Details of DPV and References
DPV NO: 221 September 1980
Family: Geminiviridae
Genus: Mastrevirus
Species: Chloris striate mosaic virus | Acronym: CSMV
Chloris striate mosaic virus
R. I. B. Francki Department of Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia
T. Hatta Department of Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Grylls (1963). Virus first purified by Francki et al.
(1979).
- Synonym:
- Wheat striate mosaic virus (Australian) (Slykhuis, 1967)
- A virus with paired (‘geminate’) particles about 18 x 30 nm containing single-stranded DNA. The host range is apparently confined to species of Gramineae. Transmitted by the leafhopper, Nesoclutha pallida, probably in a persistent manner, but not by sap inoculation.
Main Diseases
Causes striate mosaic and dwarfing of some grasses and cereals including oats, barley and maize (Grylls, 1963; Greber, 1977).
Geographical Distribution
Reported only from Australia.
Host Range and Symptomatology
Known host range confined to the Gramineae including the grasses Chloris gayana, Ixopherus unisetus and Dactylis glomerata, and cereals such as Triticum spp. (wheat), Avena sativa (oats), Hordeum vulgare (barley) and Zea mays (maize) (Grylls, 1963; Greber, 1977). Similar symptoms (broken streaks or striate mosaic) occur on all susceptible species and varieties (Grylls, 1963).
- Diagnostic species
- Chloris gayana
(Rhodes grass). Yellow or greyish-white broken streaks and reduced plant vigour (Fig. 1, Fig. 2). - Hordeum vulgare (barley). Yellowish broken streaks and pronounced
dwarfing.
- Propagation species
- C. gayana
is convenient for maintaining cultures; it can be propagated vegetatively and the leaves used as a source of virus.- Assay species
- Assays must be done by recording the proportion of test plants that become infected following inoculation with leafhopper vectors. The insects can acquire virus from infected plants or by feeding through membranes on virus preparations; they can also be rendered viruliferous by microinjection (Grylls & Waterford, 1976; Francki et al., 1979).
Strains
No information.
Transmission by Vectors
Transmissible by the leafhopper Nesoclutha (obscura) pallida after acquisition feeds of 2 h or more (Grylls, 1963). Critical evidence for a latent period is lacking. Assay plants develop symptoms after 8 to 21 days (Francki et al., 1979).
Transmission through Seed
No information.
Serology
Antisera with homologous titres of up to 1/256 in Ouchterlony double diffusion tests have been produced in rabbits by injection of purified virus fixed with glutaraldehyde (Francki et al., 1979). Single, sharp precipitin lines are produced by purified virus or by virus concentrated from sap but not from crude sap (Fig. 3). Virus particles are readily detected in crude sap by immune electron microscopy (Derrick & Brlansky, 1976).
Relationships
Particle morphology, protein M. Wt and DNA M. Wt are similar to those of bean golden mosaic, maize streak and cassava latent viruses (Francki et al., 1980) all of which are included in the geminivirus group (Matthews, 1979; Francki et al., 1980). No antigenic relationship was detected to maize streak or cassava latent viruses (Francki et al., 1979).
Stability in Sap
No information.
Purification
Small amounts of virus can be obtained by the following method (Francki et al., 1979). Freeze C. gayana leaves at -80°C and pulverize each 100 g in 400 ml 0.1 M phosphate buffer, 10 mM EDTA and 0.5% thioglycollic acid, pH 7.4. Strain through cheesecloth and clarify by centrifugation at 27,000 g for 15 min. Retain supernatant fluid and add NaCl to 0.2 M and polyethylene glycol (M. Wt 6000) to 4% (w/v); stir for 2 h, centrifuge at 12,000 g for 10 min and discard supernatant fluid. Resuspend sediment in 10 mM phosphate buffer, 1 mM EDTA, pH 7.4, and add Triton X-100 to 1% (v/v); clarify by centrifugation at 27,000 g for 15 min and discard sediment. Layer supernatant fluid over 10% sucrose (in 10 mM phosphate buffer, 1 mM EDTA, pH 7.4), centrifuge at 300,000 g for 60 min and discard supernatant fluid. Resuspend sediment and repeat low- and high-speed centrifugation. Further purification may be achieved by centrifugation in 10-40% linear sucrose density gradients.
Properties of Particles
The paired particles form a single sedimenting component in sucrose density gradients (Francki et al., 1979).
M. Wt : c. 3.8 x 106 (Francki et al., 1980).
A260/A280: c. 1.3.
Buoyant density: 1.35 g/cm3 in CsCl after glutaraldehyde fixation
(Francki et al., 1980).
