Details of DPV and References
DPV NO: 248 July 1982
Family: Reoviridae
Genus: Oryzavirus
Species: Rice ragged stunt virus | Acronym: RRSV
Rice ragged stunt virus
R. G. Milne Istituto di Fitovirologia applicata del C.N.R., Via Vigliani 104, 10135 Torino, Italy
G. Boccardo Istituto di Fitovirologia applicata del C.N.R., Via Vigliani 104, 10135 Torino, Italy
K. C. Ling International Rice Research Institute, Los Baños, Laguna, Philippines
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
First described by
Hibino et al. (1977),
Ling (1977) and
Shikata et al. (1977).
Synonym
- Rice infectious gall virus (Bergonia, 1977; De la Cruz, 1977)
-
A virus with polyhedral particles c. 50 nm in diameter possessing spikes (and possibly a complete outer shell) out to a diameter of 65 nm and containing double-stranded RNA in 8 segments. Host range confined to Gramineae and the planthopper (Delphacidae) vector. Not mechanically transmissible to plants. Causes severe and economically important disease in rice.
Main Diseases
Infected rice plants (Oryza spp.) are stunted (Fig. 1), with whitish spindle-shaped enations, sometimes later turning brown, on the backs of the leaves and leaf sheaths (Fig. 2), and twisted and ragged leaves (Fig. 3). Infected young plants may be darker green than normal and the mature plants remain green after heading, when healthy plants have turned yellow. Excess branching may occur at the nodes. Delayed flowering, incomplete panicle emergence and unfilled grains lead to loss of yield (Hibino et al., 1977; Ling, 1977; Ling, Tiongco & Aguiero, 1978a; Ling et al., 1978c, 1978d; Cabauatan & Ling, 1978; Hibino, 1979; Shikata et al., 1979). The symptoms vary with the variety of rice, date of infection and stage of growth. Most rice lines, of both japonica and indica types, are susceptible though several lines show some field or glasshouse resistance (Palmer & Soepriaman, 1977; Heinrichs & Viajante, 1978; Ling, Tiongco & Salamat, 1978b; Ghosh & John, 1980). Yield losses are difficult to estimate, being hard to distinguish from those caused by the brown planthopper itself (see Transmission by Vectors) and from those of rice grassy stunt disease (Hibino, 1979). However, in field trials of several rice lines in Indonesia, the incidence of rice ragged stunt was 34-76%, and infected plants, harvested separately, gave grain yields only 17-47% that of healthy plants (Palmer, Soepriaman & Mochida, 1978). In India, losses may be 80-100% (Velusamy, Balasubramanian & Subba Rao, 1979).
Geographical Distribution
Reported from China, India, Indonesia, Japan, Malaysia, the Philippines, Sri Lanka, Taiwan and Thailand (Hibino et al., 1977; Ling, Tiongco & Aguiero, 1977; Palmer & Soepriaman, 1977; Chettanachit, Putta & Disthaporn, 1978; Habibuddin, 1978; Heinrichs & Khush, 1978; Ling et al., 1978a, 1978b; Palmer et al., 1978; Weerapat & Pongprasert, 1978; Chen, Chiu & Wang, 1979; Ghosh, John & Reddy, 1979; Hibino, 1979; Velusamy et al., 1979; Zhou & Ling, 1979; Shinkai, Nakano & Iwasaki, 1980; Xie & Lin, 1980).
Host Range and Symptomatology
Only Oryza latifolia, O. nivara and O. sativa are infected naturally (Ling et al., 1978a, 1978b; Hibino, 1979). Hibino (1979) experimentally infected maize, barley, rye and possibly oats, though wheat and sugarcane were resistant. Shikata et al. (1979) were unable to infect maize, barley, oats or wheat. Natural infection of these or other hosts has not been reported. The planthopper vector (see Transmission by Vectors) is a host but no disease is reported in the virus-carrying insect. The symptoms in experimental plant hosts essentially resemble those occurring in rice.
-
Diagnostic species
- Oryza sativa
(rice). The symptoms in naturally infected plants are described under Main Diseases. A variety in which experimentally induced symptoms have been described in detail is Taichung Native (TN-1) (Ling et al., 1978b; Hibino, 1979).Propagation and assay species
- There is no direct plant-to-plant assay or local lesion host. The virus can be grown in Oryza spp. inoculated with the virus-carrying vectors, and the infected plants can be maintained vegetatively if necessary (Boccardo & Milne, 1980a; Milne, 1980).
Strains
None reported.
