Details of DPV and References
DPV NO: 269 July 1983
Family: Unallocated ssRNA- viruses
Genus: Tenuivirus
Species: Rice stripe virus | Acronym: RSV
There is a more recent description of this virus: DPV 375.
Rice stripe virus
S. Toriyama Laboratory of Plant Pathology, Faculty of Agriculture, University of Tokyo, Bunkyo-ku, Tokyo 113, Japan
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Disease described by
Kuribayashi (1931a). Virus characterized by
S. Toriyama (1982a,
1982b).
A virus with filamentous particles, of undetermined length but only c. 8 nm wide. The particles sediment as three components and contain four RNA species and a single type of protein. Non-structural protein material is produced abundantly in infected cells. Mechanical inoculation is difficult. The virus is transmitted by Laodelphax striatellus and three other planthopper species in a persistent manner; it is transmitted through the egg to about 90% of progeny insects. It infects many species of Gramineae but is not known to infect plants in other families. It occurs in rice-growing areas of Asia and the USSR, and causes significant reduction in rice yield.
Main Diseases
In rice the virus causes chlorotic stripes, chlorosis, moderate stunting and loss of vigour (Fig. 1); in severe infections the leaves develop brown to grey necrotic streaks and die. Diseased plants produce few or no panicles; those produced carry whitish to brown discoloured malformed spikelets (Fig. 3). Early infection of rice causes significant loss of yield (Yasuo, Ishii & Yamaguchi, 1965; Yasuo, 1969; Lee, 1969); late infection also reduces yield by retarding ear emergence and ripening (Yasuo et al., 1965). In maize and wheat the virus causes chlorotic stripes, chlorosis and stunting.
Geographical Distribution
The disease was first recognized in the early 1900s in central Japan, where severe damage was caused to rice crops. Since the 1950s the extension of early planting favoured the occurrence of the disease in Japan (Yasuo et al., 1965; Iida, 1969) and Korea (Lee, 1969). In Japan c. 200,000 ha of rice is affected each year. Damage in rice has also been reported in China (Chen, 1964), Taiwan (Lee, 1975) and the USSR (Reifman, Pinsker & Krylova, 1978).
Host Range and Symptomatology
The virus occurs naturally in rice, maize, wheat, oat, foxtail millet and wild grasses such as Cynodon dactylon, Digitaria adscendens, D. violascens, Eragrostis multicaulis and Setaria viridis. The virus is reported to infect 37 species of the family Gramineae but no species in other families. The main type of symptom induced is chlorotic striping (Kuribayashi, 1931a, 1931b; Amano, 1937; Shinkai, 1955, 1962; Yamada & Yamamoto, 1956; Sugiyama, 1966). The virus is readily transmitted to test plants by the planthopper, Laodelphax striatellus, but is difficult to transmit mechanically. Okuyama & Asuyama (1959) obtained infection in 6% of plants that were injected with crude extracts made from diseased plants with 0.01 M cysteine-HCl, and Sonku (1973) achieved infection in 0.9% of plants that were injected with extracts of viruliferous insects.
-
Diagnostic species
- Oryza sativa
(rice). Seedlings of most Japanese paddy varieties are highly susceptible to infection (See Notes). Chlorotic stripes, mostly with light yellowish broken streaks (Fig. 1), develop on systemically infected leaves 10 to 25 days after inoculation by planthoppers (L. striatellus). Characteristically, limp chlorotic leaves emerge without unfolding, elongate, droop and wilt (Fig. 2). Yellowing and moderate stunting also occur. - Triticum aestivum (wheat) and Zea mays (maize) (cv. Golden Cross Bantam). Symptoms in these species are similar to those in O. sativa. Wheat plants may produce whitish, rolled, fine leaves that quickly droop.
-
Propagation species
- Rice, wheat and maize are used to maintain cultures and as a source of virus for purification. Young rice seedlings are readily killed, and so care must be taken to use the appropriate growth stage and varieties, e.g. third leaf stage of rice cv. Norin No. 8 or Kinmaze. When maize seedlings are used, wheat, barley or rice plants must also be planted, or the planthoppers will die within 2-3 days.
