. Gámez, unpublished data).
Stability in Sap
Studied for the Colombian strain of the virus; the virus was assayed by allowing vectors to feed through membranes on crude plant extracts (Martínez-López & Rico de Cujía, 1977). Thermal inactivation point is between 60 and 65°C; longevity in vitro between 72 and 96 h at 20°C, or between 96 and 120 h at 3°C.
Purification
Extract infected maize leaves in 0.01 M phosphate buffer (pH 7.0) and clarify the extract by centrifuging at 10,000 g for 10 min. Precipitate the virus by adding polyethylene glycol, M. Wt 6000, to 8% and sodium chloride to 0.3 M and centrifuge at 10,000 g for 10 min; resuspend in phosphate buffer. Repeat this procedure three times. Centrifuge the preparation for 1 h at 87,100 g through 5% sucrose onto a 35% sucrose cushion. Collect the virus at the interphase, dilute five-fold, precipitate with polyethylene glycol, resuspend and centrifuge in 5-55% sucrose density gradients at 42,000 g for 16 h (Gámez et al., 1977; R. Gámez & T. Fukuoka, unpublished data). Purify further by isopycnic banding in caesium chloride.
Properties of Particles
The virus sediments as two components
(Fig. 4);
a top component of protein shells
(Fig. 5)
and a bottom component
(Fig. 6)
of infective nucleoprotein particles.
Sedimentation coefficients (s20,w) for the top and bottom components
(svedbergs) are 54 ± 4 and 120 ± 1 respectively.
Estimated M. Wt of the
nucleoprotein particles is 5.7 x 106.
A260/A280 for
protein shells and nucleoprotein particles is 0.87 and 1.58 respectively (uncorrected
for light-scattering).
Buoyant densities in caesium sulphate and caesium chloride, respectively, are 1.24 and 1.28 g/cm3 for the top component, and 1.37 and 1.46 g/cm3 for the bottom component (Gámez, 1980; P. León & R. Gámez, unpublished data).
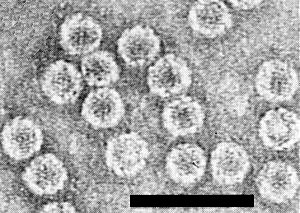
Particle Structure
Particles are isometric, 33 nm in diameter (top component) and 31.5 nm in diameter (bottom component) (Fig. 5, Fig. 6). Difference in diameter may be caused by flattening of the particles on the grid. The particles have icosahedral symmetry, with 32 morphological subunits and 180 structural subunits arranged in 20 hexamers and 12 pentamers (Gámez et al., 1977; T. Fukuoka & R. Gámez, unpublished data).
Particle Composition
Nucleic acid: single-stranded RNA, M. Wt 20 x 106, estimated to comprise 33% of particle weight (P. León & R. Gámez, unpublished data).
Protein: Electrophoresis in polyacrylamide gels containing 0.5% sodium dodecyl sulphate with or without 1% 2-mercaptoethanol revealed one main polypeptide with a M. Wt of approximately 20,500 daltons (P. León & R. Gámez, unpublished data).
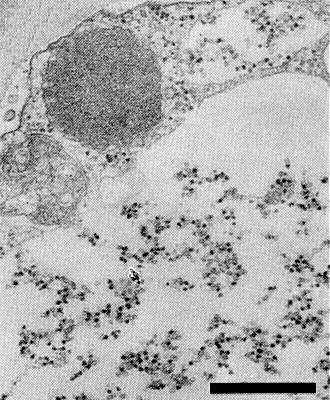
Relations with Cells and Tissues
Virus particles are commonly observed interspersed with amorphous materials in large vacuoles (Fig. 7) in the chlorenchyma and phloem parenchyma cells (Kitajima & Gámez, 1977). Particles may also occur dispersed in the cytoplasm, or in single rows in long tubules (Martínez-López, 1977), or in crystal-like arrays (Wolanski & Maramorosch, 1979).
Notes
Antigenic specificity, particle properties, identity of the leafhopper vector and virus-vector relations distinguish maize rayado fino virus from other viruses giving similar streak symptoms in maize, e.g. maize streak virus (Bock, 1974), maize stripe and maize line viruses (Kulkarni, 1973), and maize chlorotic dwarf virus (Gingery, Bradfute & Nault, 1978). The virus also differs from all other well-characterized plant viruses with small RNA-containing particles in the combination of properties displayed by its particles.
Maize rayado fino virus may be of significant economic importance. Reductions in yield of 40-50% of the weight of mature ears have been observed in Central American varieties, but losses of up to 100% occur in highly susceptible cultivars. No genetic immunity is known but tolerance has been observed (Gámez, 1977, 1980; Martínez-López, 1977).
Figures
References list for DPV: Maize rayado fino virus (220)
- Ancalmo & Davies, Pl. Dis. Reptr 45: 281, 1961.
- Bock, CMI/AAB Descriptions of Plant Viruses 133, 4 pp., 1974.
- Bradfute, Nault, Gordon, Robertson, Toler & Boothroyd, Pl. Dis. 64: 50, 1980.
- Gámez, Pl. Dis. Reptr 53: 929, 1969.
- Gámez, Ann. appl. Biol. 73: 285, 1973.
- Gámez, in Proc. int. Maize Virus Disease Colloquium and Workshop, p. 15, compiled by Ohio Agric. Res. Development Center, 1977.
- Gámez, Trop. Pest Management (formerly PANS) 26: 26, 1980.
- Gámez, Kozuka & Fukuoka, Revta Biol. trop. 25: 151, 1977.
- Gámez, Kitajima & Lin, Pl. Dis. Reptr 63: 830, 1979.
- Gingery, Bradfute & Nault, CMI/AAB Descriptions of Plant Viruses 194, 4 pp., 1978.
- González & Gámez, Turrialba 24: 51, 1974.
- Kitajima & Gámez, Turrialba 27: 71, 1977.
- Kitajima, Yano & Costa, Ciênc. Cult., S. Paulo 28: 427, 1976.
- Kulkarni, Ann. appl. Biol. 75: 205, 1973.
- Martínez-López, in Proc. int. Maize Virus Disease Colloquium and Workshop, p. 20, compiled by Ohio Agric. Res. Development Center, 1977.
- Martínez-López & Rico de Cujia, Fitopatol. Colomb. 6: 57, 1977.
- Nault & Bradfute, Proc. Am. Phytopath. Soc. 4: 172, 1977.
- Paniagua & Gámez, Turrialba 26: 39, 1976.
- Wolanski & Maramorosch, Fitopatol. Bras. 4: 47, 1979.