Details of DPV and References
DPV NO: 250 July 1982
Family: Betaflexiviridae
Genus: Carlavirus
Species: Shallot latent virus | Acronym: SLV
Shallot latent virus
L. Bos Research Institute for Plant Protection, Wageningen, The Netherlands
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Bos, Huttinga & Maat (1978b).
- A virus with straight to slightly curved particles c. 650 nm long. Manually transmissible to a few Allium species and to some dicotyledonous test plant species. Transmissible by aphids in the non-persistent manner. Very widespread in shallots.
Main Diseases
Occurs, apparently symptomlessly, in shallot (Allium ascalonicum), but virus-free plants were not available for comparison of infected with healthy plants (Bos et al., 1978b). Causes mild chlorotic streaking in leek (A. porrum) when occurring alone, and severe chlorotic or white streaking and even plant death in some cultivars of leek when in complex with leek yellow stripe virus (Paludan, 1980). Also isolated from onion (A. cepa) (Bos et al., 1978b).
Geographical Distribution
Identified in the Netherlands (Bos et al., 1978b) and Denmark (Paludan, 1980), but similar viruses recorded from England (Hollings, 1968) and France (Cadilhac et al., 1976). Probably occurs world-wide.
Host Range and Symptomatology
Host range is very limited. Has been experimentally transmitted to onion, leek, A. fistulosum and A. jailae, where systemic infection was symptomless, and to Chenopodium album, C. amaranticolor and C. quinoa, which react with local lesions. A. neapolitanum, A. schoenoprasum, freesia, hyacinth, lily (Lilium formosanum), narcissus, some tulip cultivars, and 15 other plant species tested were not infected (Bos et al., 1978b).
- Diagnostic species
- Chenopodium amaranticolor
and C. quinoa. Small necrotic (older leaves) or chlorotic (younger leaves) local lesions develop from c. 6 to 10 days after inoculation. They soon become more prominent as green rings surrounding necrotic tissue when inoculated leaves turn yellow (Fig. 1).- Propagation species
- Allium porrum
is suitable for maintaining cultures and is a good source of virus for purification (Bos et al., 1978b). A. fistulosum is a good filter host to free the virus from contamination with leek yellow stripe virus (Paludan, 1980).- Assay species
- Chenopodium amaranticolor
and C. quinoa are good local lesion assay species.
Strains
None reported.
Transmission by Vectors
Transmissible in the non-persistent manner by Myzus ascalonicus and perhaps by Aphis fabae, but not by M. persicae (Bos et al., 1978b).
Transmission through Seed
Not studied.
Serology
An antiserum with a titre of 1/1024 in microprecipitin tests was obtained from a rabbit injected with purified virus preparations, but this still reacted at dilutions of 1/1, 1/4 and 1/16 with clarified extracts from virus-free plants of onion, Nerine bowdenii and Hippeastrum sp., respectively (Bos et al., 1978b).
Relationships
Properties in vitro, particle size and morphology, and biological properties all suggest that the virus should be paced in the carlavirus group (Bos et al., 1978b). The virus reacted weakly with one of two antisera to narcissus latent virus and with antiserum to a carlavirus from Lonicera sp. (later described as lonicera latent virus; Van der Meer, Maat & Vink, 1980), but not with antisera to the following viruses: chrysanthemum B, nerine latent, pea streak, poplar mosaic, potato M, potato S, red clover vein mosaic, lily symptomless or carnation latent. These antisera had homologous litres between 1/256 and 1/4000.
Stability in Sap
In crude sap of A. porrum, infectivity was lost after 8-11 days of storage at room temperature, or heating for 10 min at 80°C; the dilution end-point was between 10-4 and 10-5 (Bos et al., 1978b).
Purification
Mucilage of Allium spp. poses problems (Huttinga, 1975). The following method, including Sephadex chromatography, was successful for onion yellow dwarf and leek yellow stripe viruses (Huttinga, 1975; Bos et al., 1978a) and also proved useful for shallot latent virus (Bos et al., 1978b). Grind 100 g tissue in 500 ml 0.1 M Tris buffer (adjusted to pH 9 with thioglycollic acid) together with 20 ml chloroform, 20 ml carbon tetrachloride and 10 ml diethyl ether. Centrifuge for 10 min at 4000 g. Centrifuge the supernatant fluid for 1.5 h at 26,500 g and resuspend pellets in 50 ml 0.1 M Tris-HCl buffer, pH 9. After 2 h at 4°C centrifuge for 10 min at 8000 g, then pass the supernatant fluid through a Sephadex G-200 colunm equilibrated with the above buffer containing 4 x 10-4 M NaN3, using a peristaltic pump allowing a flow rate of 4.6 ml/h/cm2. Pool the virus-containing ultraviolet-absorbing fractions and concentrate by centrifuging for 1.75 h at 47,000 g. Resuspend the pellets, and centrifuge through a 10-40% sucrose density-gradient for 2 h at 25,000 rev/min. For antiserum production further purification by equilibrium centrifugation in CsCl is advisable.
Properties of Particles
Sedimentation coefficient in 0.1 M Tris-HCl at 20°C at infinite dilution: about 147 S.
Buoyant density in CsCl: 1.313 g/cm3 (Bos et al., 1978b).
