Details of DPV and References
DPV NO: 284 July 1984
Family: Tombusviridae
Genus: Machlomovirus
Species: Maize chlorotic mottle virus | Acronym: MCMV
Maize chlorotic mottle virus
D. T. Gordon Department of Plant Pathology, The Ohio State University, Ohio Agriculture Research and Development Center, Wooster OH 44691, USA
O. E. Bradfute Department of Plant Pathology, The Ohio State University, Ohio Agriculture Research and Development Center, Wooster OH 44691, USA
R. E. Gingery Agricultural Research Service, USDA, The Ohio State University, Ohio Agriculture Research and Development Center, Wooster OH 44691, USA
L. R. Nault Department of Entomology, The Ohio State University, Ohio Agriculture Research and Development Center, Wooster OH 44691, USA
J. K. Uyemoto Department of Plant Pathology, Kansas State University, Manhattan, Kansas 66506, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Disease and virus were originally described by
Castillo & Hebert (1974) and
Hebert & Castillo (1973).
A virus with small isometric RNA-containing particles c. 30 nm in diameter which sediment as a single component. It causes chlorotic mottling, necrosis and stunting of maize, with poor or no ear development and premature plant death. The vectors are chrysomelid beetles which retain ability to transmit the virus for up to 6 days. The virus is transmissible by mechanical inoculation, and infects only species of Gramineae. Reported from Peru, Argentina, Mexico and USA but economically important only in limited coastal areas of Peru and in Kansas and Nebraska, USA.
Main Diseases
The virus causes a variety of symptoms in maize (Zea mays), depending upon genotype, age of infection and environmental conditions. They range from a relatively mild chlorotic mottle to severe stunting, leaf necrosis, premature plant death, shortened male inflorescences with few spikes, and/or shortened, malformed, partially filled ears (Castillo Loayza, 1977; Castillo & Hebert, 1974; Niblett & Claflin, 1978; Uyemoto et al., 1981). The virus caused crop losses of 10-15% in natural field infections and losses of up to 59% in experimental maize plots (Castillo Loayza, 1977).
In the USA, the virus acts synergistically with maize dwarf mosaic virus strains A or B, or with wheat streak mosaic virus, to produce the economically damaging corn lethal necrosis disease (Niblett & Claflin, 1978; Uyemoto, Bockelman & Claflin, 1980; Uyemoto et al., 1981); crop losses as high as 91% have been reported (Niblett & Claflin, 1978). Leaves of corn plants inoculated early (three to seven leaf stage) with both maize chlorotic mottle virus and maize dwarf mosaic virus turn chlorotic then necrotic and plants become stunted, die prematurely, and bear small, deformed and partially filled or no ears (Uyemoto et al., 1981). Plants inoculated later (14-leaf stage) are not stunted but terminal leaves develop chlorosis and necrosis (Uyemoto et al., 1981) and ears are of normal size but kernel weight is greatly reduced. The virus complex was also associated with some late-infected plants that showed normal green foliage but bore prematurely yellowed and necrotic ears with shrivelled kernels (Uyemoto et al., 1980).
Geographical Distribution
The virus has been reported only in the Western Hemisphere countries of Peru (Castillo Loayza, 1977; Castillo & Hebert, 1974; Hebert & Castillo, 1973; Nault et al., 1979), Argentina (Teyssandier, Nome & Dalbo, 1983), Mexico (D. T. Gordon & O. E. Bradfute, unpublished data) and the USA (Doupnik, Lane & Wysong, 1982; Niblett & Claflin, 1978; Uyemoto et al., 1980). It is probably present in other Western Hemisphere countries wherever its chrysomelid vectors are found in maize growing areas. Its verified field vectors (Diabrotica species) occur only in the New World (Krysan & Branson, 1983).
Host Range and Symptomatology
The experimental host range is restricted to the Gramineae (Poaceae) with maize as a natural host. In mechanical inoculations, both Kansas and Peru serotypes systemically infected Bromus spp., Digitaria sanguinalis, Eragrostis trichodes, Hordeum spp., Panicum spp., Setaria spp., Sorghum spp., Triticum aestivum (Bockelman, Claflin & Uyemoto, 1982; Castillo & Hebert, 1974; Niblett & Claflin, 1978) and Zea mays ssp. mays and mexicana (Castillo & Hebert, 1974; Nault et al., 1982). The Kansas serotype 1 also infected Z. mays ssp. parviglumis and Z. luxurians but not Z. perennis and Z. diploperennis (Nault et al., 1982). Dicotyledonous species were not infected when mechanically inoculated (Castillo & Hebert, 1974; Niblett & Claflin, 1978).
