Details of DPV and References
DPV NO: 266 July 1983
Family: Alphaflexiviridae
Genus: Potexvirus
Species: Plantain virus X | Acronym: PlVX
Plantain virus X
J. Hammond U.S. Department of Agriculture, Agricultural Research Service, Beltsville, Maryland 20705, USA
R. Hull John Innes Institute, Norwich NR4 7UH, UK
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
First reported by
Hammond (1977)
and described by
Hammond & Hull (1981).
A virus with filamentous particles c. 12 x 570-580 nm; found in Great Britain in Plantago lanceolata, often in mixed infections with other viruses. Readily sap-transmissible to a moderate range of hosts. No vector known.
Main Diseases
The virus occurs commonly in plantain (Plantago lanceolata) in Great Britain, often in mixed infections with other viruses (Hammond, 1981), but causes no obvious symptoms. Of no known economic importance.
Geographical Distribution
Reported only from Great Britain (Hammond & Hull, 1981; Hammond, 1981).
Host Range and Symptomatology
Plantain virus X infects at least 22 species from 6 dicotyledonous families (Hammond & Hull, 1981).
Diagnostic species
- Nicotiana x edwardsonii.
Necrotic flecks form in inoculated leaves after 10-15 days (Fig. 1). Systemically infected leaves produce similar symptoms. When plants are grown at high temperature (32°C) white ringspots are formed in inoculated and early systemically infected leaves. Later symptoms are similar to those formed at lower temperatures.- Nicotiana clevelandii. In plants grown in high light intensities,
systemic symptoms of sunken necrotic flecking
(Fig. 2)
appear 10-15 days after inoculation.
- Plantago rugelii. Spreading local and systemic necrotic spots form in 10-15 days.
Propagation species
- Nicotiana clevelandii.
Assay species
- Nicotiana x edwardsonii
is a reliable whole plant assay host. The symptoms in inoculated leaves are not suitable for local lesion assays.- Plantago rugelii is an alternative, but develops too few lesions to be useful for local lesion assays.
Strains
All isolates tested were serologically indistinguishable, and no host range differences were detected (J. Hammond, unpublished data).
Transmission by Vectors
Myzus persicae allowed acquisition access feeds of 15 min to 24 h did not transmit the virus from either Plantago lanceolata or Nicotiana benthamiana to N. benthamiana (J. Hammond, unpublished data).
Transmission through Seed
Not detected in N. benthamiana (J. Hammond, unpublished data).
Serology
Antisera with homologous titres of at least 1/1024 in the ring-precipitin test were prepared by subcutaneous injections of rabbits with a total of c. 2.4 mg purified virus emulsified with Freund’s adjuvant (Hammond & Hull, 1981). Agar gel double-diffusion tests, in which virus disrupted by sonication is used as antigen, are also useful (Fig. 4).
Relationships
The properties of the virus place it in the potexvirus group but no reactions were obtained in gel diffusion tests between plantain virus X antigen and antisera to cactus X, clover yellow mosaic, cymbidium mosaic, narcissus mosaic, papaya mosaic, potato X or white clover mosaic viruses (Fig. 4); nor between antiserum to plantain virus X and sonicated preparations of papaya mosaic virus and potato virus X, or the dissociated proteins of clover yellow mosaic, narcissus mosaic, nerine X or white clover mosaic viruses (Hammond & Hull, 1981). No serological relationship was found between plantain virus X and plantago severe mottle virus (Gracia, Koenig & Lesemann, 1983).
Stability in Sap
Infectivity was retained in Nicotiana clevelandii sap heated for 10 min at 60°C but not 70°C (Hammond & Hull, 1981).
Purification
Homogenize fresh or frozen leaves of Nicotiana clevelandii in 0.5 M phosphate buffer (pH 7.6) containing 10 mM EDTA. Add Triton X-100 to 2-2.5% (v/v) and mix for 1 h prior to differential centrifugation (10 min at 8000 g; 3 h at 85,600 g). Resuspend the pellets in 5 mM EDTA, pH 7.6, clarify by low speed centrifugation and concentrate the virus by centrifuging (3 h at 85,600 g) through a sucrose pad prior to further purification by sucrose gradient centrifugation (Hammond & Hull, 1981).
Properties of Particles
Sedimentation coefficient: the particles sediment as a single component with s°20,w = 119 S.
Particle weight: c. 40.5-41.1 x 106, based on particle length and structure, and the protein and nucleic acid M. Wts reported by Hammond & Hull (1981).