-
Assay species
- Young rice and wheat seedlings are suitable for assaying transmission by insect vectors.
Strains
An isolate that causes mild symptoms on rice (Ishii & Ono, 1966) and two variants that differ in symptoms induced in wheat and in insect transmissibility (Kisimoto, 1965) are reported but there are no detailed studies of these isolates.
Transmission by Vectors
The most active vector in the field is the small brown planthopper, Laodelphax striatellus (Kuribayashi, 1931a, 1931b); three other planthopper species, Unkanodes sapporona (Shinkai, 1966), U. albifascia (Hirao, 1968) and Terthron albovittatum (Shinkai, 1970) also transmit. The proportion of active transmitters of L. striatellus is about 20% (Shinkai, 1962). Although the shortest acquisition feeding period is 15 min, best transmission is obtained with planthoppers that acquire the virus by feeding for 1 day. The incubation period of the virus in L. striatellus ranges from 5 to 21 days but incubation is complete within 5 to 10 days for most individuals. Minimum inoculation feeding time is 3 min: about half or more of the infective planthoppers infect rice seedlings after feeding for 1 h (Yamada & Yamamoto, 1955; Shinkai, 1962). Ability of the insects to transmit the virus decreases markedly with age. Females of L. striatellus are more efficient transmitters than males (Shinkai, 1962). The virus passes through a high percentage of eggs to progeny, about 90% in L. striatellus (Kuribayashi, 1931b; Yamada & Yamamoto, 1954, 1955; Shinkai, 1954, 1962). L. striatellus was selected and bred for high or low ability to acquire and transmit the virus (50-60% and less than 10% of the insects transmitting, respectively), and this was correlated with the frequency of transmission of the virus through the eggs (Kisimoto, 1967). Evidence of virus multiplication in L. striatellus has been obtained by repeated passage through the eggs for 6 years through 40 generations (Shinkai, 1962), by serial transfer of the virus from insect to insect by injection (Okuyama, Yora & Asuyama, 1968) and by detection of virus particle antigen in various organs of viruliferous insects with the fluorescent antibody staining technique (Kitani, Kiso & Yamamoto, 1968).
Transmission through Seed
None found in rice (Kuribayashi, 1931b; Okuyama, 1959).
Serology
The virus is a good immunogen. Rabbit antisera with titres of 1/512 to 1/1024 in precipitin tests are readily obtained. In agar gel diffusion tests, it is necessary to disrupt virus particles in purified preparations or crude sap of infected tissues by treatment with 0.5% SDS. The haemagglutination test was used to detect the virus particle antigen at high dilutions in vectors or plants (Saito & Iwata, 1964; Yasuo & Yanagita, 1963; Sonku & Sakurai, 1973b).
Relationships
The virus is serologically related to maize stripe virus (Gingery, Nault & Bradfute, 1981) but not to rice hoja blanca virus (Yasuo, Yanagita & Yamaguchi, 1961). The particle structure of these three viruses is similar (S. Toriyama, 1982b). In recent work (H. Hibino, personal communication) a filamentous virus purified from rice with grassy stunt disease was found to be distantly serologically related to rice stripe virus.
Stability in Sap
Assayed by observing transmission by injected insects. Dilution end-point: 10-4-10-5 in extracts from viruliferous insects and 10-3-10-4 in sap from diseased leaves. Thermal inactivation point (5 min): 50-55°C. Longevity: 4 days in extracts of viruliferous insects kept at 4°C, 8-12 months in viruliferous insects and diseased rice plants kept at -20°C, and 1-2 months in purified preparations kept at -20°C (Kiso, Yamamoto & Kitani, 1974).