-
Diagnostic species
- Zea mays
(maize). Numerous dent inbreds and hybrids as well as sweet corn and popcorn types are very susceptible. Symptoms include chlorotic mottling (Fig. 1), leaf necrosis and plant stunting (Castillo Loayza, 1977; Uyemoto et al., 1981). Tolerant genotypes develop chlorotic mottle or mosaic, or latent infections. These genotypes may also be susceptible to the mechanically transmissible maize dwarf mosaic and wheat streak mosaic viruses, which cause symptoms indistinguishable from those induced by maize chlorotic mottle virus. - Sorghum bicolor (sorghum) cv. Asgrow Bugoff. Immune. Also immune to wheat streak
mosaic virus. This sorghum cultivar is useful in distinguishing the virus from maize dwarf mosaic
virus strain A, which causes chlorotic mottle or red leaves, and strain B, which causes intense
systemic leaf reddening
(Uyemoto et al., 1980).
- Triticum aestivum (wheat) cv. Parker. Occasionally develops chlorotic spots and rarely a faint systemic mottle. This host is useful in differentiating the virus from wheat streak mosaic virus, which causes systemic chlorotic streaks, and from maize dwarf mosaic virus, which causes no infection (Uyemoto et al., 1980).
-
Propagation and assay species
- Maize inbreds H84, N28, N28Ht and Oh28, maize hybrid PM-205, and the sweet corn cultivar Aristogold Bantam Evergreen served as virus assay or propagation hosts for studies on mechanical and beetle transmission and on virus characterisation (Bockelman et al., 1982; Castillo & Hebert, 1974; Ferguson, Shahin & Uyemoto, 1978; Nault et al., 1978; Niblett & Claflin, 1978; Phillips, Uyemoto & Wilson, 1982; Uyemoto et al., 1980, 1981) or for virus purification (R. E. Gingery, unpublished data; Niblett & Claflin, 1978; Uyemoto, 1980). No reliable local lesion assay host has been reported.
Strains
Three strains or serotypes have been reported: Kansas serotypes 1 and 2 (Uyemoto et al., 1980) and the Peru serotype (Niblett & Claflin, 1978; Niblett, Claflin & Hebert, 1977). All three serotypes were readily differentiated by formation of spurs in double immunodiffusion tests in agar gels (Niblett & Claflin, 1978; Niblett et al., 1977; Uyemoto, 1980); in addition, the Kansas serotype 1 and the Peru serotype were differentiated by enzyme-linked immunosorbent assay (ELISA) (Uyemoto, 1980) and differential host responses (Bockelman et al., 1982). Both Kansas serotypes were detected in Nebraska (J. K. Uyemoto, unpublished data). The serotypes of the isolates from Argentina and Mexico have not been determined.
Transmission by Vectors
Adults of six species of chrysomelid beetle transmitted the virus experimentally in the USA (Jensen, 1979; Nault & Knoke, 1981; Nault et al., 1978): Diabrotica virgifera virgifera (western corn rootworm) (Fig. 2), D. barberi (=longicornis) (northern corn rootworm), D. undecimpunctata howardi (southern corn rootworm), Chaetocnema pulicaria (corn flea beetle), Systena frontalis (a flea beetle) and Oulema melanopa (cereal leaf beetle). In Peru, adults of D. viridula and D. decempunctata sicuanica were vectors (Castillo, 1983). Larval forms of D. undecimpunctata (Jensen, 1979), D. virgifera virgifera, D. barberi (S. G. Jensen, personal communication), D. viridula (Castillo, 1983) and O. melanopa (Nault et al., 1978) also transmitted the virus. Only Diabrotica species have been implicated as vectors in the field. They occur only in the New World, most species having a tropical distribution (Krysan & Branson, 1983), whereas the experimental vectors O. melanopa and C. pulicaria occur worldwide. Both adult and larval forms of Diabrotica spp. appear important in virus epidemiology (Uyemoto, 1983a, 1983b).
