Details of DPV and References
DPV NO: 27 October 1970
Family: Alphaflexiviridae
Genus: Potexvirus
Species: Cymbidium mosaic virus | Acronym: CymMV
Cymbidium mosaic virus
R. I. B. Francki Department of Plant Pathology, Waite Agricultural Research Institute, University of Adelaide, South Australia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Jensen (1951).
Selected synonyms
- Cymbidium black streak virus (Rev. appl. Mycol. 30: 469)
- Orchid mosaic virus (Rev. appl. Mycol. 23: 261)
-
An RNA-containing virus with flexuous filamentous particles c. 475 x 13 nm. Readily transmissible by sap inoculation to a narrow range of host plants. Causes diseases in several genera of orchids. No known vector. World-wide distribution.
Main Diseases
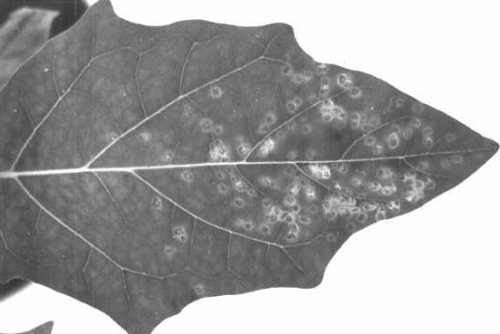
Causes mosaic (Fig. 1, Fig. 2) and necrosis in several genera of orchids (Jensen, 1951; Jensen & Gold, 1955; Murakishi, 1958a, 1958b; Kado & Jensen, 1964). Together with a strain of tobacco mosaic virus causes blossom brown necrotic streak in Cattleya (Thornberry & Phillippe, 1964).
Geographical Distribution
World-wide in cultivated orchids.
Host Range and Symptomatology
Infects species in the Orchidaceae and a few in other families. Transmissible by sap inoculation, for example, to the following:
-
Diagnostic species
- Chenopodium amaranticolor.
Slow-developing, large, blotchy lesions in inoculated leaves, not earlier than 20 days after inoculation (Fig. 3). - Cassia occidentalis, C. tora and C. bicapsularis. Small lesions in
inoculated leaves within 5 days after inoculation
(Fig. 4).
- Datura stramonium. Slow-developing, necrotic lesions in inoculated leaves, not earlier than 10 days after inoculation (Fig. 5).
-
Propagation species
- Cymbidium
plants are suitable for maintaining cultures and as a source of virus for purification (Corbett, 1960; Francki, 1966). Datura stramonium has also been used as a source of virus for purification (Francki, unpublished).Assay species
- Chenopodium amaranticolor, Datura stramonium
and Cassia occidentalis are all suitable local-lesion assay plants.
Strains
None reported.
Transmission by Vectors
No known vector.
Transmission through Seed
None reported.
Transmission by Dodder
No information.
Serology
The virus is strongly immunogenic. Serological tube-precipitin or gel-diffusion tests are convenient. A single precipitin band is produced in gel-diffusion tests.
Relationships
A member of the potato virus X group: a distant serological relationship to potato virus X has been reported and it has particles of similar length (Brandes & Bercks, 1965).
Stability in Sap
In Cymbidium sap, the thermal inactivation point (10 min) is 65-70°C and sap remains infective for at least 7 days at room temperature. Stable in the presence of organic solvents such as chloroform.
Purification
Easily purified by differential centrifugation. Yields of up to 360 mg virus may be obtained from 1 kg of Cymbidium leaves (Kado & Jensen, 1964; Francki, 1966).
Properties of Particles
Appear similar to those of potato virus X but details are unknown.
Particle Structure
Particles are flexuous filaments with helical symmetry about 475 nm long and 13 nm wide as observed in electron micrographs (Fig. 6).
Particle Composition
Contains about 6% RNA (Francki, unpublished).
Relations with Cells and Tissues
No information.
Notes
Tobacco mosaic virus is also common in orchids but can easily be distinguished from cymbidium mosaic virus by testing on herbaceous hosts, serological testing or examination of leaf-dip preparations by electron microscopy.
Figures
References list for DPV: Cymbidium mosaic virus (27)
- Brandes & Bercks, Adv. Virus Res. 11: 1, 1965.
- Corbett, Phytopathology 50: 346, 1960.
- Francki, Aust. J. biol. Sci. 19: 555, 1966.
- Jensen, Phytopathology 41: 401, 1951.
- Jensen & Gold, Phytopathology 45: 327, 1955.
- Kado & Jensen, Phytopathology 54: 974, 1964.
- Murakishi, Phytopathology 48: 132, 1958a.
- Murakishi, Phytopathology 48: 137, 1958b.
- Thornberry & Phillipe, Pl. Dis. Reptr 48: 936, 1964.