Details of DPV and References
DPV NO: 299 September 1985
Family: Unallocated ssRNA- viruses
Genus: Tenuivirus
Species: Rice hoja blanca virus | Acronym: RHBV
Rice hoja blanca virus
F. J. Morales Centro Internacional de Agricultura Tropical, Apartado Aéreo 6713, Cali, Colombia
A. I. Niessen Centro Internacional de Agricultura Tropical, Apartado Aéreo 6713, Cali, Colombia
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Disease first reported in Colombia by Garcés (1940). Virus isolated by Morales & Niessen (1983).
A virus with RNA-containing filamentous particles of undetermined length and 3-4 nm in diameter. The virus induces the formation of a non-structural protein in infected plant cells. It is efficiently transmitted by the planthopper Sogatodes orizicola in the persistent (propagative) manner and passes transovarially to the progeny. It is not transmitted to susceptible plants by mechanical inoculation. Plant host range is confined to the Gramineae. The virus is known to occur in most rice-growing countries of the American Continent, where it can cause yield losses of economic importance.
Main Diseases
Infected rice (Oryza sativa) plants usually exhibit characteristic chlorotic stripes on the leaf blade (Fig. 1) and are stunted; the panicles contain few or no seeds. New tillers may not show symptoms (Atkins & Adair, 1957). Varieties differ in symptom expression and the severity of the symptoms is inversely proportional to the age of the plant at inoculation. Necrosis and death of leaves may occur when plants are infected young. When infection takes place after emergence of the panicle, only slight yield reduction may occur (McGuire et al., 1960). Hoja blanca virus infection predisposes rice plants to brown spot caused by Helminthosporium oryzae (Lamey & Everett, 1967; Anon., 1984a).
Geographical Distribution
Reported only in the American Continent: Brazil, Belize, Colombia, Costa Rica, Cuba, Dominican Republic, El Salvador, Ecuador, Guatemala, Guyana, Honduras, Mexico, Nicaragua, Panama, Peru, Puerto Rico, Surinam, USA and Venezuela (Everett & Lamey, 1969). The disease is restricted largely to tropical America, mainly because of the poor overwintering capacity of the insect vector in cooler climates.
Host Range and Symptomatology
The virus is restricted to species of Gramineae. Not transmitted by manual inoculation but transmitted experimentally by the planthopper vector to Avena sativa, Digitaria horizontalis, Hordeum vulgare, Leptochloa filiformis and Secale cereale (Gálvez et al., 1961; Gálvez, 1968). In addition, the following plant species are reported to exhibit hoja blanca symptoms, although there is no unequivocal evidence that rice hoja blanca virus is the causal agent: Brachiaria plantaginea, Echinochloa colona, E. crus-galli, E. walteri, Panicum fasciculatum, P. capillare, Paspalum sp., Rottboellia exaltata and Triticum spp. (McGuire et al., 1960; Gibler et al., 1961).
-
Diagnostic species
- Oryza sativa
(rice). The characteristic symptoms are described under Main Disease. Rice cv. Bluebonnet 50 is highly susceptible whereas the cv. Colombia 1 is considered tolerant (Anon., 1984b). Most Japonica varieties are tolerant to the virus (Lamey, 1967).Propagation species
- Oryza sativa
(rice) cvs. Bluebonnet 50, CICA 8 and 1R22 are those most frequently used to propagate the virus (Morales & Niessen, 1983).Assay species
- There are no specific assay species other than susceptible rice cultivars. Bluebonnet 50 is the preferred cultivar in vector transmission tests but it is very susceptible to direct feeding damage by the vector. Other cultivars, such as CICA 8, are resistant to the planthopper vector but susceptible to the virus.
Strains
There is no evidence for the existence of strains in rice: differences in varietal reaction in different rice-producing countries are a consequence of variable vector incidence (Anon., 1984b). A virus with similar morphological and physicochemical properties (F. J. Morales & A. I. Niessen, unpublished data) occurs in Echinochloa colona, which is a common weed in rice fields and has been considered the main reservoir of rice hoja blanca virus. However, reciprocal gel double diffusion tests and ELISA (F. J. Morales & A. I. Niessen, unpublished data) suggest that the two viruses, though closely serologically related, are not identical; moreover, the virus from E. colona is not easily transmitted to rice (F. J. Morales & A. I. Niessen, unpublished data; Gálvez, 1968) and its specific vector, Sogatodes cubanus (van Hoof, 1959), is a poor vector of typical rice isolates. The virus from F. colona therefore seems best regarded as a strain of rice hoja blanca virus.
Transmission by Vectors
The virus is specifically transmitted by the rice planthopper Sogatodes orizicola (Fig. 7) (Malaguti et al., 1957; Acuña et al., 1958; Gálvez & Jennings, 1959; McGuire et al., 1961). S. cubanus transmits the virus with difficulty experimentally but cannot survive for long on rice (Gálvez et al., 1960; Gálvez, 1967; Everett & Lamey, 1969; Anon., 1984b). The minimum acquisition access periods reported for 100% of the vectors to acquire the virus range from approximately 1 h (Gálvez, 1967) to at least 12 h (McMillian et al., 1962). The incubation period of the virus in S. orizicola ranges from 10 to 40 days for most vector insects (Anon., 1984b). The minimum incubation period reported is 5 days (McGuire et al., 1960). Inoculation access periods of 24 h (McMillian et al., 1962) and 3 h (Gálvez, 1967) are reported to give 100% transmission. The virus has been shown to multiply in cells of the insect vector (Shikata & Gálvez, 1969), and to pass transovarially through 10 successive generations (Gálvez, 1967). Male and female adults as well as nymphs can transmit the virus. Males, because of their short life span, can only transmit virus acquired through the egg; females, with a longer life span, can transmit virus acquired during the nymphal stage.
