Details of DPV and References
DPV NO: 287 July 1984
Family: Pospiviroidae
Genus: Cocadviroid
Species: Coconut cadang-cadang viroid | Acronym: CCCVd
There is a more recent description of this viroid: DPV 402
Coconut cadang-cadang viroid
J. W. Randles Waite Agricultural Research Institute, The University of Adelaide, South Australia, Australia
J. S. Imperial Philippine Coconut Authority, Guinobatan, Albay, Philippines
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Disease first described by Ocfemia (1937), the viroid by Randles (1975).
A viroid consisting of circular or linear single-stranded RNA with a basic size of 246 or 247 nucleotides occurring as monomeric and dimeric forms in Cocos nucifera. Larger molecular forms appear as the disease progresses. Mechanically transmissible using nucleic acid inocula, but mode of natural transmission unknown. Detected only in Palmae. Distributed in the Philippines and Guam.
Main Diseases
Cadang-cadang is a lethal disease of coconut (Cocos nucifera) recognised by a generalised yellow-bronze coloration of the lower two-thirds of fronds in the crown, cessation of nut production, crown diminution and death (Fig. 1). The third or fourth fronds below the spear leaf show small chlorotic leaf spots which expand and coalesce during ageing of the frond to produce a chlorotic mottle (Fig. 5). As the spots appear, the nuts become smaller and rounded (Fig. 3) (Mohamed et al., 1982), and many of them develop equatorial scarifications. Inflorescences later become necrotic and infertile, and nut production ceases. Frond production and size gradually decline (Randles et al., 1977), leaflets become brittle, and death of the crown ensues. Palms die between 8 and 16 years after the onset of symptoms (Zelazny & Niven, 1980). Palms less than 10 years old are very rarely affected, and the incidence increases nearly linearly up to palm age of about 40 years, thereafter remaining constant (Zelazny et al., 1982). Total losses since recognition of the disease are estimated at c. 30 million palms (Zelazny et al., 1982). Tinangaja is a similar disease, but differs in that the nuts are characteristically small and elongated, and lacking a kernel, i.e. ‘mummified’ (Fig. 2; Boccardo et al., 1981).
Naturally infected oil palm (Elaeis guineensis) shows a progression from general chlorosis on younger fronds to development of translucent orange leaf spots (Fig. 4) and cessation of inflorescence production (Randles, Boccardo & Imperial, 1980).
Geographical Distribution
Cadang-cadang disease is recognised in the central Philippines (southern Luzon, Samar, Masbate and smaller islands within a zone about 600 km x 300 km). Tinangaja disease occurs on Guam (Marianas Islands). Distribution is unaffected by water barriers, but incidence is negatively correlated with altitude (Zelazny, 1980).
Host Range and Symptomatology
Known hosts are confined to the Palmae. Transmitted experimentally by high pressure injection combined with pricking (Randles et al., 1977).
-
Diagnostic species
- Cocos nucifera
(coconut). Inoculated seedlings show spotting on leaflets of fronds that are at least three positions below the unexpanded spear leaf (Fig. 5), together with reduction in palm growth, frond size and production, and failure to bear nuts at the expected time. - Chlorotic or orange spotting of leaflets is characteristic of other inoculated
species of palm such as betel nut palm (Areca catechu), bori palm (Corypha
elata), Manila palm (Adonidia merrillii), oil palm, palmena
(Chrysalidocarpus lutescens) and royal palm (Oreodoxa regia).
Propagation species
- Naturally infected Cocos nucifera.
Assay species
- Cocos nucifera
is used as a systemic assay host; the viroid is detected by gel electrophoretic or molecular hybridisation assay 1-2 years after inoculation.
Strains
All sequenced isolates of the coconut cadang-cadang viroid have the same minimal nucleotide sequence (Haseloff, Mohamed & Symons, 1982); modifications to the structure are not strain differences but are related to stage of disease development (Imperial, Rodriguez & Randles, 1981). The symptoms of tinangaja disease differ somewhat from those of cadang-cadang disease and the causal viroid is regarded as a strain, although the nucleotide sequence is not available.
Transmission by Vectors
Mode of natural transmission is unknown.
Transmission through Seed
Very low, e.g. 1 out of 320 of seedlings tested (Anon., 1982).
Serology
No viroid-specific antibodies were detected in rabbits injected with the viroid-containing polyethylene glycol-insoluble fraction from coconut leaves (J. W. Randles & O. W. Barnett, unpublished data).
Relationships
