Details of DPV and References
DPV NO: 265 July 1983
Family: Betaflexiviridae
Genus: Carlavirus
Species: Helenium virus S | Acronym: HVS
Helenium virus S
Renate Koenig Institut für Viruskrankheiten der Pflanzen, Biologische Bundesanstalt für Land- und Forstwirtschaft, D 3300 Braunschweig, Germany
D.-E. Lesemann Institut für Viruskrankheiten der Pflanzen, Biologische Bundesanstalt für Land- und Forstwirtschaft, D 3300 Braunschweig, Germany
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Described by Kuschki et al. (1978) and Koenig et al. (1983).
- A virus with slightly flexuous or curved particles c. 670-700 nm long, occurring, often symptomlessly, in Helenium hybrids and Impatiens holstii. Transmissible by inoculation of sap and (some isolates) by aphids in the non-persistent manner. Occurrence probably world-wide.
Main Diseases
The effect of the virus on Helenium hybrids is not precisely known; in naturally infected plants showing spotting of leaves as well as colour breaking and distortion of flowers, the virus occurs in mixed infections with a potyvirus (Kuschki et al., 1978). In Impatiens holstii. growth reduction and leaf distortion were more pronounced with an isolate from Helenium than with one from Impatiens (Fig. 1; Koenig et al., 1983). Mild strains for each host are probably selected by horticultural practices.
Geographical Distribution
Identified in Germany in Helenium hybrids (Kuschki et al., 1978) and in the USA in Impatiens holstii (Koenig et al., 1983). Probably occurs world-wide.
Host Range and Symptomatology
The Helenium isolate has been transmitted only to a few Chenopodium spp. and to Impatiens holstii. The Impatiens isolate also infects Gomphrena globosa, Tetragonia expansa and a few Beta spp. (Kuschki et al., 1978; Koenig et al., 1983).
- Diagnostic species
- Chenopodium album, C. amaranticolor
and C. quinoa. Local chlorotic or necrotic lesions (Fig. 3) develop about 10 days after inoculation and are followed by systemic veinal chlorosis and mosaic (Fig. 4). In C. quinoa, intercostal areas later become yellow while bands along the veins remain green (Fig. 5).- Propagation species
- C. quinoa
is suitable for maintaining cultures and is a good source of virus for purification.- Assay species
- No local lesion host reliable for all isolates is known.
Strains
The Helenium and Impatiens isolates differ in their reported experimental host ranges, and in their transmissibility by aphids (Kuschki et al., 1978; Koenig et al., 1983).
Transmission by Vectors
The Helenium, but not the Impatiens isolate, is readily transmitted from Impatiens or Chenopodium quinoa to C. quinoa by Myzus persicae in the non-persistent manner (Koenig et al., 1983).
Transmission through Seed
Not found in C. quinoa (Koenig et al., 1983).
Serology
Antisera with titres of more than 1:2000 in the slide precipitin test are readily obtained. These antisera can also be used in the agar gel double diffusion test (Fig. 2) to detect the degradation products of the virus obtained after treating crude sap or purified preparations with 2.5% pyrrolidine (Kuschki et al., 1978; Koenig et al., 1983).
Relationships
The Helenium and Impatiens isolates are serologically closely related, if not identical. Similar homologous and heterologous titres are obtained in the slide precipitin test and no spurs are formed when the pyrrolidine degradation products of the two isolates are placed in adjacent wells in the agar gel double diffusion test (Fig. 2; Koenig et al., 1983). The virus is a member of the carlavirus group, being serologically related to carnation latent, chrysanthemum B, dandelion latent, hop mosaic, hop latent, lily symptomless and potato M viruses (Kuschki et al., 1978; Adams & Barbara, 1982a, 1982b; Johns, 1982; Koenig, unpublished data).
Stability in Sap
In crude sap of C. quinoa, the virus has a thermal inactivation point (10 min) of 60-70°C, dilution end-point of 10-4 - 10-5 and retains infectivity for 2-3 weeks.
Purification
Mince 100 g C. quinoa leaves in 100 ml of a solution containing 2.5% Na2B4O7. 10 H2O, 2.69% H3BO4, 0.2% ascorbic acid and 0.2% Na2SO3 at pH 7.8. Centrifuge expressed sap at low speed. To 1 vol. of the supernatant fluid add 0.15 vol. of 0.4% silver nitrate and leave at room temperature for 2-3 h. Centrifuge at low speed and stir 1 vol. of supernatant fluid with 0.25 vol. of chloroform for 10 min. Centrifuge at low speed and add 4% (w/v) of polyethylene glycol 6000 to the supernatant fluid. Leave at 4°C overnight. Centrifuge at low speed, resuspend the pellets in 10 ml of a solution containing 2.5% Na2B4O7. 10 H2O, 2.69% H3BO4, 3% urea and 0.1% (v/v) 2-mercaptoethanol at pH 7.8. Subject the virus to 1 or 2 cycles of differential centrifugation and purify further by sucrose density gradient centrifugation (Koenig et al., 1983).
Properties of Particles
Sedimentation coefficient (relative to marker viruses): 160 S (Kuschki et al., 1978).
A260/A280: 1.1 (Koenig et al., 1983).
Particle Structure
Slightly curved filamentous particles measuring 676 nm in length in phosphotungstate and 693 nm in uranyl acetate (Fig. 6); c. 13 nm wide (Koenig et al., 1983).
Particle Composition
Nucleic acid: No information.
Protein: M. Wt of the undegraded coat protein subunit is c. 31,000. Especially in the Impatiens isolate, the coat protein easily becomes partially degraded, yielding a residual protein of M. Wt c. 27,000 (Koenig et al., 1983).
Relations with Cells and Tissues
Infected leaf cells of Helenium, Impatiens and C. quinoa contain conspicuous cytoplasmic inclusions of irregular shape which are mainly composed of much-branched cisternae of the endoplasmic reticulum and scattered virus particles. Densely packed bundles of virus particles and large lipid globules occur in variable numbers in the inclusions (Fig. 7; Kuschki et al., 1978; Koenig et al., 1983).
Notes
In naturally infected Helenium hybrids the virus was always found together with a potyvirus, helenium virus Y, from which it can easily be differentiated by serology.
Acknowledgements
The work reported here was supported by the Deutsche Forschungsgemeinschaft.
Figures