Purification
(S. Toriyama, 1982a, 1982b). Grind infected leaves in 0.1 M Na2HPO4 containing 10 mM DIECA, and adjust to pH 7.2 with solid ascorbic acid. After clarification by treatment with 20% chloroform, collect the virus by centrifugation for 2 h at 123,000 g or by precipitation from 8% polyethylene glycol. Resuspend the virus in 0.01 M phosphate buffer, pH 7.5, and purify by repeated low and high speed centrifugation. Host plant impurities can be precipitated by adding solid ammonium sulphate to 30% saturation. The preparation can be further purified by centrifugation in 10-40% linear sucrose density gradients. The infective component (nB) aggregates readily and sediments to the bottom of sucrose gradient tubes; it may be necessary to resuspend the pellet and repeat the density gradient centrifugation. The yield of virus is 20-30 mg per 100 g fresh leaves. Addition of Mg2+, Ca2+, EDTA or bentonite to the buffer did not improve preservation of the virus particles (S. Toriyama, unpublished data).
Properties of Particles
The virus particles sediment as three main components: middle (M), bottom (B) and nB component (Fig. 5) (S. Toriyama, 1982a, 1982b). A ‘top component’ also occurs but consists of degraded particles (S. Toriyama, 1982a).
Sedimentation coefficients (s20,w) determined on a preparation with A260= 10: 72 S (M) (Koganezawa, 1977); 65 S (M), 80 S (B) and 98 S (nB) (S. Toriyama, unpublished data).
A260/A280: 1.49, determined on an unfractionated preparation (Koganezawa, Doi & Yora, 1975).
Absorbance at 260 nm (1 mg/ml, 1 cm light path): 4.4, determined on an unfractionated preparation; not corrected for light-scattering (S. Toriyama, unpublished data).
Buoyant density in caesium chloride: 1.282 g/cm3 (all components) (S. Toriyama, unpublished data)
Isoelectric point: particles precipitate at around pH 4.5 (S. Toriyama, unpublished data).
Particle Structure
The virus was formerly thought to have spherical particles (Koganezawa et al., 1975; Saito, 1977) but it is now known that the particles are filamentous, and only c. 8 nm wide (Fig. 8) (S. Toriyama, 1982b). In negatively stained dip preparations, the virus is mostly observed as apparently branched structures. The length of particles of M component (including length of branches) was 400 nm (Koganezawa et al., 1975). The branched appearance is caused by supercoiling: completely unfolded particles possess a helical structure 3 nm wide which in turn form secondary coils about 8 nm wide (S. Toriyama, 1982b) (Fig. 6, Fig. 7, Fig. 8).
Particle Composition
Nucleic acid: RNA, single-stranded. Four species, of M. Wt (x 106) 1.9, 1.4, 1.0 and 0.9 (S. Toriyama, 1982a, 1982b). The two smallest RNA species occur in M component (which is a mixture of two components), the 1.4 x 106 M. Wt RNA occurs in B component and the 1.9 x 106 M. Wt RNA in nB component (Fig. 4). Only the nB component particles are infective. The RNA constitutes about 12% of the particle weight, as judged by the A260/A280 value of purified preparations. The three RNA species from M and B components are linear molecules about 0.7-1.0 µm long (S. Toriyama, 1982a).
Protein: One species, of M. Wt 3.2 x 104 (Koganezawa, 1977; S. Toriyama, 1982b). Amino acid composition: asp 25 residues; thr 28; ser 21; glu 22; pro 5; gly 21; ala 23; cys ?; val 14; met 8; ileu 14; leu 24; tyr 9; phe 8; lys 27; his 5; arg 7; trp ? (S. Toriyama, unpublished data).
Other components: No significant amounts of lipid or polyamine are found in purified preparations (S. Toriyama, unpublished data).
Relations with Cells and Tissues
Large inclusions, shaped like rings, rods or figures of eight, are present in infected cells (Kawai, 1939; Hirai et al., 1964; Reifman et al., 1978). The inclusions usually contain many granules, but some have no granules and resemble crystalline inclusions. The inclusions probably consist of the non-structural protein which is serologically unrelated to the coat protein and is produced abundantly in infected cells of plants with severe symptoms but less so or not at all in infected cells of tolerant or resistant varieties. The isoelectric point is pH 5.4. Purified non-structural protein forms needle crystals at pH 4-5. Its sedimentation coefficient (s20,w) is 3 (Kiso & Yamamoto, 1973). One polypeptide species, of M. Wt 2 x 104 was reported by Koganezawa (1977). Amino acid composition: asp 19 residues; thr 11; ser 11; glu 18; pro 12; gly 8; ala 4; cys ?; val 5; met 4; ileu 6: leu 20; tyr 6; phe 8; lys 13; his 7; arg 4; trp ? (S. Toriyama, unpublished data; see also Kiso & Yamamoto, 1973).