Transmission by Vectors
The rice brown planthopper Nilaparvata lugens (Fig. 6) is the only known vector (Hibino et al., 1977; Ling et al., 1978c, 1978d; Ghosh et al., 1979; Shikata et al., 1979; Xie & Lin, 1980). The planthoppers Sogatella furcifera and Laodelphax striatellus, and the leafhoppers Nephotettix cincticeps, N. nigropticus, N. virescens and Recilia (= Inazuma) dorsalis are unable to transmit the virus (Chettanachit et al., 1978; Daquioag & Ling, 1978; Ghosh et al., 1979; Shikata et al., 1979; Xie & Lin, 1980). The minimum acquisition access period for the vector is 3 h, the latent period averages 9 days (range 2-33 days) and the minimum inoculation access period is 1 h. Symptoms in plants appear 10-36 days after inoculation. About 40% (range 6-76%) of hoppers transmit the virus. Nymphs appear to be more efficient transmitters than adults but males and females, and brachypterous and macropterous forms, appear equally efficient. Hoppers continue to transmit after moulting. Transmission by any one individual is usually intermittent. The hopper may remain infective for 1-4 weeks or until death (Hibino et al., 1977; Ling et al., 1977, 1978c, 1978d; Chettanachit et al., 1978; Hibino, 1979; Shikata et al., 1979; Ghosh & John, 1980). Four brown planthopper biotypes tested showed similar transmission characteristics (Ling et al., 1978c, 1978d). The virus is not transmitted through the egg (H. Hibino, unpublished data; Ling et al., 1978a, 1978c, 1978d). These properties and evidence from electron microscopic examination of thin sections (see Relations with Cells and Tissues) indicate that the virus multiplies in its vector.
Transmission through Seed
None found (Ling et al., 1978c, 1978d; Chen et al., 1979; Huang, Xiao & Zhan, 1980; Xie & Lin, 1980).
Serology
The virus particle is a good but complex immunogen. Senboku et al. (1979) described an antiserum with a precipitin tube-test titre of 1/512. Milne, Luisoni & Ling (1979) used an antiserum with gel diffusion titres of 1/64 against virus particles and 1/16 against host components but no titre against the virus RNA or poly[I]:poly[C] to search for possible relationships to other similar viruses (see Relationships). Hibino & Kimura (1982) used an antiserum with a precipitin tube-test titre of 1/1024 against virus particles to detect virus in plant and hopper extracts by enzyme-linked immunosorbent assay (ELISA). Luisoni, Milne & Roggero (1982) used an antiserum with a gel-diffusion titre of 1/512 against virus particles and no titre against host components, virus dsRNA or poly[I]:poly[C] to compare the sensitivities of ELISA and immunosorbent electron microscopy (ISEM) for detecting virus infection in rice plants.
Relationships
The virus has affinities with the fijiviruses and phytoreoviruses, having a partly (or completely) double-shelled isometric particle 50-65 nm in diameter containing dsRNA. In symptoms, host range and mode of transmission it resembles the fijiviruses (Matthews, 1979), but cannot be firmly placed in this group because it contains 8 (not 10) segments of dsRNA and may lack a complete outer shell (see Particle Composition and Particle Structure); neither can it be placed among the phytoreoviruses which have particles with a different morphology containing 12 segments of dsRNA (Matthews, 1979). Serologically, it is unrelated to rice black-streaked dwarf (Senboku et al., 1979), maize rough dwarf, pangola stunt, oat sterile dwarf or sugarcane Fiji disease viruses (Milne et al., 1979), all recognized members of the fijivirus group, or leafhopper A virus, a morphologically similar virus of uncertain classification (Boccardo et al., 1980). The possibility of a serological relationship with the phytoreoviruses has not been tested. The vector, Nilaparvata lugens, is not known to transmit any phytoreovirus or fijivirus, and vectors of these viruses, for example Laodelphax striatellus, Sogatella furcifera, Recilia (= Inazuma) dorsalis or Nephotettix spp., do not transmit rice ragged stunt virus.
Stability in Sap
Extracts of virus-infected leaves, when injected into vector hoppers, were infective at dilutions to 10-5 but lost infectivity after being heated to 60°C for 10 min. Infectivity was unaffected by freezing and thawing or by varying the pH of the extraction medium between 6 and 9 (Hibino, Saleh & Roechan, 1979).
