Details of DPV and References
DPV NO: 271 July 1983
Family: Secoviridae
Genus: Nepovirus
Species: Artichoke yellow ringspot virus | Acronym: AYRSV
Artichoke yellow ringspot virus
G. L. Rana Istituto di Patologia vegetale, Università di Bari, 70126 Bari, Italy
Panayota E. Kyriakopoulou Benaki Phytopathological Institute, Kiphissia, Athens, Greece
G. P. Martelli Istituto di Patologia vegetale, Università di Bari, 70126 Bari, Italy
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Disease described by Kyriakopoulou & Bem (1973) and Rana, Rosciglione & Cannizzaro (1978), virus characterized by Rana et al. (1980).
A virus with isometric particles c. 30 nm in diameter, containing two functional RNA species. Readily transmitted by inoculation of sap to many herbaceous hosts and frequently transmitted through seed and pollen. No vector known. Occurs in a range of cultivated and wild plants in Greece and Italy.
Main Diseases
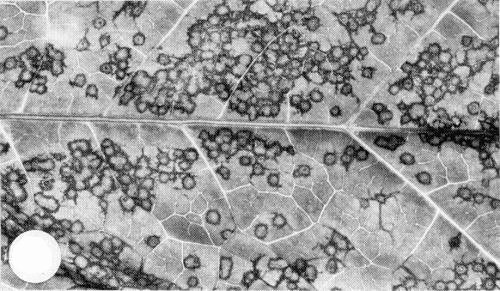
Causes chrome-yellow blotches, rings and lines (Fig. 1) on the leaves of artichoke (Cynara scolymus) and cardoon (Cynara cardunculus) (Kyriakopoulou & Bem, 1973; Rana et al., 1978); yellow/necrotic rings and oakleaf patterns in tobacco (Nicotiana tabacum) cv. Black of Argos (P. E. Kyriakopoulou, unpublished information); stunting, diffuse yellowing and leaf malformation in French bean (Phaseolus vulgaris) and broad bean (Vicia faba) (P. E. Kyriakopoulou, unpublished information). In naturally infected wild plant species, induces chlorotic to yellow rings, mottling and, occasionally, malformation of the leaves.
Geographical Distribution
Reported from north-east Peloponnesus (Greece) and Sicily (Italy).
Host Range and Symptomatology
In nature the virus was found infecting 32 different plant species in 13 dicotyledonous families, of which 5 species are cultivated (artichoke, cardoon, tobacco, French and broad beans) and 27 are wild (P. E. Kyriakopoulou, unpublished information). Experimental host range is wide, including 56 species in 11 families (Rana et al., 1980).
- Diagnostic species
- Gomphrena globosa. Reddish local lesions followed by systemic yellow
ringspots and line patterns.
- Chenopodium quinoa. Chlorotic/necrotic local lesions, systemic mottling and tip necrosis.
- Phaseolus vulgaris (French bean). Reddish necrotic local lesions, systemic mosaic and leaf deformation.
- Cucumis sativus (cucumber). Chlorotic lesions on cotyledons followed by mosaic, malformation and enations in non-inoculated leaves.
- Nicotiana tabacum (tobacco). Local and systemic chlorotic rings, lines and oakleaf patterns (Fig. 2).
- Chenopodium quinoa. Chlorotic/necrotic local lesions, systemic mottling and tip necrosis.
- Propagation species
- Phaseolus vulgaris, Chenopodium quinoa and Cucumis sativus
are good sources of virus for purification. G. globosa is suitable for
maintaining virus cultures.
- Assay species
- Chenopodium quinoa, Cucumis sativus and Canavalia ensiformis (Fig. 3) are satisfactory local lesion hosts.
Strains
Virus isolates from Italy and Greece differ in host range responses but are serologically indistinguishable (Rana et al., 1980).
Transmission by Vectors
No vector known. Italian and Greek isolates were not transmitted by Myzus persicae (Rana et al., 1980) and an intensive search for possible longidorid nematode vectors in Greece was unsuccessful (F. Roca & G. L. Rana, unpublished information).