In sections of infected plant cells (Koganezawa, 1977), recognition of individual filamentous virus particles is difficult but granular regions, sometimes enclosed by membranes, can be observed in the cytoplasm. These regions are possibly formed from virus aggregates. Virus particle antigen was detected in phloem tissue and mesophyll of infected wheat leaves by fluorescent antibody staining (Kiso et al., 1974). The virus moves downwards in the phloem at a rate of 25-30 cm/h at 30°C and multiplies when young tissues are reached (Sonku & Sakurai, 1973a, 1973b).
Notes
Rice stripe is readily distinguished from other viruses infecting rice, wheat and maize by its characteristic symptoms and its transmissibility by L. striatellus. The resistance of rice (Oryza sativa) to the virus has been extensively studied. Most Japanese paddy varieties are highly susceptible but Japanese upland varieties and indica-type rice varieties are resistant and/or tolerant. Inheritance of resistance appears to be governed by multiple alleles with various levels of resistance: gene St2 in Japanese upland varieties and gene St2i in indica-type varieties. A gene St1 at another locus as well as St2 is essential for resistance in Japanese upland varieties. Resistant Japanese paddy varieties (Chugoku No. 31 and Mineyutaka) have been bred (Yamaguchi, Yasuo & Ishii, 1965; Sakurai & Ezuka, 1964; Washio et al., 1967, 1968a, 1968b; Sakurai, 1969; K. Toriyama, 1969, 1972; K. Toriyama et al., 1966, 1972).
Figures

Chlorotic stripe symptoms on infected leaves of rice (Oryza sativa) compared with a healthy leaf (extreme left).

Malformed and immature ears, and chlorotic stripes on flag-leaves, of rice plants infected at a late growth stage.

Ribonucleic acid species on polyacrylamide gels: (left), three RNA segments from M + B components; (right), four RNA segments in a preparation from nB component, all except the uppermost being caused by contamination with M + B components.
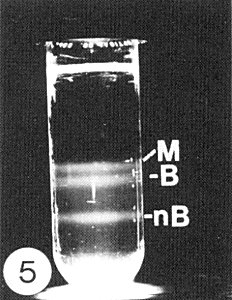
Bands formed by rice stripe virus after sedimentation in a sucrose gradient column: middle component (M), bottom component (B) and nB component (nB).
References list for DPV: Rice stripe virus (269)
- Amano, J. Pl. Prot., Tokyo 24: 774, 1937.
- Chen, Chekiang agric. Sci. 3: 123, 1964.
- Gingery, Nault & Bradfute, Virology 112: 99, 1981.
- Hirai, Suzuki, Kimura, Nakazawa & Kashiwagi, Phytopathology 54: 367, 1964.
- Hirao, Jap. J. appl. Ent. Zool. 12: 137, 1968.
- Iida, in The Virus Diseases of the Rice Plant, p. 3, Baltimore: Johns Hopkins, 1969.
- Ishii & Ono, Ann. phytopath. Soc. Japan 32: 83, 1966.
- Kawai, Ann. phytopath. Soc. Japan 9: 97, 1939.
- Kisimoto, Proc. Conf. on Relationships between Arthropods and Plant-Pathogenic Viruses (mimeo), Tokyo, p. 73, 1965.
- Kisimoto, Virology 32: 144, 1967.
- Kiso & Yamamoto, Rev. Pl. Prot. Res. 6: 75, 1973.
- Kiso, Yamamoto & Kitani, Bull. Shikoku agric. Exp. Stn 27: 1, 1974.
- Kitani, Kiso & Yamamoto, Bull. Shikoku agric. Exp. Stn 18: 117, 1968.
- Koganezawa, Symp. Virus Dis. trop. Crops, Trop. Agric. Res. Ser. No.10, p. 151, Trop. Agr. Res. Cent. Japan, 1977.