The virus was acquired and inoculated by beetles after feeding periods of less than 24 h each (Nault & Knoke, 1981). It persisted in adult D. virgifera virgifera for 3 days and in O. melanopa for 4-6 days at room temperature (Nault et al., 1978). There is no latent period in beetles (Jensen, 1979). Adult O. melanopa, developed from viruliferous larvae, failed to transmit (Nault et al., 1978), suggesting that beetle-transmissible virus neither is retained in the gut and regurgitated during feeding nor does it multiply or circulate in the vector to be transmitted via the saliva.
Transmission through Seed
The virus was not transmitted through seed of maize inbreds (2153 seedlings) or hybrids (1898 seedlings) (Bockelman et al., 1982) or hybrid PM-205 (800 seedlings) (Castillo Loayza, 1977; Castillo & Hebert, 1974) nor through seed of Panicum miliaceum (842 seedlings), Setaria lutescens (135 seedlings) or S. viridis (2676 seedlings) (Bockelman et al., 1982).
Serology
The virus is moderately to highly immunogenic. Antisera were produced in rabbits by intramuscular injections, at 7-day intervals, of purified virus emulsified in Freund's complete or incomplete adjuvant (Castillo & Hebert, 1974; Niblett & Claflin, 1978; Paul et al., 1980) or by a combination of intramuscular and intravenous injections spaced at 10-day intervals (Uyemoto, 1980). The antisera had titres of 1/512 to 1/2048 in gel double diffusion tests (Paul, Querfurth & Huth, 1980; Uyemoto, 1980), homologous reactants producing a single precipitin line (Uyemoto & Claflin, 1981). Serological tests most commonly used are gel double diffusion (Bockelman et al., 1982; Castillo & Hebert, 1974; Nault et al., 1978; Niblett & Claflin, 1978; Paul et al., 1980; Phillips et al., 1982: Uyemoto, 1980; Uyemoto et al.. 1980, 1981) and ELISA (Bockelman et al., 1982; Nault et al., 1982; Phillips et al., 1982; Teyssandier et al., 1983; Uyemoto, 1980; Uyemoto et al., 1980). Other tests employed are microprecipitin assay (Nault et al., 1978, 1979, 1982), immunoelectrophoresis (Paul et al., 1980), immunosorbent electron microscopy (Lesemann & Paul, 1980) and immunofluorescence (Gingery & Gordon, 1981; Nault et al., 1979).
Relationships
Maize chlorotic mottle virus resembles a number of other viruses that have host ranges confined to the Gramineae, high infectivity titres, high thermal inactivation points and isometric particles of c. 28 nm diameter, and that are easily transmitted mechanically, sediment as single components, and contain c. 21% RNA (Paul et al., 1980). However, it is unrelated serologically to several such viruses: brome stem leaf mottle, cocksfoot mild mosaic, cocksfoot mottle, cynosurus mottle, holcus transitory mottle, molinia streak, panicum mosaic, phleum mottle and St Augustine decline viruses (Castillo & Hebert, 1974; Hebert & Castillo, 1973; Niblett & Claflin, 1978; Paul et al., 1980). It is also unrelated serologically to brome mosaic, rice yellow mottle and sowbane mosaic viruses (Castillo & Hebert, 1974; Hebert & Castillo, 1973; Niblett & Claflin, 1978), barley stripe mosaic, foxtail mosaic, maize dwarf mosaic and wheat streak mosaic viruses (Niblett & Claflin, 1978), and to maize chlorotic dwarf, maize rayado fino, maize streak, maize stripe and maize white line mosaic viruses (D. T. Gordon, unpublished data). It has most properties in common with the sobemoviruses or with the viruses related to cocksfoot mild mosaic virus.
Stability in Sap
Sap from infected maize had a thermal inactivation point of 80-85°C (Castillo Loayza, 1977; Castillo & Hebert, 1974; Ferguson et al., 1978; Hebert & Castillo, 1973; Uyemoto & Claflin, 1981), a dilution end-point of 10-6 (Castillo Loayza, 1977; Castillo & Hebert, 1974; Hebert & Castillo, 1973) or 10-8 (Ferguson et al., 1978; Uyemoto & Claflin, 1981) and a longevity in vitro at 20°C greater than 12 days (Castillo Loayza, 1977; Castillo & Hebert, 1974) or 33 days (Ferguson et al., 1978; Uyemoto & Claflin, 1981).
