Details of DPV and References
DPV NO: 24 October 1970
Family: Caulimoviridae
Genus: Caulimovirus
Species: Cauliflower mosaic virus | Acronym: CaMV
There is a more recent description of this virus: DPV 243
Cauliflower mosaic virus
R. J. Shepherd Department of Plant Pathology, University of California, Davis, California, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Tompkins (1937).
Selected synonyms
- Brassica virus 3 (Rev. appl. Mycol. 36: 303)
- Broccoli mosaic virus (Rev. appl. Mycol. 22: 122)
- Cabbage mosaic virus (Rev. appl. Mycol. 19: 65)
- Cabbage virus B (Rev. appl. Mycol. 24: 438)
- Broccoli mosaic virus (Rev. appl. Mycol. 22: 122)
A DNA-containing virus with isometric particles about 50 nm in diameter. It is transmitted by several aphid species in a non-persistent or semi-persistent manner and is readily transmissible by mechanical means. It is found only in cruciferous plants in which it is widely distributed in temperate regions.
Main Diseases
Induces mosaic and mottle diseases in a wide range of cruciferous crop and ornamental species, particularly the various cultivars of Brassica campestris and B. oleracea. Often found in mixed infection with turnip mosaic virus.
Geographical Distribution
World wide, especially in temperate regions.
Host Range and Symptomatology
With the exception of Nicotiana clevelandii (Hills & Campbell, 1968) only members of the Cruciferae have been reported as hosts. The virus is readily transmissible mechanically using an abrasive.
-
Diagnostic species
- Brassica oleracea
var. botrytis (cauliflower): Inoculated leaves usually symptomless; systemically infected leaves show initial vein-clearing, gradually replaced by green vein-banding (Fig. 1). Some varieties show only faint persistent vein-clearing, which may become masked in chronically infected plants, particularly at high temperatures. - B. campestris (Tendergreen mustard; also known as B. perviridis).
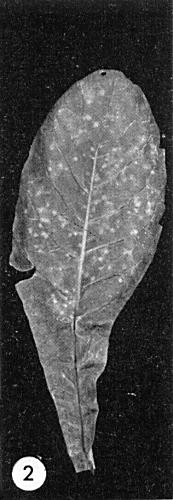
Some inoculated leaves develop chlorotic local lesions
(Fig. 2);
systemic symptoms
are vein-clearing, followed by chlorotic mottle
(Fig. 4).
- Matthiola incana var. annua (annual stock). Systemic chlorotic mottle.
-
Propagation and assay species
- Tendergreen mustard (B. campestris).
Strains
Minor variants can be distinguished by their virulence in cauliflower or turnip (Broadbent & Tinsley, 1953). Most isolates consist of a mixture of strains, of which some become systemic in Tendergreen mustard without inducing local lesions, and others induce local chlorotic lesions but do not become systemic.
Transmission by Vectors
At least 27 aphid spp. have been reported to transmit the virus (Kennedy, Day & Eastop, 1962) in a non-persistent or semi-persistent manner. All instars transmit and there is no latent period. Aphids can acquire the virus in 1-2 min and immediately thereafter can inoculate plants in less than 1 min. The virus is retained by Myzus persicae for more than 3 h, i.e., considerably longer than the typical non-persistent type viruses. Retention is unaffected by post-acquisition feeding activity (Hamlyn, 1955).
Transmission through Seed
None reported.
Serology
The virus is moderately immunogenic in rabbits. Purified virus gives a single band of precipitate in gel-diffusion tests in 1% agar but the virus is usually present in infected plants in insufficient amounts, or is liberated from inclusion bodies too slowly, to give reactions in agar gel-diffusion tests or tube precipitin tests with clarified leaf extracts (Pirone, Pound & Shepherd, 1961). In the presence of sufficient salt (0.2 M acetate) the virus adsorbs to polystyrene latex particles and these provide an extremely sensitive indicator system for serology.
Relationships
The virus is serologically closely related to dahlia mosaic virus (Brunt, 1966).
Stability in Sap
In cauliflower sap, the thermal inactivation point is 75-80°C (10 min), dilution end-point c. 10-3, and longevity at 20°C 5-7 days.
Purification
The concentration of virus in infected plants is usually less than 5 mg/kg of plant tissue. The following method is useful (Pirone, Pound & Shepherd, 1960). Homogenize tissue in 0.5 M phosphate (pH 7.5), add n-butanol to 8.5% (v/v) and centrifuge at 8000 g. Retain supernatant fluid and concentrate virus by 2-3 cycles of differential centrifugation using water as the solvent. Finally purify by sucrose density-gradient centrifugation. For large quantities of infected tissue (10-20 kg), precipitate the virus from butanol-clarified extracts by adding sodium chloride (to 0.05 M) and polyethylene glycol, 6000 M. Wt (100 g/l of extract); resuspend the precipitated virus in water and concentrate by 2 cycles of differential centrifugation (Shepherd, Bruening & Wakeman, 1970). The virus can be freed of host material by incubation with pancreatic ribonuclease (40 µg/ml) and centrifugation to equilibrium in CsCl gradients (Shepherd, Wakeman & Romanko, 1968).
Properties of Particles
Sedimentation coefficient (s20,w): 220 S (Pirone et al., 1961).