- Koganezawa, Doi & Yora, Ann. phytopath. Soc. Japan 41: 148, 1975.
- Kuribayashi, Bull. Nagano agric. Exp. Stn 2: 45, 1931a.
- Kuribayashi, J. Pl. Prot., Tokyo 18: 565, 636, 1931b.
- Lee, in The Virus Diseases of the Rice Plant, p. 67, Baltimore: Johns Hopkins, 1969.
- Lee, Taiwan Agric. 11: 95, 1975.
- Okuyama, Doct. Thesis, Univ. Tokyo, 155 pp., 1959.
- Okuyama & Asuyama, Ann. phytopath. Soc. Japan 24: 35, 1959.
- Okuyama, Yora & Asuyama, Ann. phytopath. Soc. Japan 34: 255, 1968.
- Reifman, Pinsker & Krylova, Arch. Phytopath. PflSchutz. 14: 273, 1978.
- Saito, Rev. Pl. Prot. Res. 10: 83, 1977.
- Saito & Iwata, Virology 22: 426, 1964.
- Sakurai, in The Virus Diseases of the Rice Plant, p. 275, Baltimore: Johns Hopkins, 1969.
- Sakurai & Ezuka, Bull. Chugokunatn. agric. exp. Stn A 10: 51, 1964.
- Shinkai, Ann. phytopath. Soc. Japan 18: 169, 1954.
- Shinkai, Ann. phytopath. Soc. Japan 20: 100, 1955.
- Shinkai, Bull. natn. Inst. agric. Sci. Tokyo C 14: 1, 1962.
- Shinkai, Ann. phytopath. Soc. Japan 32: 317, 1966.
- Shinkai, Ann. phytopath. Soc. Japan 36: 375, 1970.
- Sonku, Bull. Chugoku natn. agric. exp. Stn E 8: 1, 1973.
- Sonku & Sakurai, Ann. phytopath. Soc. Japan 39: 53, 1973a.
- Sonku & Sakurai, Ann. phytopath. Soc. Japan 39: 109, 1973b.
- Sugiyama, Ann. phytopath. Soc Japan 32: 83, 1966.
- K. Toriyama, in The Virus Diseases of the Rice Plant, p. 313, Baltimore: Johns Hopkins, 1969.
- K. Toriyama, Rev. Pl. Prot. Res. 5: 22, 1972.
- K. Toriyama, Sakurai, Washio & Ezuka, Bull. Chugoku natn. agric. exp. Stn A 13: 41, 1966.
- K. Toriyama, Washio, Sakurai, Ezuka, Shinoda, Sakamoto, Yamamoto, Morinaka & Sekizawa, Bull. Chugoku natn. agric. exp. Stn A 21: 1, 1972.
- S. Toriyama, Ann. phytopath. Soc. Japan 48: 482, 1982a.
- S. Toriyama, J. gen. Virol. 61: 187, 1982b.
- Washio, Ezuka, Sakurai & K. Toriyama, Jap. J. Breed. 17: 91, 1967.
- Washio, K. Toriyama, Ezuka & Sakurai, Jap. J. Breed. 18: 96, 1968a.
- Washio, K. Toriyama, Ezuka & Sakurai, Jap. J. Breed. 18: 167, 1968b.
- Yamada & Yamamoto, Ann. phytopath. Soc. Japan 18: 169, 1954.
- Yamada & Yamamoto, Spec. Bull. Okoyama pref. Agric. Exp. Stn 52: 93, 1955.
- Yamada & Yamamoto, Spec. Bull. Okayama pref Agric. Exp. Stn 55: 33, 1956.
- Yamaguchi, Yasuo & Ishii, J. cent. agric. Exp. Stn 8: 109, 1965.
- Yasuo, in The Virus Diseases of the Rice Plant, p. 167, Baltimore: Johns Hopkins, 1969.
- Yasuo & Yanagita, Ann. phytopath. Soc. Japan 28: 84, 1963.
- Yasuo, Yanagita & Yamaguchi, Ann. phytopath. Soc. Japan 26: 68, 1961.
- Yasuo, Ishii & Yamaguchi, J. cent. agric. Exp. Stn 8: 17, 1965.