Purification
The method recommended (E. Luisoni, G. Boccardo & R. G. Milne, unpublished data) is as follows. Crush each 1 g roots and stem bases with 1.5 ml chilled 0.4 M K phosphate buffer, pH 7.6, containing 0.02 M sodium sulphite, 0.01 M diethyldithiocarbamate and 0.005 M ethylenediamine-tetraacetate (sodium salt) (EDTA) (extraction solution). Filter the juice through nylon stocking, shake 30 min with an equal volume of Freon 113 (1,1,2-trifluoro-1,2,2-trichloroethane) and recover the virus-containing aqueous phase. Add Nonidet P-40 to 1.5% (v/v) final concentration and let stand at 0°C for 30 min. After 15 min centrifugation at 20,000 g, sediment the virus from the clarified supernatant fluid by ultracentrifugation at 303,000 g for 30 min. Cover the pellet with 10-20 ml extraction solution and allow to resuspend overnight at 4°C. Repeat the Freon and detergent clarification steps and centrifuge the virus at 22,000 rev/min for 150 min in a Beckman SW 25.2 rotor through a ‘cushion’ of 35% (w/v) sucrose dissolved in extraction solution. Resuspend the pellet in 0.1 M K phosphate buffer, pH 7.0, containing 0.005 M EDTA and centrifuge the virus into preformed linear 20-60% (w/v) Cs2SO4 density gradients prepared in the same buffer, in a Beckman SW 39L rotor at 36,000 rev/min for 60 min. The preparation forms two bands, the lower containing virus particles and the upper containing some particles trapped in host material.
Hibino & Kimura (1982) obtained infective virus by extracting infected tissues in the presence of 0.2 M glycine, pH 7.6, containing 5 mM EDTA and 0.5% (w/v) sodium ascorbate and clarifying the extracts with carbon tetrachloride and fluorocarbon; the virus was then pelleted from the aqueous phase by adding polyethylene glycol M. Wt 6000 to 7% (w/v) and NaCl to 0.2 M, and was further purified by sucrose density gradient centrifugation. Two other methods, yielding partially purified particles, have been reported (Milne et al., 1979; Shikata et al., 1979).
Properties of Particles
Particles in purified preparations (Fig. 9) resemble the B-spiked subviral particles of fijiviruses, are rather stable, and are infective; it is not clear whether parts corresponding to the outer shell of fijivirus particles are absent or extremely labile (see Particle Structure). Treatment with Na or K phosphotungstate, pH 7.0, causes loss of the B spikes. So does digestion with 100 µg/ml a-chymotrypsin in 0.1 M Tris-HCl buffer, pH 8.3, containing 0.2 M CsCl (Milne, 1980). Such treatment does not affect the B spikes of the subviral particles of maize rough dwarf virus (Milne, 1980), reovirus (Borsa et al., 1973) or leafhopper A virus (Boccardo et al., 1980). The particles of cytoplasmic polyhedrosis viruses also remain intact after such treatment (Matthews, 1979). Other particle properties not investigated.
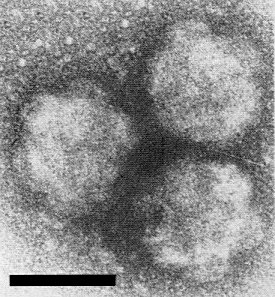
Particle Structure
In thin sections of virus-infected plants, Shikata et al. (1979) found isometric particles 50-70 nm in diameter. In sections of infected plants and insects, Hibino (1979) and Hibino et al., (1979) described particles having dense cores about 45 nm in diameter surrounded by outer shells to a diameter of 65 nm; and in sections of infected rice, Milne (1980) described dense cores 35-40 nm in diameter with less dense shells of outer diameter 50 nm supporting projections about 7 nm long, giving an outer particle diameter of about 63 nm. Such particles are similar to those described by Hibino et al. (1979) in sections of cells of infected plants and viruliferous insects. These structures resemble either the double-shelled particles of fijiviruses or the subviral particles with or without the B spikes. In negatively stained preparations of rice ragged stunt virus, ‘B-spiked subviral’ particles have been seen but not intact double-shelled particles, though a variety of fixatives, stains and techniques have been used (Shikata et al., 1979; Hibino, 1979; Hibino et al., 1979; Milne, 1980). Particles in uranyl acetate are about 50 nm in diameter with spikes 12-18 nm wide and 7-8 nm long (Fig. 9) (Hibino, 1979; Hibino et al., 1979; Milne, 1980). However, the spikes on the surface of rice ragged stunt virus particles appear different from the B spikes of the fijivirus subviral particles in being much broader at the base, giving the appearance of a truncated pyramid. Perhaps only damaged particles have been observed so far, as suggested by Hibino (1979) or, alternatively, a true outer shell may be lacking, as proposed by Milne (1980). A model representing a possible structure of the virus particle is shown in Fig. 8.