Impatiens holstii 'Bellizzy Orange' infected with Helenium (left) and Impatiens (middle) isolates; healthy control (right). (From Koenig et al., 1983.).

Fusion of precipitin lines formed by the pyrrolidine degradation products of the Helenium (dH) and Impatiens (dI) isolates with antiserum to the Impatiens isolate (as I).
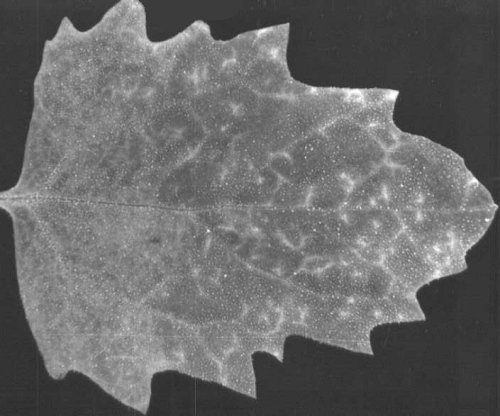
Chenopodium quinoa leaves infected with Impatiens isolate. Early systemic symptoms: veinal chlorosis and mosaic.

Chenopodium quinoa leaves infected with Impatiens isolate. Late systemic symptoms: yellowing of intercostal areas and green bands along the veins.
References list for DPV: Helenium virus S (265)
- Adams & Barbara, Ann. appl. Biol. 101: 483, 1982a.
- Adams & Barbara, Ann. appl. Biol. 101: 495, 1982b.
- Johns, Phytopathology 72: 1239, 1982.
- Koenig, Lesemann, Lockhart, Betzold & Weidemann, Phytopath. Z. 106: 133, 1983.
- Kuschki, Koenig, Düvel & Kühne, Phytopathology 68: 1407, 1978.