Transmission through Seed
Transmitted through seed at rates ranging from 15 to 100% in artificially infected Chenopodium amaranticolor, C. quinoa, Datura stramonium, Nicotiana clevelandii, N. glutinosa, N. rustica and Petunia hybrida, and in naturally infected Anethum graveolens, Foeniculum sp., N. glauca, N. tabacum, Reseda alba and Stellaria media (P. E. Kyriakopoulou, unpublished information). Pollen transmission to seeds occurs in D. stramonium, N. clevelandii, N. glutinosa, N. tabacum, P. hybrida and transmission through pollen to pollinated plants was found in N. clevelandii (P. E. Kyriakopoulou, unpublished information).
Serology
The virus is weakly immunogenic. Antisera with titres not higher than 1/32 were obtained. The best antisera still had a titre of 1/2 to healthy plant proteins. In gel double diffusion tests no precipitin lines form unless virus particles are dissociated with SDS or ethanolamine (Rana et al., 1980). Immunosorbent electron microscopy and antibody coating can be used for virus identification.
Relationships
The virus is classified in the nepovirus group on the basis of particle size and morphology, physico-chemical properties, transmission through seed and intracellular behaviour. However, it was serologically unrelated to any of 26 different viruses with isometric particles, including the following 13 nepoviruses: artichoke Italian latent (grapevine and artichoke isolates), arabis mosaic, cherry leaf roll (rhubarb and walnut strains), chicory yellow mottle, grapevine Bulgarian latent, cocoa necrosis, tomato black ring (potato bouquet and beet ringspot strains), tomato ringspot, grapevine fanleaf, myrobalan latent ringspot, strawberry latent ringspot (type, peach and olive strains), grapevine chrome mosaic and peach rosette mosaic (Rana et al., 1980).
Stability in Sap
In expressed sap of French bean the virus was still infective after dilution to 10-3 or 10-4. Infectivity was lost after heating for 10 min at 60°C or storing at 22-24°C for 48-72 h.
Purification
Virus particles show a strong tendency to aggregate during extraction and purification. However, the following method (Rana et al., 1980) gives satisfactory results: homogenise each 100 g French bean leaves, collected 10-12 days after inoculation, in 200 ml each of chloroform and neutral 0.1 M phosphate buffer containing 0.6% sodium ascorbate, 4% Triton X-100 and 25% sucrose. Centrifuge at low speed and precipitate the virus from the supernatant fluid by adding polyethylene glycol to 10% (w/v) and NaCl to 1% (w/v); centrifuge at low speed and resuspend the pellets overnight at 4°C in neutral 0.02 M phosphate buffer. The virus particles can then be separated from most host constituents by alternate cycles of low speed centrifugation (20 min at 7000 g) in 25% sucrose and high speed centrifugation (1 h at 90,000 g) in 12.5% sucrose, retaining the supernatant fluid and pellets, respectively. Further purification can be obtained by centrifuging partially purified preparations to equilibrium using the ‘step’ isopycnic CsCl method as modified by Sehgal et al. (1970).
Properties of Particles
Particles are of two types: ‘empty’ shells (T component) and ‘full’ particles containing RNA. Because of the strong aggregation of particles during purification, preparations cannot be satisfactorily fractionated. In ‘step’ isopycnic CsCl centrifugation, most of the ‘empty’ shells band in the upper part of the gradients (density 1.20-1.30 g/cm3) whereas ‘full’ particles sediment in the lower part (density 1.40-1.53 g/cm3) (Rana et al., 1980).
Particle Structure
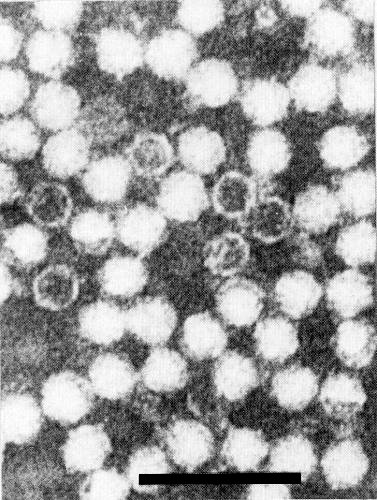
Particles are isometric, c. 30 nm in diameter and show angular outlines. ‘Empty’ particles (possibly T component) are penetrated by negative stain (Fig. 4).