Particle Composition
Nucleic acid: RNA, double-stranded. The melting transition temperature in 0.15 M NaCl-0.015 M Na citrate, pH 7.0, is about 83°C and the hyperchromic shift is about 43%. The RNA, extracted directly from virus-induced enations as described by Luisoni et al. (1979), consists of 8 segments occurring roughly in equimolar amounts. Taking the maize rough dwarf virus genome segments as standards (Reddy et al., 1975), the RNA had a total M. Wt of about 11.6 million (Boccardo & Milne, 1980a). Reddy et al. (1975) assigned the same M. Wt to the 3rd and 4th segments of the maize rough dwarf virus genome, which exhibit different mobilities (Boccardo & Milne, 1980b). However, G. Boccardo & M. d'Aquilio (unpublished data), using Bombyx mori cytoplasmic polyhedrosis virus RNA (Fuji-Kawata, Miura & Fuke, 1970) as standards, obtained the following revised M. Wt (x 10-6) of rice ragged stunt virus RNA segments (Fig. 4): 2.39, 2.33, 1.72, 1.37, 1.23, 1.21, 0.55 and 0.52, giving a total M. Wt of about 11.3 million. This is only about three quarters that of any phytoreovirus or fijivirus.
Protein: no information.
Relations with Cells and Tissues
Virus particles occur in large numbers in the cytoplasm of phloem parenchyma cells and in sieve tubes, usually embedded in fibrillar ‘viroplasms’ (Fig. 5). The virus is not found elsewhere (Hibino, 1979; Hibino et al., 1979; Shikata et al., 1979; Milne, 1980). The phloem parenchyma cells multiply and hypertrophy, giving rise to enations on the leaves and leaf sheaths (Hibino et al., 1979; Hatta, Boccardo & Francki, 1982). A similar pathology is found in salivary gland cells of the infected vector, though without cell hypertrophy (Hibino, 1979; Hibino et al., 1979). Virus particle-containing tubules (Fig. 10), similar to those found in plant and insect tissues infected with fijiviruses or phytoreoviruses (Milne & Lovisolo, 1977) are found in infected planthopper tissues but have not been reported in infected rice cells.
Notes
Three similar viruses, all occurring in the same geographical areas, are known to infect rice: rice dwarf (Iida, Shinkai & Kimura, 1972), rice black-streaked dwarf (Shikata, 1974) and rice gall dwarf (Omura et al., 1980; Putta et al., 1980; Inoue & Omura, 1982). Rice dwarf and rice gall dwarf are phytoreoviruses transmitted by leafhoppers (Nephotettix spp.) but not by planthoppers; rice black-streaked dwarf, a fijivirus, is transmitted by Laodelphax striatellus and Unkanodes spp., two planthoppers that do not transmit rice ragged stunt virus. Only rice dwarf is easily distinguishable from the rest by symptoms because it does not induce enations but causes mosaic on the leaves (Iida et al., 1972). Both rice black-streaked dwarf (Shikata, 1974) and rice gall dwarf (Omura et al., 1980; Putta et al., 1980) viruses resemble rice ragged stunt virus in inducing enations (galls) on the leaves of the infected plants. Although some symptom differences can be recognised, they are greatly affected by the age of the plants, date of infection and environmental conditions. Therefore, diagnosis on symptomatology alone may be difficult and misleading, especially in mixed infections, and sensitive and reliable screening techniques based on ELISA or ISEM (Hibino & Kimura, 1982; Luisoni et al., 1982) should be used where possible.
Figures

Polyacrylamide gel electrophoretic separation of the double-stranded RNA segments of maize rough dwarf virus (left), rice ragged stunt virus (centre) and Bombyx mori cytoplasmic polyhedrosis virus (from C. C. Payne) (right). (Extremities of the gels not shown.)
References list for DPV: Rice ragged stunt virus (248)
- Bergonia, Philipp. Phytopath. 13: 2, 1977.
- Boccardo & Milne, Intervirology 14: 57, 1980a.
- Boccardo & Milne, CMI/AAB Descr. Pl. Viruses 217, 6 pp., 1980b.
- Boccardo, Hatta, Francki & Grivell, Virology 100: 300, 1980.
- Borsa, Sargent, Copps, Long & Chapman, J. Virol. 11: 1017, 1973.
- Cabauatan & Ling, Int. Rice Res. Newsl. 3(2): 9, 1978.
