Details of DPV and References
DPV NO: 268 July 1983
Family: Rhabdoviridae
Genus: Unassigned Rhabdoviridae
Species: Beet leaf curl virus | Acronym: BLCV
Beet leaf curl virus
G. Proeseler Institut für Phytopathologie Aschersleben der Akademie der Landwirtschaftswissenschaften, Germany
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Disease first described by Wille (1928; 1929). Virus characterized by Eisbein & Proll (1978).
- Selected synonyms
- Beet Kräuselkrankheit virus (Rev. appl. Mycol. 7: 692)
- Beet leaf crinkle virus (Rev. appl. Mycol. 7: 692)
- Beta virus 3 (Rev. appl. Mycol. 36: 303)
- Rübenkräusel-Virus (Rev. Pl. Path. 56: 3275)
- Beet leaf crinkle virus (Rev. appl. Mycol. 7: 692)
A virus with bacilliform particles c. 225 x 80 nm. Transmitted in the persistent (propagative) manner by the bug Piesma quadratum (Heteroptera), but not by inoculation of sap. Host range moderate, mainly in the Chenopodiaceae. The virus affects sugar beet in Central Europe on sandy soils only, but is no longer of economic importance.
Main Diseases
Causes leaf curl disease and stunting in Beta vulgaris (sugar beet, fodder beet, etc.) and in Spinacia oleracea.
Geographical Distribution
Restricted to Germany, Poland and sporadic occurrences in Czechoslovakia and USSR. The apparent northern limit of distribution is the 18°C isotherm for the month of July. Found especially in crops on light sandy soils, because these are preferred by Piesma quadratum.
Host Range and Symptomatology
Confined to the Chenopodiaceae and Aizoaceae, the most important hosts being sugar beet and fodder beet. Also infects other members of the genus Beta, species of Atriplex, Chenopodium and Spinacia, and Tetragonia tetragonioides (syn. T. expansa) (Wille, 1929; Proeseler, 1966a; Schmutterer & Ehrhardt, 1966). Transmitted by Piesma quadratum but not by inoculation with sap.
-
Diagnostic species
- Beta vulgaris
(sugar beet). The first symptom is vein clearing in the youngest leaves (Fig. 1); soon afterwards the leaves begin to curve inwards, forming closed braids that resemble a head of lettuce (Fig. 2). Because new leaves are constantly being formed, the bushy plant may continue to grow upwards. The growth of the root ceases prematurely. - Chenopodium quinoa. Systemic vein clearing (Fig. 3) and leaf curling; severe stunting.
-
Propagation species
- Beta vulgaris
(sugar beet) is ideal for maintaining cultures.Assay species
- Sugar beet and fodder beet are the best plants for transmission tests with Piesma quadratum.
Strains
No strains reported.
Transmission by Vectors
Transmitted by the bug Piesma quadratum (Fig. 4) in the persistent (propagative) manner. The virus multiplies in its vector, and the bugs remain infective for life. There is no evidence of transmission to progeny insects. Minimum acquisition feeding period, 30 min; minimum inoculation feeding period, 40 min. There is a latent period of 7 to 35 days. Both the adult and nymphal stages can acquire and transmit the virus (Proeseler, 1966a; 1978a; 1978b).
Transmission through Seed
Not known to occur.
Transmission by Dodder
Five species of Cuscuta did not transmit (Proeseler, 1966a).
Serology
No antiserum has been prepared.
Relationships
Sugar beet savoy virus (Coons et al., 1958), which is transmitted by Piesma cinereum in North America, is poorly characterized and its relationship, if any, to beet leaf curl virus is unknown. Particle morphology places beet leaf curl virus in the plant rhabdovirus group, possibly in Peters’ (1981) sub group II because the particles accumulate in the perinuclear space.
Stability in Sap
Determined by injecting virus-free Piesma quadratum with beet sap. The thermal inactivation point (10 min) is between 54 and 58°C, the dilution end-point of unconcentrated sap is between 10-4 and 10-5 and infectivity in sap is retained for less than 24 h at 25°C or more than 28 days at - 20°C (Proeseler, 1966b).