Buoyant density in CsCl: 1.37 g/ml.
Uncorrected absorbance at 260 nm (1 mg/ml, 1 cm light path): 7.
Specific refractive increment: 1.8 x 10-4/mg/ml.
A260/A280: 1.40-1.48.
Particle Structure
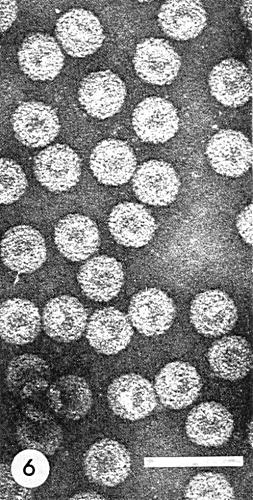
The particle is roughly spherical with a diameter of c. 50 nm. It flattens considerably when preparations are air-dried on electron microscope grids. This can be prevented by freeze-drying (Pirone et al., 1961). The particles are penetrated by potassium phosphotungstate and appear ‘empty’, with a diameter of 50 nm and a hollow centre of 20 nm diameter. In uranyl acetate the particles (Fig. 6) appear 45 nm in diameter and the stain does not penetrate (Hills & Campbell, 1968).
Particle Composition
DNA: Double stranded, about 16% of particle weight, molecular weight c. 4.5 x 106; buoyant density in CsCl: 1.702 g/ml. Melts at 87.2°C and has 43% G+C (Shepherd et al., 1970).
Protein: No information.
Relations with Cells and Tissues
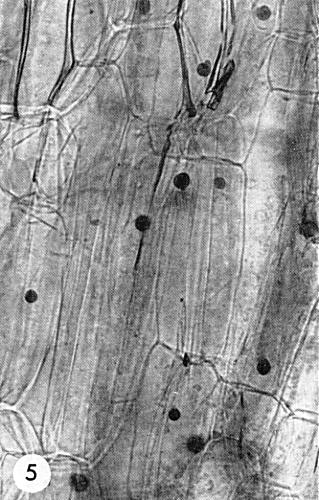
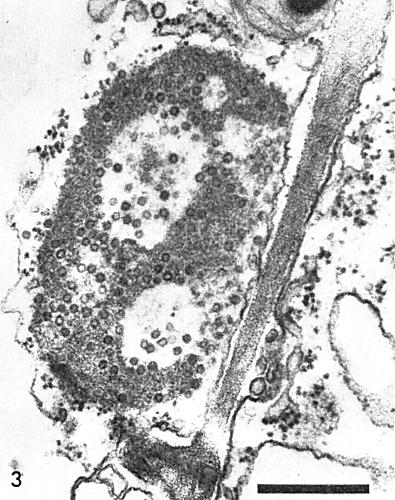
Virus particles are found only in the cytoplasm of infected cells, usually in association with a granular, electron-dense material that forms the bulk of the prominent inclusion bodies present in chronically infected plants. These inclusion bodies, when fully developed, consist of compact, elliptical masses of material with vacuoles, but without membranes. Virus particles are randomly scattered throughout the granular matrix and most are near the periphery and within the vacuoles (Fig. 3). In epidermal strips stained for 1 min with 0.5% phloxine, followed by thorough removal of excess stain with water, the deeply stained (reddish-pink) roughly spherical inclusion bodies are visible in the cytoplasm of virtually every cell (Fujisawa et al., 1967) (Fig. 5). This is useful in diagnosis of the virus.
Notes
Cauliflower mosaic virus may be differentiated from other non-persistent, aphid-borne viruses found in crucifers (e.g., turnip mosaic virus) by its relatively high thermal inactivation point (75-80°C), its longevity in vitro (5-7 days) and restricted host range (Walker, LeBeau & Pound, 1945). Unlike turnip mosaic virus, it does not infect solanaceous or chenopodiaceous plants, with the exception of Nicotiana clevelandii. The characteristic inclusion bodies it induces in chronically infected plants provide a convenient means of diagnosis.
Figures
Inclusion body in epidermal cell of B. campestris (Tendergreen mustard), showing virus particles embedded in the densely-staining granular matrix. Bar represents 500 nm. (Photo courtesy A. Allison).
References list for DPV: Cauliflower mosaic virus (24)
- Broadbent & Tinsley, Pl. Path. 2: 88, 1953.
- Brunt, Virology 28: 778, 1966.
- Kennedy, Day & Eastop, A conspectus of aphids as vectors of plant viruses, London, Commonwealth Institute of Entomology, 1962.
- Fujisawa, Rubio-Huertos, Matsui & Yamaguchi, Phytopathology 57: 1130, 1967.
- Hamlyn, Pl. Path. 4: 13, 1955.
- Hills & Campbell, J. Ultrastruct. Res. 24: 134, 1968.
- Pirone, Pound & Shepherd, Nature, Lond., 186: 656, 1960.
- Pirone, Pound & Shepherd, Phytopathology 51: 541, 1961.
- Shepherd, Wakeman & Romanko, Virology 36: 150, 1968.
- Shepherd, Bruening & Wakeman, Virology 41: 339, 1970.
- Tompkins, J. agric. Res. 55: 33, 1937.
- Walker, LeBeau & Pound, J. agric. Res. 70: 379, 1945.