Particle Composition
Nucleic acid: Single-stranded RNA. In polyacrylamide gel electrophoresis the RNA migrates as two species (RNA-1 and RNA-2) with estimated M. Wt (x 10-6) of 2.3 and 1.9 respectively under non-denaturing conditions (Bishop, Claybrook & Spiegelman, 1967) or 2.17 and 1.85 respectively under denaturing conditions (Reijnders et al., 1974). Both RNA species are necessary for infectivity (Rana et al., 1980).
Protein: In polyacrylamide/SDS gels, protein preparations from unfractionated virus contained a single polypeptide of estimated M. Wt 53,000.
Relations with Cells and Tissues
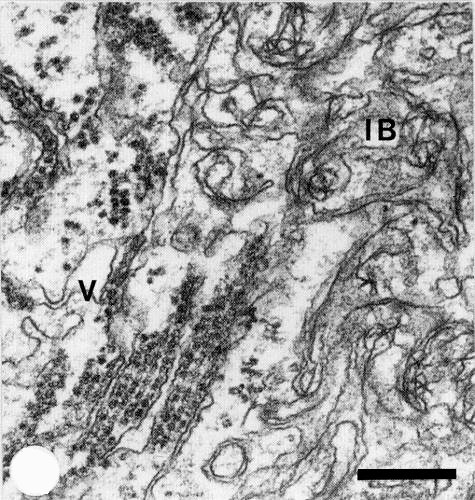
The virus is present in most plant tissues both in leaves and roots. Cytopathological modifications (Russo et al., 1978) consist of cytoplasmic inclusion bodies made up of accumulations of tangled membranes (Fig. 5) and vesicles containing finely stranded material resembling nucleic acid. Dictyosomes are much affected and may take part in some steps of virus multiplication (possibly assembly of virions). Virus particles occur in the cytoplasm at the periphery of the inclusions in stacked layers or between membranes (Fig. 5) or within tubular structures. Aggregates of empty particles may be present in the nucleoplasm. Modifications of cell walls (callose deposition and wall outgrowths) and of the cell wall-plasmalemma interface (paramural bodies) are very common.
Notes
The virus has biological, physico-chemical and ultrastructural properties of nepoviruses. However, it is not serologically related to any of the known members of this group that have been tested and it has no known vector. Because of the M. Wt of its RNA-2, artichoke yellow ringspot virus can be assigned to the cherry leaf roll virus subgroup (Martelli et al., 1978). The virus is transmitted to plants through pollen, as may happen with cherry leaf roll virus (Mircetich, Sanborn & Ramos, 1981). Artichoke yellow ringspot virus may be distinguished from other viruses occurring in the same natural hosts by differences in artificial host range and symptomatology, by its strong tendency to aggregate during extraction and purification, and by serological reactions (Rana et al., 1980). The field syndrome observed in artichoke and cardoon plants is distinctive and unlike that shown by the same species when infected by other viruses (Martelli, Russo & Rana, 1981).
Figures
References list for DPV: Artichoke yellow ringspot virus (271)
- Bishop, Claybrook & Spiegelman, J. molec. Biol. 26: 373, 1967.
- Kyriakopoulou & Bem, Symp. Agric. Res., Athens, 1973: 64, 1973.
- Martelli, Quacquarelli, Gallitelli, Savino & Piazolla, Phytopath. Medit. 17: 145, 1978.
- Martelli, Russo & Rana, Atti 3° Congr. Internaz. Carciofo, Bari, 1979: 895, 1981.
- Mircetich, Sanborn & Ramos, Phytopathology 71: 962, 1981.
- Rana, Rosciglione & Cannizzaro, Phytopath. Medit. 17: 63, 1978.
- Rana, Gallitelli, Kyriakopoulou, Russo & Martelli, Ann. appl. Biol. 96: 177, 1980.
- Reijnders, Aalbers, Van Kammen & Thuring, Virology 60: 515, 1974.
- Russo, Martelli, Rana & Kyriakopoulou, Microbiologica 1: 81, 1978.
- Sehgal, Jean, Bhalla, Soong & Krause, Phytopathology 60: 1778, 1970.