The coconut cadang-cadang viroid is the smallest known pathogen, and is biologically distinct from other viroids. It has two partially complementary regions of about 20 nucleotides (I, Fig. 8) that are almost identical to regions in the potato spindle tuber, chrysanthemum stunt and citrus exocortis viroids, but no other regions are homologous. It shows no significant nucleotide sequence homology with avocado sunblotch viroid (Symons, 1981) which is only one nucleotide larger than the smallest form of coconut cadang-cadang viroid.
The viroid associated with tinangaja disease is similar in size to the cadang-cadang viroid and has nucleotide sequence homology with it. The extent of the differences between these two viroids has yet to be determined (Boccardo et al., 1981).
Stability in Sap
No information on survival of infectivity in sap.
Purification
Chop leaflets from fronds four or more below the spear leaf, blend with 3 vol. 0.1 M Na2SO3, filter the extract through cloth mesh, clarify by centrifugation at 10,000 g for 10 min, and add polyethlene glycol (PEG), M. Wt 6000, to 5% (w/v). After 1-2 h incubation at 4°C, centrifuge at low speed and extract nucleic acids from the precipitate by protease, phenol-sodium dodecyl sulphate, or chloroform procedures (Randles, 1975; Randles et al., 1976). The viroid may also be recovered directly from the PEG precipitate by resuspending it in 1 mM EDTA and precipitating the viroid with 3 vol. ethanol (J. W. Randles and J. S. Imperial, unpublished data). Subject extracts to electrophoresis in 5% polyacrylamide gels, and detect the bands by staining in aqueous toluidine blue. Excise the bands and lay them over a gel containing 8 M urea (Randles, Steger & Riesner, 1982). A second electrophoresis under these partially denaturing conditions separates circular and linear forms of the viroid monomer and dimer. Again excise the bands and extract the RNA by maceration and extraction of gel followed by sucrose density gradient centrifugation (Randles & Palukaitis, 1979), or by preparative gel electrophoresis, or by electrophoresis into agarose gel (Randles et al., 1982) followed by gel maceration and extraction. RNA obtained by either method is precipitated with ethanol, dried, resuspended in 0.2 M NaCl, then reprecipitated with 1% cetyl trimethyl ammonium bromide to remove gel residue which remains in solution.
Viroid samples are assayed by electrophoresis in polyacrylamide gels (Fig. 6) and the viroid is detected by staining with toluidine blue, ethidium bromide or silver nitrate (Schumacher, Randles & Riesner, 1983).
Properties of Infective Nucleic Acid
Occurs as monomer and dimer, with sedimentation coefficients of 7 S and
10 S
(Riesner, Kaper & Randles, 1982).
Monomer M. Wt is 83,000.
A260/A280: 2.1.
Buoyant density in Cs2SO4: c. 1.60 g/cm3 (Randles et al., 1976).
Molecular Structure
Both monomer and dimer are single-stranded RNA occurring as covalently linked circular molecules (Fig. 7) or as linear molecules. The complete nucleotide sequence is known (Fig. 8). The smallest form comprises 246 nucleotides, but addition of a cytosine residue (*, Fig. 8) increases the size to 247 nucleotides (Haseloff et al., 1982). The dimer is detected simultaneously with the monomer. As infection develops these small forms are replaced by large forms (Fig. 6) which arise through reiteration of a sequence at the ‘right hand’ end of the native molecule (region II, Fig. 8).
The native monomer shows two thermal transitions in 10 mM Na+, at 49° and 58°C; the first represents cooperative melting of the double-stranded regions to allow the formation of a stable intermediate, the second represents melting of the intermediate to yield a covalently linked open circle (Randles et al., 1982). The native viroid has 70% GC pairs, the half-width of the first thermal transition (DT½) is 1.4°C (Randles et al., 1982).
The structure of the viroid is more stable than that of cell RNA species in incubated polyethylene glycol concentrates of coconut leaf sap (Randles, 1975; Randles, Rillo & Diener, 1976).
Relations with Cells and Tissues
The viroid has been detected by molecular hybridisation in the inflorescence and in the meristems, in fronds of various ages and in roots (J. W. Randles and G. Boccardo, unpublished data). Light microscopy of yellow leaf spots shows hypoplasia, and loss of chloroplast pigments; electron microscopy shows accumulation of dark structures which are presumed to be tannin bodies, but no disease-specific cytoplasmic changes have been recognised (Randles, 1985).
Notes
Coconut cadang-cadang is the most serious of the viroid diseases because of its lethality. Its area of distribution in the Philippines has increased very little in the last 26 years, but epidemics arise and decline within this zone (Zelazny et al., 1982). The mode of natural transmission is unknown, and eradication measures fail to control the disease. The viroid is not associated with Kerala wilt or Tatipaka disease of coconut palm in India, nor with a lethal disease of coconut palm in Vanuatu (J. W. Randles and M. Dollet, unpublished data).
Figures

