Details of DPV and References
DPV NO: 263 July 1983
Family: Betaflexiviridae
Genus: Carlavirus
Species: Elderberry symptomless virus | Acronym: ElSLV
Elderberry carlavirus
Jeanne Dijkstra Department of Virology, Agricultural University, Binnenhaven 11, 6709 PD Wageningen, The Netherlands
J. W. M. van Lent International Institute of Tropical Agriculture, Oyo Road, P.M.B. 5320, Ibadan, Nigeria
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Van Lent, Wit & Dijkstra (1980).
- Synonym
- Elderberry virus A (Jones, 1970; 1972).
- A virus with straight to slightly flexuous particles c. 12 x 678 nm, occurring in Sambucus spp. Transmitted in the non-persistent manner by Myzus persicae and by mechanical inoculation with sap.
Main Diseases
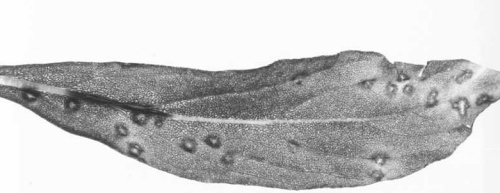
Occurs in species and cultivars of Sambucus (Caprifoliaceae) viz. S. nigra, S. racemosa and S. racemosa ‘Plumosa Aurea’. The virus probably causes no symptoms in Sambucus spp. because, although it was isolated from elderberry leaves with bright line-pattern symptoms it was also recovered from leaves without symptoms, and in experimental transmissions the virus infected S. pubens seedlings symptomlessly. However, in mixed infections with cherry leaf roll virus it enhanced the expression of line-pattern symptoms (Van Lent et al., 1980).
Geographical Distribution
Found in the Netherlands (Van Lent et al., 1980); a closely related virus, elderberry virus A, was found in Scotland in Sambucus canadensis cv. Adams imported from USA (Jones, 1970; 1972).
Host Range and Symptomatology
Species from four dicotyledonous families (Amaranthaceae, Caprifoliaceae, Chenopodiaceae and Leguminosae) were infected, mostly symptomlessly, by mechanical inoculation (Van Lent et al., 1980).
- Diagnostic species
- Chenopodium amaranticolor
(Fig. 1) and C. quinoa. In late summer, irregular, greyish-white necrotic local lesions with chlorotic haloes were formed 10-14 days after inoculation; throughout the rest of the year the inoculated leaves were infected symptomlessly and systemic infection occurred. - Cyamopsis tetragonoloba (guar). When inoculation is restricted to the
cotyledons, the virus infects them symptomlessly but fails to spread systemically;
when the first undivided leaf is inoculated too, it shows local chlorotic ringspots
(Fig. 2) and the virus spreads systemically, inducing a faint chlorotic mottle and
mosaic on the second leaf, and sometimes also on the third undivided leaf and on
the first trifoliolate leaf. The symptoms usually fade about 4 weeks after
inoculation.
- Gomphrena globosa. Necrotic local lesions with red or reddish-brown borders appeared c. 9 days after inoculation (Fig. 3).
- Gomphrena globosa. Necrotic local lesions with red or reddish-brown borders appeared c. 9 days after inoculation (Fig. 3).
- Propagation species
- The virus can be maintained in Gomphrena globosa and purified from
Sambucus nigra and S. racemosa.
- Assay species
- Gomphrena globosa is a good assay host.
Strains
No critical comparisons have been made between isolates from different regions in the Netherlands and from Britain (elderberry virus A).
Transmission by Vectors
The aphid Myzus persicae transmitted the virus with low frequency when allowed acquisition access periods of 10-45 min on cotyledons of Cyamopsis tetragonoloba and then transferred to cotyledons of C. tetragonoloba test plants. In similar tests, Aphis fabae did not transmit the virus from C. tetragonoloba or Sambucus racemosa to C. tetragonoloba (Van Lent et al., 1980).
Transmission through Seed
Not studied.
Serology
The virus is moderately immunogenic. Antiserum with a precipitin titre of 1/512 was prepared by injecting rabbits once intramuscularly and twice intravenously with purified virus (c. 10 mg total virus). The microprecipitin test and the enzyme-linked immunosorbent assay were useful (Van Lent et al., 1980).
Relationships
Properties of the virus in vitro and particle morphology suggest that it is a member of the carlavirus group. Serologically, the virus is related closely to elderberry virus A (Jones, 1970; 1972) and distantly to carnation latent virus (Van Lent et al., 1980).
Stability in Sap
In crude sap of Gomphrena globosa, infectivity was lost after 2-4 days at 20°C or 10 min at 70-75°C; the dilution end-point was between 10-3 and 10-4 (Van Lent et al., 1980).
Purification
Two methods were used by Van Lent et al. (1980).
1. The method described by Van Oosten (1972) for the purification of plum pox virus. Homogenize leaf material in 0.01 M citrate buffer (pH 6.7) containing 0.015 M sodium diethyldithiocarbamate (DIECA) and 0.0005 M sodium ethylenediamine-tetraacetate. Clarify by low speed centrifugation. Add Triton X-100 to 5% (v/v) and stir at room temperature for 30 min. Centrifuge at 78,000 g for 90 min. Resuspend the pellet in 0.01 M citrate buffer (pH 6.7) containing 1 M urea and 0.1% 2-mercaptoethanol. Centrifuge at low speed and recover the virus from the supernatant liquid by centrifugation through a 20% sucrose cushion. Purification is completed by ultracentrifugation in 10-40% sucrose gradients.