Purification
The following methods gave good yields (up to 1 mg/g fresh weight tissue).
1. Adaptations of the Niblett & Paulsen (1975) method for panicum mosaic virus (Ferguson et al., 1978; Niblett & Claflin, 1978; Uyemoto, 1980). Blend infected maize leaves in cold 0.1 M potassium phosphate, pH 7.0, plus 1% 2-mercaptoethanol (1 g tissue/3 ml buffer) and strain through cheesecloth. Clarify extract by slowly adding an equal volume of cold chloroform/butanol (1:1, v/v) with gentle stirring. Incubate emulsion for 30 min at 4°C and recover aqueous phase by centrifugation (5000 g, 7 min). Filter through glass wool and pellet virus particles (147,000 g, 2 h); suspend the pellets in 0.02 M phosphate buffer, pH 7, and further purify by two cycles of differential centrifugation (12,000 g, 10 min; 269,000 g, 90 min); resuspend finally in 0.02 M phosphate (2.5 ml/ 100 g). For large extract volumes, centrifuge (12,000 g, 10 min) and precipitate the virus by adding polyethylene glycol (PEG), M. Wt 6000, to 8% (w/v) and NaCl to 0.2 M (2 h, room temperature) and recover precipitate (8000 g, 10 min). Suspend in 0.1 M phosphate buffer, clarify with cold chloroform:butanol (1:1, w/v), and concentrate the virus by two cycles of differential centrifugation. Additional purification is achieved by rate-zonal centrifugation (Castillo & Hebert, 1974; Niblett & Claflin, 1978) or zone electrophoresis (Ferguson et al., 1978; Uyemoto, 1980).
2. (R. E. Gingery, unpublished data). Blend infected maize leaves in 0.2 M K2HPO4, adjusted to pH 5.0 with 0.1 M citric acid, plus 0.5% 2-mercaptoethanol (1 g tissue/ml buffer). Clarify with 0.25 vol. chloroform and precipitate virus from the aqueous phase by adding PEG to 6% (w/v) and KCl to 4% (w/v). Recover precipitate by centrifugation (10,000 g, 20 min) and further purify by rate-zonal sucrose density-gradient centrifugation and isopycnic banding in CsCl.
Properties of Particles
Purified virus preparations contain a single sedimenting component.
Sedimentation coefficient, s20,w: 109 S (Niblett & Claflin, 1978; Niblett et al., 1977), not adjusted for infinite dilution.
A260/A280: 1.69-1.73 (R. E. Gingery, unpublished data).
Buoyant density in CsCl: 1.365 g/cm3 (R. E. Gingery, unpublished data).
Particle Structure
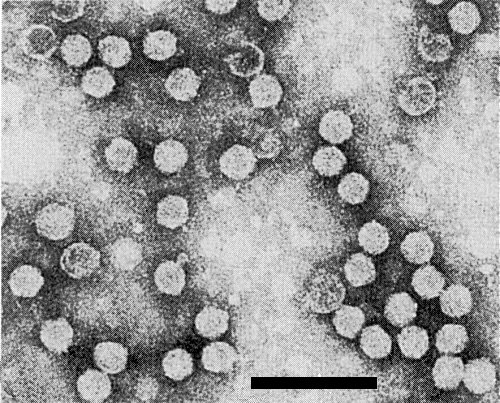
Particles were isometric, c. 30 nm in diameter when negatively stained in potassium phosphotungstate, pH 6.9 (Fig. 3) (O. E. Bradfute, unpublished data; Castillo Loayza, 1977; Castillo & Hebert, 1974) and c. 33 nm when stained in uranyl acetate (Fig. 4) (O. E. Bradfute, unpublished data). Many particles were hexagonal in outline and some were penetrated by stain. The detailed particle structure is unknown.
Particle Composition
Nucleic acid: RNA, probably single-stranded, with M. Wt 1.4-1.6 x 106 (Ferguson et al., 1978; R. E. Gingery, unpublished data; Niblett et al., 1977). Constitutes c. 18% of the particle weight (R. E. Gingery, unpublished data). Molar percentage of nucleotides: G22-25; A24-26; C15-16; U34-37 (R. E. Gingery, unpublished data),
Protein: Single migrating protein species, of M. Wt 23.9-27.0 x 103 (Ferguson et al., 1978; Niblett et al., 1977; Paul et al., 1980), constituting c. 82% of the particle weight.
