Details of DPV and References
DPV NO: 26 October 1970
Family: Rhabdoviridae
Genus: Cytorhabdovirus
Species: Lettuce necrotic yellows virus | Acronym: LNYV
There is a more recent description of this virus: DPV 343
Lettuce necrotic yellows virus
R. I. B. Francki Waite Agricultural Research Institute, Glen Osmond, South Australia
J. W. Randles Waite Agricultural Research Institute, Glen Osmond, South Australia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Stubbs & Grogan (1963).
An RNA-containing virus with enveloped, bacilliform particles about 227 x 66 nm. It has a narrow host range, is transmitted by inoculation of sap and by two species of aphid in the persistent manner. Reported only from Australia and New Zealand.
Main Diseases
Causes a serious disease of lettuce in Australia (Fig. 1) similar to that caused by tomato spotted wilt virus or virulent isolates of lettuce mosaic virus (Stubbs & Grogan, 1963). Also causes a mosaic in Calendula officinalis (Fig. 2) (Francki, unpublished).
Geographical Distribution
Widespread in Australia and has been identified in the South Island of New Zealand (R. C. Close, personal communication). Its distribution in South Australia appears to coincide with that of Sonchus oleraceus though Reichardia picroides and Embergeria megalocarpa have recently been shown to be hosts of virus and vector (Randles, unpublished), and are potentially important sources of the disease in the coastal areas of the state.
Host Range and Symptomatology
Narrow host range. Transmissible by sap inoculation with great difficulty to lettuce (Lactuca sativa) and sowthistle (Sonchus oleraceus), a weed which acts as a symptomless reservoir for the virus in the field.
-
Diagnostic species
- Nicotiana glutinosa.
Following mechanical inoculation, mild isolates induce slight stunting of the plant with downward curling of the leaves and a faint mosaic in the systemically infected leaves (Fig. 3). Virulent isolates produce necrotic local lesions 6-8 days after mechanical inoculation; systemic symptoms are vein-yellowing followed by chlorosis, curling and cessation of growth in terminal leaves (Fig. 4). -
Datura stramonium. Faint interveinal chlorosis in systemically infected leaves (Fig. 5).
Calendula officinalis. Chlorotic flecks on systemically infected leaves (Fig. 2).
Some isolates induce symptoms in Petunia hybrida, Spinacia oleracea, Lycopersicon esculentum, Gomphrena globosa (Stubbs & Grogan, 1963) and Nicotiana tabacum (Randles, unpublished).
-
Propagation species
- Nicotiana glutinosa
is suitable for maintaining cultures; both N. glutinosa and naturally infected lettuce are good sources of virus for purification.Assay species
- Nicotiana glutinosa
can be used as a local lesion host for some isolates (Fig. 4). Sonchus oleraceus is used for aphid transmission tests with Hyperomyzus lactucae; infection of S. oleraceus must be detected by return inoculation to N. glutinosa.
Strains
Variants isolated from the field have been distinguished by the severity of symptoms produced in Nicotiana glutinosa. The best known variant is the severe SE3 isolate (Fig. 4) (Stubbs & Grogan, 1963).
Transmission by Vectors
Transmissible by the aphids Hyperomyzus lactucae (Stubbs & Grogan, 1963) and H. carduellinus (Randles & Carver, unpublished). The virus systemically infects the tissues of H. lactucae (O’Loughlin & Chambers, 1967) but details of the acquisition, latent and inoculation periods are not known. Epidemiological studies indicate that H. lactucae is probably the sole important vector of LNYV in lettuce crops in South Australia (Randles & Crowley, 1970). Sonchus oleraceus appears to be the main field source of both LNYV and H. lactucae. Lettuce, which is not a host of the aphid, apparently becomes infected when probed by migrating aphids.
Transmission through Seed
No evidence of transmission through seed of lettuce varieties, Sonchus oleraceus or Nicotiana glutinosa.
Transmission by Dodder
Not tested.
Serology
An antiserum to the virus has been prepared in rabbits by intramuscular and intravenous injections (Harrison & Crowley, 1965). In gel-diffusion tests preparations of the virus give several specific bands, but concentrated virus must be used (McLean & Francki, unpublished). Judging from their positions, the bands of precipitate are formed by fragments of the virus rather than intact particles. Tube precipitin tests have been used but can be complicated by formation of non-specific precipitates.
Relationships
These have not been investigated. However, in particle structure, host range and mode of transmission, LNYV resembles the Gomphrena virus (Kitajima & Costa, 1966), sowthistle yellow vein virus (Richardson & Sylvester, 1968) and broccoli necrotic yellows virus (Hills & Campbell, 1968).