Nuts typical of cadang-cadang (left) compared with mummified nuts associated with tinangaja. (Photograph courtesy of G. Boccardo, Istituto di Fitovirologia Applicata, Turin.)

Scarified and rounded nuts (right) from a palm at an early stage of infection, compared with normal nuts.

Chlorosis and yellow-orange spotting of two leaves (centre and right) of infected oil palm compared with healthy leaf (left).

Leaf spot development on second youngest open frond (top) to ninth frond of cadang-cadang infected coconut palm.
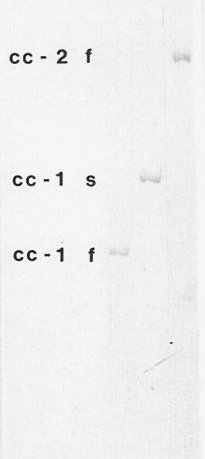
Polyacrylamide gel electrophoretic separation of small (cc-1f) and large (cc-1s) monomeric forms of the viroid and the small dimeric form (cc-2f).

Denatured circles of monomeric (above) and dimeric forms (below) of the viroid (Randles & Hatta, 1979). Bar represents 150 nm.
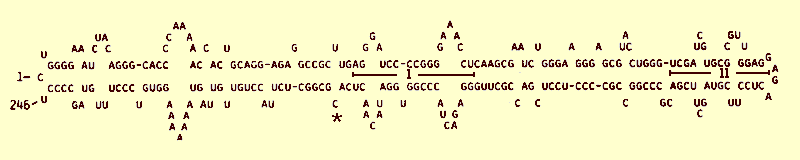
Primary sequence of the minimal infective form of coconut cadang-cadang viroid (from data of Haseloff et al., 1982, sequence J02049). Note the conserved sequences (I), the sequences (II) that are reiterated in variant molecules produced at later stages of infection, and the site (*) where cytosine occurs either as a single or double residue.
References list for DPV: Coconut cadang-cadang viroid (287)
- Anonymous, Cadang-cadang research, 1971-1982. Tech. Rep. F.A.O./U.N. Dev. Proj. AG:DP/PH1/523, 75 pp., 1982.
- Boccardo, Beaver, Randles & Imperial, Phytopathology 71: 1104, 1981.
- Haseloff, Mohamed & Symons, Nature, Lond. 299: 316, 1982.
- Imperial, Rodriguez & Randles, J. gen. Virol. 56: 77, 1981.
- Mohamed, Haseloff, Imperial & Symons, J. gen. Virol. 63: 181, 1982.
- Ocfemia, Philipp. Agric. 26: 338, 1937.
- Randles, Phytopathology 65: 163, 1975.
- Randles, in Subviral Pathogens of Plants and Animals: Viroids and Prions, ed. K. Maramorosch & J. J. McKelvey, New York, Academic Press, p. 39, 1985.
- Randles & Hatta, Virology 96: 47, 1979.
- Randles & Palukaitis, J. gen. Virol. 43: 649, 1979.
- Randles, Rillo & Diener, Virology 74: 128, 1976.
- Randles, Boccardo, Retuerma & Rillo, Phytopathology 67: 1211, 1977.
- Randles, Boccardo & Imperial, Phytopathology 70: 185, 1980.
- Randles, Steger & Riesner, Nucleic Acids Res. 10: 5569, 1982.
- Riesner, Kaper & Randles, Nucleic Acids Res. 10: 5587, 1982.
- Schumacher, Randles & Riesner, Analyt. Biochem. 135: 288, 1983.
- Symons, Nucleic Acids Res. 9: 6527, 1981.
- Zelazay, Phytopathology 70: 700, 1980.
- Zelazny & Niven, Pl. Dis. 64: 841, 1980.
- Zelazny, Randles, Boccardo & Imperial, Scientia Filipinas 2: 45, 1982.