Particle Structure
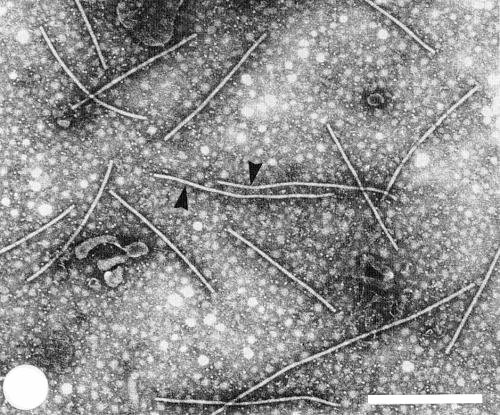
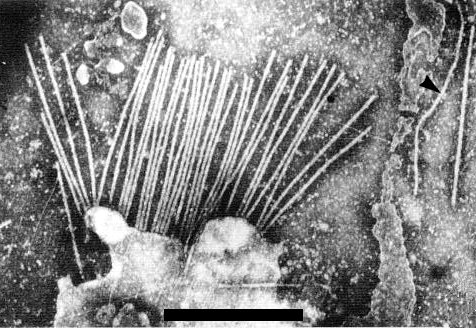
Virus particles are straight or slightly curved filaments (Fig. 2, Fig. 3), c. 650 nm long, which often aggregate end-to-end in crude sap preparations and may appear as dimers after purification (Bos et al., 1978b). In mixed crude preparations they can usually be easily distinguished from particles of two potyviruses, onion yellow dwarf and leek yellow stripe viruses, which are slightly longer and more flexuous (Fig. 2, Fig. 3).
Particle Composition
M. Wt of coat protein subunit is c. 23,000 (Bos et al., 1978b).
Relations with Cells and Tissues
In crude leaf extracts viewed with the electron microscope the particles are usually present in high concentrations. They often occur in bundles, or in groups with one end attached to membranes (Fig. 3) (Cadilhac et al., 1976; Bos et al., 1978b), as observed for pea streak virus (Bos, 1973). In ultrathin sections of leek, accumulations of particles in semi-crystalline array have been reported (Paludan, 1980).
Notes
The virus obviously belongs to the carlavirus group on grounds of its particle morphology and size, high concentration and behaviour in crude sap, serological relationships to some carlaviruses, limited host range and latency in important hosts. However, it did not react with antisera to the carlaviruses nerine latent virus or lily symptomless virus, and reacted only weakly with one out of two antisera to narcissus latent virus; also, unlike these viruses, it failed to infect monocotyledonous ornamental species.
Wetter & Milne (1981) have kept the virus outside the carlavirus group because
of its lower sedimentation coefficient, lower buoyant density and the lower M. Wt
of its coat protein subunit. However, correction of the sedimentation coefficient
for water would bring it close to values summarized for the group (156-172 S),
and its buoyant density differs only slightly from that of narcissus latent virus
(1.32 g/cm3 Brunt, 1977), erroneously listed as 1.33 by Wetter &
Milne (1981).
The virus appears to be symptomless in shallot, but whether this is entirely true
cannot be established until virus-free plants become available. It causes symptoms
in leek in Denmark, where it also intensifies the symptoms of leek yellow stripe
virus (Paludan, 1980). In shallot in France, a carlavirus (which may have been
shallot latent virus) was associated with mild symptoms but an unidentified
potyvirus occurred in plants with more severe symptoms (Cadilhac et al.,
1976). A combination of onion yellow dwarf virus and shallot latent virus must
have been responsible for shallot yellows ascribed by Hollings (1968) to a virus
with particles 620-640 nm long, because the diseased plants contained intracellular
inclusions readily detected by light microscopy, which is typical of onion yellow
dwarf virus.
Similar complexes occur in garlic in which mosaic symptoms have been ascribed
variously to a carlavirus (Cadilhac et al., 1976) serologically unrelated
to shallot latent virus (Delecolle & Lot, 1979), or to the potyviruses
causing onion yellow dwarf (Delecolle & Lot, 1979) and garlic yellow streak
(Mohamed & Young, 1981). Other unidentified potyviruses are reported from
garlic in France (Cadilhac et al., 1976), Venezuela (Lastra, Ladera &
Debrot, 1979) and Japan (Lee et al., 1979).
Figures

(a, b) Chenopodium amaranticolor and (c, d) C. quinoa with local lesions, 14 (c) and 18 days (a, b, d) after inoculation. (After Bos et al., 1978b).
References list for DPV: Shallot latent virus (250)
- Bos, CMI/AAB Descr. Pl. Viruses 112, 4 pp., 1973.
- Bos, Huijberts, Huttinga & Maat, Neth. J Pl. Path. 84: 185, 1978a.
- Bos, Huttinga & Maat, Neth. J. Pl. Path. 84: 227, 1978b.
- Brunt, Ann. appl. Biol. 87: 335, 1977.
- Cadilhac, Quiot, Marrou & Leroux, Annals Phytopath. 8: 65, 1976.
- Delecolle & Lot, Abstr. 3rd Conf ISHS Working Group on Vegetable Viruses, Bari, Italy, 1979: 83, 1979.
- Hollings, Ann. Rept. Glass. Crops Res. Inst., 1967: 108, 1968.
- Huttinga, Neth. J. Pl. Path. 81: 81, 1975.
- Lastra, Ladera & Debrot, Phytopathology 69: 1036, 1979.
- Lee, Yamazaki, Osaki & Inouye, Ann. phytopath. Soc. Japan 45: 727, 1979.
- Mohamed & Young, Ann. appl. Biol. 97: 65, 1981.
- Paludan, Tidsskr. PlAvl 84: 371, 1980.
- Van der Meer, Maat & Vink, Neth. J. Pl. Path. 86: 69, 1980.
- Wetter & Milne, in Handbook of Plant Virus Infections and Comparative Diagnosis, p. 695, ed. E. Kurstak, Amsterdam: Elsevier/North-Holland: 943 pp., 1981.