Van Lent et al. (1980) found that with elderberry carlavirus this method yielded rather pure preparations of infective virus (2-3 mg/100 g leaf material), but the particles showed much end-to-end aggregation. Better results were obtained with the following method.
2. The method described by Veerisetty & Brakke (1978) for the purification of alfalfa latent virus. Homogenize leaf material (stored at - 20°C) in 0.165 M disodium phosphate, 0.018 M trisodium citrate buffer (pH 9) containing 0.1% DIECA and 0.5% 2-mercaptoethanol. Strain the homogenized material through cheesecloth and centrifuge at low speed. Clarify the supernatant liquid by adding 1/20 volume 0.2 M Na2HPO4 and 1/100 volume 1.0 M CaCl2 with constant stirring for 20 min. Centrifuge at low speed. Precipitate the virus from the clarified supernatant fluid by adding 6% (w/v) solid polyethylene glycol, M. Wt 6000. Collect the precipitated virus by low speed centrifugation and resuspend it in the phosphate-citrate buffer (one-tenth of the original volume of extract) containing 1% Triton X-100. Centrifuge at low speed and recover the virus from the supernatant fluid by centrifugation twice through sucrose cushions, first of 20% sucrose, then of 30% sucrose. Resuspend the pellet in the extraction buffer (one tenth original molarity) without DIECA or 2-mercaptoethanol, and use this buffer for ultracentrifugation in 10-40% sucrose gradients.
The yield of elderberry carlavirus with this method was about the same as
with method no. 1 (2-3 mg virus per 100 g leaf material), but the particles
showed much less end-to-end aggregation (Van Lent et al., 1980).
Properties of Particles
In the analytical ultracentrifuge the virus sedimented as a single component (Van Lent et al., 1980).
Sedimentation coefficient (s20,w in phosphate-citrate buffer, pH 9.0: about 155 S.
Buoyant density in CsCl: 1.315 g/cm3.
A260/A280: 1.17 (not corrected for light-scattering).
Particle Structure
Straight to slightly flexuous filamentous particles c. 12 x 678 nm (Fig. 4).
Particle Composition
Nucleic acid: No information.
Protein: A single type of protein is found by electrophoresis in polyacrylamide/SDS gels with estimated M.Wt of 31,000 (Van Lent et al., 1980).
Relations with Cells and Tissues
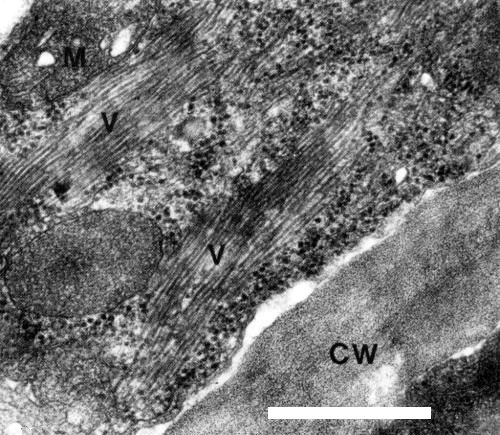
In ultrathin sections of leaf tissues of Sambucus racemosa ‘Plumosa Aurea’, large numbers of filamentous virus particles could be observed in the cytoplasm of most of the cells (Fig. 5).
Notes
Apart from elderberry carlavirus and its serological relative, elderberry virus A, morphologically similar viruses have been recorded from a cultivar of Sambucus canadensis in USA (Uyemoto & Gilmer, 1971; Uyemoto et al., 1971), from S. nigra in Czechoslovakia (Brcák, 1964; Brcák, & Polák, 1966) and Scotland (Jones, 1972), from a cultivar of S. racemosa in Scotland (Jones, 1972), and from elderberry (species not named) in S.E. England (Brunt & Stace-Smith, 1972). However, the relationship if any of these viruses to elderberry carlavirus is not known. Other viruses found in Sambucus species do not have filamentous particles.
Figures
References list for DPV: Elderberry carlavirus (263)
- Brcák, Proc. 5th Conf. Czechosl. Pl. Virologists, Prague 1962: 280, 1964.
- Brcák & Polák, Meded. Rijksfak. Landbwet. Gent 31: 397, 1966.
- Brunt & Stace-Smith, Rep. Glasshouse Crops Res. Inst., 1971: 121, 1972.
- Jones, Rep. Scott. hort. Res. Inst., 1969: 58, 1970.
- Jones, Ann. appl. Biol. 70: 49, 1972.
- Uyemoto & Gilmer, Phytopathology 61: 132, 1971.
- Uyemoto, Gilmer & Williams, Pl. Dis. Reptr 55: 913, 1971.
- Van Lent, Wit & Dijkstra, Neth. J. Pl. Path. 86: 117, 1980.
- Van Oosten, Neth. J. Pl. Path. 78: 33, 1972.
- Veerisetty & Brakke, Phytopathology 68: 59, 1978.