Other components: None reported.
Relations with Cells and Tissues
No studies reported.
Notes
Two other mechanically transmissible viruses that commonly infect maize are maize dwarf mosaic virus and wheat streak mosaic virus. Maize chlorotic mottle virus may be distinguished from these two filamentous viruses by the reactions of sorghum and wheat and by particle morphology. Maize chlorotic mottle virus is easily differentiated from other viruses commonly infecting maize in the Western Hemisphere (maize chlorotic dwarf virus, maize mosaic virus, maize rayado fino virus, maize stripe virus and maize white line mosaic virus), because none of these is mechanically transmissible. All except the last, which is soil-borne, are transmitted by either leafhoppers or planthoppers. Among viruses rarely identified from diseased maize in the Western Hemisphere, brome mosaic virus and cucumber mosaic virus are mechanically transmissible but have quite different host ranges from that of maize chlorotic mottle virus, and barley yellow dwarf virus is obligately aphid transmitted.
Figures

Symptoms on corn hybrid B73 x FR16 inoculated at or near tassel stage of growth and photographed about 4 weeks later.
References list for DPV: Maize chlorotic mottle virus (284)
- Bockelman, Claflin & Uyemoto, Pl. Dis. 66: 216, 1982.
- Castillo Loayza, Proc. int. Maize Virus Dis. Colloq. Workshop, Wooster, Ohio, 1976: 40, 1977.
- Castillo, Proc. int. Maize Virus Dis. Colloq. Workshop, Wooster, Ohio, 1982: 87, 1983.
- Castillo & Hebert, Fitopatologia 9: 79, 1974.
- Doupnik, Lane & Wysong, Phytopathology 72: 939, 1982.
- Ferguson, Shahin & Uyemoto, Phytopath. News 12: 87, 1978.
- Gingery & Gordon, South. Coop. Ser. Bull. 247: 19, 1981.
- Hebert & Castillo, Abstr. 2nd int. Congr. Pl. Path., Minneapolis, Minn., 1973: Abstr. No. 0072, 1973.
- Jensen, Abstr. IXth int. Congr. Pl. Prot., Washington, D.C., 1979: Abstr. No. 459, 1979.
- Krysan & Branson, Proc. int. Maize Virus Dis. Colloq. Workshop, Wooster, Ohio, 1982: 144, 1983.
- Lesemann & Paul, Acta Hort. 110: 119, 1980.
- Nault & Knoke, South. Coop. Ser. Bull. 247: 77, 1981.
- Nault, Styer, Coffey, Gordon, Negi & Niblett, Phytopathology 68: 1071, 1978.
- Nault, Gordon, Gingery, Bradfute & Castillo Loayza, Phytopathology 69: 824, 1979.
- Nault, Gordon, Damsteegt & Iltis, Pl. Dis. 66: 61, 1982.
- Niblett & Claflin, Pl. Dis. Reptr 62: 15, 1978.
- Niblett & Paulsen, Phytopathology 65: 1157, 1975.
- Niblett, Claflin & Hebert, Proc. Am. Phytopath. Soc. 4: 90, 1977.
- Paul, Querfurth & Huth, J. gen. Virol. 47: 67, 1980.
- Phillips, Uyemoto & Wilson, Pl. Dis. 66: 376, 1982.
- Teyssandier, Nome & Dalbo, Proc. int. Maize Virus Dis. Colloq. Workshop., Wooster, Ohio, 1982: 93, 1983.
- Uyemoto, Phytopathology 70: 290, 1980.
- Uyemoto, Pl. Dis. 67: 7, 1983a.
- Uyemoto, Proc. int. Maize Virus Dis. Colloq. Workshop, Wooster, Ohio, 1982: 141, 1983b.
- Uyemoto & Claflin, South. Coop. Ser. Bull. 247: 163, 1981.
- Uyemoto, Bockelman & Claflin, Pl. Dis. 64: 99, 1980.
- Uyemoto, Claflin, Wilson & Raney, Pl. Dis. 65: 39, 1981.