Purification
Eisbein & Proll (1978) used the following procedure which is based on that used by Lee (1968) for purification of wheat striate mosaic virus. Extract 5-10 g fresh or frozen beet leaves or roots with 20 ml 0.05 M phosphate buffer, pH 7.0, containing 0.15 M D-mannitol, 0.001 M ethylenediamine-tetraacetate, 0.01 M MgCl2, and 0.1% bovine serum albumin. Concentrate and clarify the virus by two cycles of high and low speed centrifugation, resuspending the pellets obtained at high speed in the same buffer. Subject the final product to density gradient centrifugation.
Properties of Particles
Two narrow zones are produced by sedimentation in sucrose density gradients (Eisbein & Proll, 1978). The upper zone contains intact bacilliform particles (about 186 x 75 nm in negative stain) and numerous deformed virus particles and fragments; the lower zone contains aggregated particles. Attempts to purify the virus further led to disruption of the virus particles, and a decrease of infectivity (Eisbein & Proll, 1978).
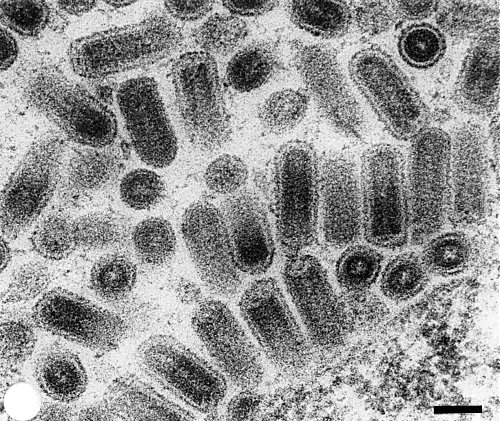
Particle Structure
In ultrathin sections the particles are bacilliform (Fig. 5), 80 nm in diameter, with an average length of 225 nm in diseased leaves, and 350 nm in roots. They have an electron-dense core with a central channel and an enveloping membrane with small protrusions. Many bullet-shaped particles are also observed (Eisbein, 1976).
Particle Composition
Unknown.
Relations with Cells and Tissues
Large concentrations of particles occur in the phloem parenchyma of leaves as well as in the parenchyma cells of roots. They accumulate predominantly in the perinuclear spaces and also in the cisternae of the endoplasmic reticulum. In the cytoplasm every group of virus particles is surrounded by a membrane (Eisbein, 1976).
Notes
Although at one time a serious problem, beet leaf curl virus is of little economic importance at present. The first symptoms it causes in sugar beet may be confused with beet mosaic, but the subsequent intense leaf curling symptoms, its transmission by Piesma quadratum, and its characteristic bacilliform particles distinguish this virus from all others infecting beet in Europe. However, P. quadratum also transmits a rickettsia-like organism, the cause of the beet latent rosette disease described from Germany (Proeseler, 1980).
Figures
References list for DPV: Beet leaf curl virus (268)
- Coons, Stewart, Bockstahler & Schneider, Pl. Dis. Reptr 42: 502, 1958.
- Eisbein, Arch. Phytopath. PflSchutz 12: 299, 1976.
- Eisbein & Proll, Arch. Phytopath. PflSchutz 14: 81, 1978.
- Lee, Virology 34: 583, 1968.
- Peters, CMI/AAB Descr. Pl. Viruses No. 244, 6 pp., 1981.
- Proeseler, Phytopath. Z. 56: 191, 1966a.
- Proeseler, Phytopath. Z. 56: 213, 1966b.
- Proeseler, Arch. Phytopath. PflSchutz 14: 95, 1978a.
- Proeseler, NachrBl. PflSchutz DDR 32: 254, 1978b.
- Proeseler, in Vectors of Plant Pathogens, p. 97, ed. K. F. Harris & K. Maramorosch, New York: Academic Press, 1980.
- Schmutterer & Ehrhardt, Z. PflKrankh., PflPath., PflSchutz 73: 271, 1966.
- Wille, Arb. Biol. Reichsanst. Land-u. Forstw. 16: 115, 1928.
- Wille, Monogr. zum PflSchutz No 2, 116 pp., Berlin: Springer, 1929.