Stability in Sap
In Nicotiana glutinosa sap, the thermal inactivation point (10 min) is about 52°C, dilution end-point about 10-2, and infectivity is retained for 8 to 24 h. lnfectivity is stabilized by 0.1% bovine serum albumin but not by reducing agents (Crowley, 1967). Inactivation of virus is accelerated by some chelating agents such as sodium diethyl dithiocarbamate (Atchison, Francki & Crowley, 1969). The virus is rapidly inactivated by organic solvents such as n-butanol, chloroform and ether (Crowley, Harrison & Francki, 1965; Harrison & Crowley, 1965). Maximum infectivity is obtained with inocula at pH 6 (Francki, unpublished).
Purification
McLean & Francki (1967). Extract the virus from experimentally-infected N. glutinosa or field-infected lettuce by homogenising leaves in 0.2 M Na2HPO4, and clarify the extract by adding charcoal and DEAE-cellulose powder and filtering through Celite. Sediment the virus by centrifugation at 50,000 g for 15 min and resuspend in water. Purify further by column chromatography on calcium phosphate gel and/or centrifugation in sucrose density gradients.
Properties of Particles
Sedimentation coefficient (s20,w) approximately 940 S, buoyant density in sucrose solution in the range 1.19-1.20 g/cm3 (Harrison & Crowley, 1965).
Particle Structure
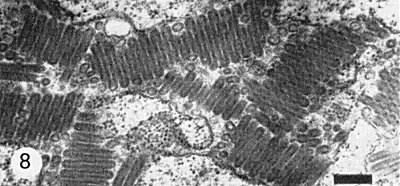
Particles are bacilliform, about 227 nm long and 66 nm wide in negatively stained preparations (Fig. 7) (Harrison & Crowley, 1965) but only about 50 nm wide in tissue sections (Fig. 8) (Chambers, Crowley & Francki, 1965). Bullet-shaped (Fig. 6) or globular particles are artefacts of preparations negatively stained and dried for electron microscopy (Wolanski, Francki & Chambers, 1967; Wolanski & Francki, 1969). The particle has an outer envelope covered with uniformly arranged projections enclosing an internal component consisting of a long filament, coiled in a regular shallow helix of pitch 4.5 nm (Wolanski et al., 1967).
Particle Composition
Purified virus preparations contain RNA and substantial amounts of lipid, but no detectable DNA (Hansen, Francki & Crowley, unpublished).
Relations with Cells and Tissues
Particles are seen in the cytoplasm of mesophyll, epidermal, hair cells, immature xylem and sieve tubes of Nicotiana glutinosa and Sonchus oleraceus, generally enclosed in membranes (Chambers, et al., 1965; Chambers & Francki, 1966; T. C. Lee, personal communication). Degeneration of nuclei, chloroplasts and mitochondria (Wolanski, 1969) and loss of 70 S chloroplast ribosomes (Randles & Coleman, 1970) occurs after symptoms appear.
Particles are seen in the muscles, fat body, brain, mycetome, tracheae, epidermis, salivary glands, and cells of the alimentary canal of Hyperomyzus Iactucae (O’Loughlin & Chambers, 1967). Particles with and without the outer coat are seen in H. lactucae.
Notes
Lettuce necrotic yellows virus can be distinguished from tomato spotted wilt and lettuce mosaic viruses, which also commonly occur in lettuce, by inoculation to Nicotiana glutinosa and Datura stramonium, serological testing, or examination of leaf-dip preparations by electron microscopy.
Figures
References list for DPV: Lettuce necrotic yellows virus (26)
- Atchison, Francki & Crowley, Virology 37: 396, 1969.
- Chambers, Crowley & Francki, Virology 27: 320, 1965.
- Chambers & Francki, Virology 29; 673, 1966.
- Crowley, Virology 31: 107, 1967.
- Crowley, Harrison & Francki, Virology 26: 290, 1965.
- Harrison & Crowley, Virology 26: 297, 1965.
- Hills & Campbell, J. Ultrastruct. Res. 24: 134, 1968.
- Kitajima & Costa, Virology 29: 523, 1966.
- McLean & Francki, Virology 31: 585, 1967.
- O’Loughlin & Chambers, Virology 33: 262, 1967.
- Randles & Coleman, Virology (in press), 1970.
- Randles & Crowley, Aust. J. agric. Res. 21: 447, 1970.
- Richardson & Sylvester, Virology 35: 347, 1968.
- Stubbs & Grogan, Aust. J. agric. Res. 14: 439, 1963.
- Wolanski, Ph.D. Thesis, Melbourne University, 1969.
- Wolanski & Francki, Virology 37: 437, 1969.
- Wolanski, Francki & Chambers, Virology 33: 287, 1967.