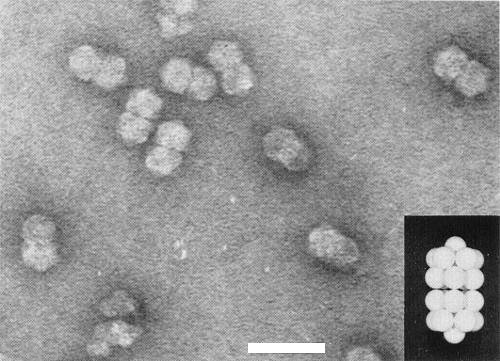
Particle Structure
Particles appear to be paired (‘geminate’), c. 18 x 30 nm. In fact each ‘pair’ is a single particle consisting of two incomplete icosahedra with a T = 1 surface lattice and a total of 22 capsomeres (Fig. 7; Hatta & Francki, 1979) and containing a single molecule of DNA (Francki et al., 1980). Particles disintegrate when mounted in phosphotungstate but remain intact in uranyl acetate.
Particle Composition
Nucleic acid: Circular single-stranded DNA, M. Wt c. 7.1 x 105 (Fig. 6) sedimenting at about 16 S in 0.15 M NaCl, 0.015 M Na citrate, pH 7, and accounting for about 19% of the weight of each particle ‘pair’ (Francki et al., 1980).
Protein: Subunits have M. Wt c. 2.8 x 104 (Francki et al., 1980).
Relations with Cells and Tissues
In C. gayana leaf cells, the virus particles are almost completely confined to the nuclei, forming aggregates (Fig. 4, Fig. 5) which may occupy the major part of the nucleus. Only very rarely can particles be detected in the cytoplasm or vacuoles (Francki et al., 1979). At least some of the particles in situ are paired (Hatta & Francki, 1979). Particles can be detected in all tissues of C. gayana leaves except the epidermis (Francki et al., 1979).
Notes
A number of viruses of cereals and grasses induce symptoms similar to those induced by chloris striate mosaic virus, and several of these are also transmitted by leafhoppers (Slykhuis, 1967). One of them, maize streak, is a geminivirus (Bock, 1974; Harrison et al., 1977) but is serologically unrelated to chloris striate mosaic virus (Francki et al., 1979).
Figures

Immunodiffusion test. Centre well: antiserum, diluted 1/128, to chloris striate mosaic virus. Outer wells: (a) sap from healthy leaf; (b) sap from virus-infected leaf; (c) as in (a) but concentrated by ultracentrifugation so that sediment from 1 g of leaf tissue was resuspended in 0.1 ml buffer; (d) as in (b) but concentrated as in (c).
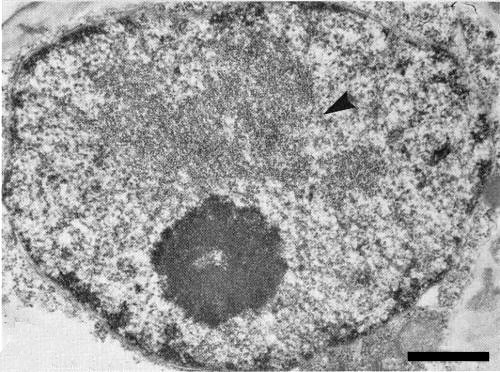
Section of nucleus from a leaf cell of infected C. guyana showing aggregate of virus particles (arrow). Bar represents 500 nm.
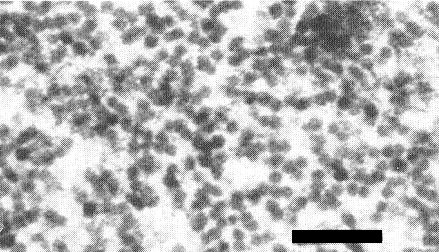
Enlarged view of an aggregate of virus particles in the nucleus of a leaf cell of infected C. gayana. Bar represents 100 nm.
References list for DPV: Chloris striate mosaic virus (221)
- Bock, CMI/AAB Descriptions of Plant Viruses 133, 4 pp., 1974.
- Derrick & Brlansky, Phytopathology 66: 815, 1976.
- Francki, Hatta, Grylls & Grivell, Ann. appl. Biol. 91: 51, 1979.
- Francki, Hatta, Boccardo & Randles, Virology 101: 233, 1980.
- Greber, Newsl. Aust. Pl. Path. Soc. 6: 4, 1977.
- Grylls, Aust. J. agric. Res. 14: 143, 1963.
- Grylls & Waterford, Newsl. Aust. Pl. Path. Soc. 5 (Suppl.): 89, 1976.
- Harrison, Barker, Bock, Guthrie, Meredith & Atkinson, Nature, Lond. 270: 760, 1977.
- Hatta & Francki, Virology 92: 428, 1979.
- Matthews, Intervirology 12: 132, 1979.
- Slykhuis, Rev. appl. Mycol. 46: 401, 1967.