Absorption coefficient (A0.1%, 10mm) at 260 nm: c. 3.0.
A260/A280: c. 1.19 (partially corrected for light-scattering)
Buoyant density: 1.31 g/cm3 in CsCl, 1.259 g/cm3 in Cs2SO4.
Particle Structure
Particles are filamentous, c. 12 x 570-580 nm, with helical symmetry, having 8.8 subunits per turn, a pitch of 3.4 nm and true repeat of 17 nm. In uranyl acetate or uranyl formate many particles show distinct cross-banding (Fig. 6). In some isolates a loose external helical structure was observed in negatively stained epidermal dip preparations (Fig. 5), similar to those observed with cowpea mild mottle virus (Brunt & Kenten, 1973), white bryony mosaic virus (Tomlinson & Carter, 1971), and a second virus from white bryony (Milne et al., 1980).
Particle Composition
Nucleic acid: single-stranded RNA of c. 2.1 x 106 daltons, forming 5-6% of particle weight.
Protein: c. 94-95% of particle weight, c. 1475-1500 subunits of a single protein of c. 26,000 daltons (estimated by amino acid analysis and SDS/polyacrylamide gel electrophoresis). The amino acid composition is reported by Hammond & Hull (1981).
Relations with Cells and Tissues
Fibrous masses of particles in parallel arrays were observed in a ribosome-rich cytoplasmic matrix by electron microscopy of thin sections of Nicotiana benthamiana (Fig. 3). Particles were also observed scattered throughout the cytoplasm of N. benthamiana and N. × edwardsonii.
Notes
Another potexvirus, plantago severe mottle virus, has been reported from Plantago major in Canada (Rowhani & Peterson, 1980). This is not serologically related to plantain virus X (Gracia & Koenig, 1983) and is further distinguished by having shorter particles (c. 536 nm) and a distinct host range. Plantago severe mottle virus failed to infect P. lanceolata or any Nicotiana species tested (Rowhani & Peterson, 1980), whereas plantain virus X did not infect Gomphrena globosa (Hammond & Hull, 1981). Plantain virus X is readily distinguished from other viruses infecting Plantago by electron microscopy and host range (Hammond, 1981; 1982).
Acknowledgements
Figs 1, 3, 5 and 6 courtesy of Journal of General Virology.
Figures

Symptoms in inoculated leaves of Nicotiana × edwardsonii grown at (left) about 18°C and (right) about 32°C.
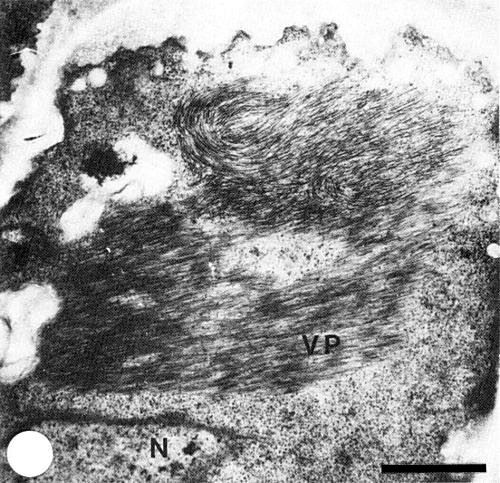
Thin section of infected Nicotiana benthamiana leaf cell showing arrays of virus particles (VP) in a ribosome-rich region; N=nucleus. Bar represents 1 µm.

Double diffusion agarose gel showing reaction of a sonicated preparation of plantain virus X (c. 1 mg/ml) in well V with antiserum of 1) plantain virus X; 2) potato virus XHR; 3) potato virus X; 4) clover yellow mosaic virus; 5) white clover mosaic virus; 6) papaya mosaic virus.
References list for DPV: Plantain virus X (266)
- Brunt & Kenten, Ann. appl. Biol. 74: 67, 1973.
- Gracia, Koenig & Lesemann Phytopathology 73: 1488, 1983.
- Hammond, Rep. John Innes Inst., 1976: 109, 1977.
- Hammond, Pl. Path. 30: 237, 1981.
- Hammond, Adv. Virus Res. 27: 103, 1982.
- Hammond & Hull, J. gen. Virol. 54: 75, 1981.
- Milne, Masenga & Lovisolo, Phytopath. Medit. 19: 115, 1980.
- Rowhani & Peterson, Can. J. Pl. Path. 2: 12, 1980.
- Tomlinson & Carter, Rep. natn. Veg. Res. Stn, 1970: 107, 1971.