- Chen, Chiu & Wang, Pl. Prot. Bull. F.A.O 21: 447, 1979.
- Chettanachit, Putta & Disthaporn, Int. Rice Res. Newsl. 3(4): 14, 1978.
- Daquioag & Ling, Philipp. Phytopath. 14: 2, 1978.
- De la Cruz, Greenfields 7: 17, 1977.
- Fuji-Kawata, Miura & Fuke, J. molec. Biol. 51: 247, 1970.
- Ghosh & John, Pl. Dis. 64: 1032, 1980.
- Ghosh, John & Reddy, Curr. Sci. 48: 493, 1979.
- Habibuddin, Res. Bull. Malaysian agric. Res. Dep. Inst. 6: 113, 1978.
- Hatta, Boccardo & Francki, Physiol. Pl. Path. 20: 43, 1982.
- Heinrichs & Khush, Int. Rice Res. Newsl. 3(2): 13, 1978.
- Heinrichs & Viajante, Int. Rice Res. Newsl. 3(4): 9, 1978.
- Hibino, Rev. Pl. Prot. Res. 12: 98, 1979.
- Hibino & Kimura, Phytopathology 72: 656, 1982.
- Hibino, Roechan, Sudarisman & Tantera, Contr. cent. Res. Inst. Agric., Bogor, Indonesia No. 35, 15 pp., 1977.
- Hibino, Saleh & Roechan, Ann. phytopath. Soc. Japan 45: 228, 1979.
- Huang, Xiao & Zhan, Fujian agric. tech. Bull. 2: 30, 1980.
- Iida, Shinkai & Kimura, CMI/AAB Descr. Pl. Viruses 102, 4 pp., 1972.
- Inoue & Omura, Pl. Dis. 66: 57, 1982.
- Ling, Int. Rice Res. Newsl. 2(5): 6, 1977.
- Ling, Tiongco & Aguiero, Int. Rice Res. Newsl. 3(6): 11, 1977.
- Ling, Tiongco & Aguiero, Pl. Dis. Reptr 62: 701, 1978a.
- Ling, Tiongco & Salamat, Int. Rice Res. Newsl. 3(3): 8, 1978b.
- Ling, Tiongco, Aguiero & Cabauatan, Int. Rice Res. Inst. Res. PaperSer. 16: 1, 1978c.
- Ling, Tiongco, Aguiero & Cabauatan, Philipp. Phytopath. 14: 1, 1978d.
- Luisoni, Boccardo, Milne & Conti, J. gen. Virol. 45: 651, 1979.
- Luisoni, Milne & Roggero, Pl. Dis. 66: 929, 1982.
- Matthews, Intervirology 12: 129, 1979.
- Milne, Intervirology 14: 331, 1980.
- Milne & Lovisolo, Adv. Virus Res. 21: 267, 1977.
- Milne, Luisoni & Ling, Pl. Dis. Reptr. 63: 445, 1979.
- Omura, Inoue, Morinaka, Saito, Chettanachit, Putta, Parajarearn & Disthaporn, Pl. Dis. 64: 795, 1980.
- Palmer & Soepriaman, Int. Rice Res. Newsl. 2(5): 5, 1977.
- Palmer, Soepriaman & Mochida, Int. Rice Res. Newsl. 3(3): 15, 1978.
- Putta, Chettanachit, Morinaka, Parejarearn & Disthaporn, Int. Rice Res. Newsl. 5(3): 10, 1980.
- Reddy, Boccardo, Outridge, Teakle & Black, Virology 63: 287, 1975.
- Senboku, Chou, Noda & Shikata, Ann. phytopath. Soc. Japan, 45: 564, 1979.
- Shikata, CMI/AAB Descr. Pl. Viruses 135, 4 pp., 1974.
- Shikata, Leelapanang, Tiongco & Ling, Int. Rice Res. Newsl. 2(5): 7, 1977.
- Shikata, Senboku, Kamjaipai, Chou, Tiongco & Ling, Ann. phytopath. Soc. Japan 45: 436, 1979.
- Shinkai, Nakano & Iwasaki, Ann. phytopath. Soc. Japan 46: 411, 1980.
- Velusamy, Balasubramanian & Subba Rao, Int. Rice Res. Newsl. 4(1): 4, 1979.
- Weerapat & Pongprasert, Int. Rice Res. Newsl. 3(1): 11, 1978.
- Zhou & Ling, Int. Rice Res. Newsl. 4(6): 10, 1979.
- Xie & Lin, J. Fujian agric. Coll. 1: 98, 1980.