Transmission through Seed
The virus does not appear to be transmitted through the seed produced by infected plants (Malaguti et al., 1957; Gálvez & Jennings, 1959; Anon., 1984b).
Serology
The virus is a good immunogen. The relatively weak reactions observed in double immunodiffusion tests in agar containing SDS (Purcifull & Batchelor, 1977) could be a consequence of the composition of the medium, which was originally prepared for viruses with larger flexuous filamentous particles. Enzyme-linked immunosorbent assay (ELISA) has proved a highly sensitive and specific serological test to detect the virus in infected plants and in individual planthoppers (F. J. Morales & A. I. Niessen, unpublished data). The non-structural protein induced by the virus in infected plants is also a good immunogen and antiserum to this protein reacts specifically with extracts from rice hoja blanca-affected plants (but not with extracts from viruliferous vectors) in ELISA (F. J. Morales & A. I. Niessen, unpublished data).
Relationships
Rice hoja blanca virus has similar particle morphology and vector relations to rice stripe virus (Koganezawa et al., 1975) and maize stripe virus (Gingery et al., 1981). However, no serological relationship to these viruses was observed in reciprocal Ouchterlony tests or in ELISA (F. J. Morales & A. I. Niessen, unpublished data). Moreover, antiserum to rice hoja blanca virus did not react with rice grassy stunt virus, another pathogen that apparently belongs to this novel group of viruses (F. J. Morales & A. I. Niessen, unpublished data). Rice hoja blanca virus is therefore regarded as a distinct member of the rice stripe virus group.
Stability in Sap
No information. The consistent isolation of the virus from young and old infected rice plants (Morales & Niessen, 1983), and acquisition of the virus by vector insects injected with extracts of infected leaves (Gálvez, 1967), suggest that the virus is relatively stable.
Purification
(Morales & Niessen, 1983). The whole plant, including the roots, can be used to isolate the virus. Homogenize the tissue with three volumes of a cold mixture of 0.2 M potassium phosphate buffer (PB), pH 7.6, containing 0.1% thioglycollic acid, 1 mM sodium diethyldithiocarbamate, and one volume of a 1:1 (v/v) chloroform : carbon tetrachloride mixture. Centrifuge the emulsion for 5 min at 8300 g. Filter the supernatant fluid through glass wool and add polyethylene glycol (M. Wt 6000) to 10% (w/v). Stir the mixture for 2 h at 5°C and centrifuge for 20 min at 9500 g. Resuspend the pellet overnight at 5°C in 0.1 M PB, pH 7.6. Clarify the suspension by centrifugation for 10 min at 12,000 g, and centrifuge the supernatant fluid for 2 h at 103,000 g. Allow the pellet to resuspend overnight in 0.01 M PB, pH 7.6, then clarify the suspension by centrifugation for 10 min at 12,000 g. Subject the virus to equilibrium centrifugation (90,000 g for 17 h) in 30% (w/w) CsCl or Cs2SO4 prepared in 0.01 M PB, pH 7.6. Collect the virus bands and dilute two-fold with 0.01 M PB, pH 7.6, and concentrate by differential centrifugation as before. The purified preparations are not infective.
Properties of Particles
Sedimentation coefficient s°20,w: 63-97 S depending on the conformation (filamentous, circular, aggregates) adopted by purified virus particles (F. J. Morales & A. I. Niessen, unpublished data).
Isoelectric point: about pH 4.5.
Migration velocity: -3.5 x 10-5 cm2/volt. sec on cellulose acetate in 0.01 M sodium phosphate buffer, pH 7.0. Electrophoretic mobility in the same test, relative to tobacco mosaic virus: 0.37.
A260/A280: 1.4 corrected for light-scattering.
Buoyant density in CsCl: 1.288 g/cm3.
Particle Structure
The virus particles are filaments, c. 3 nm in diameter. In sodium phosphotungstate, pH 6.5, they appear c. 8 nm in diameter because of supercoiling and are of no definite length (Fig. 3, Fig. 4) (Morales & Niessen, 1983); some particles adopt a helical configuration and may aggregate side by side (Fig. 4). In 1% uranyl acetate, pH 4.6, the particles have a loose open appearance which is difficult to interpret (Fig. 5).
Particle Composition
Nucleic acid: RNA, approximately 12% of the particle weight as determined by the A260/A280 value of purified virus preparations.
Protein: One species, of M. Wt 3.4 x 104, determined by electrophoresis in 7.5% polyacrylamide/SDS gels (Fig. 2) (Morales & Niessen, 1983).
Relations with Cells and Tissues
Virus aggregates have been observed in cells of the epidermis, palisade and spongy parenchyma, phloem, and vessels of leaves showing hoja blanca symptoms, both in the nucleus and in the cytoplasm (Fig. 6) (Shikata & Gálvez, 1969; A. I. Niessen & F. J. Morales, unpublished data). Virus particles have also been seen in the lumen and epithelial cells of the intestine of vector insects (Shikata & Gálvez, 1969).
Notes
Hoja blanca is the only virus disease of rice recognized in the American Continent. Rice hoja blanca virus can be distinguished from other rice viruses by differences in particle morphology, antigenic and vector specificity, and/or symptomatology. Rice stripe virus, which induces similar striping symptoms, causes malformation in the newly developed leaves whereas the new leaves of hoja blanca-affected plants unfold normally (Ling, 1972).
Figures

Polyacrylamide gel electrophoresis of sodium dodecyl sulphate-treated, partially purified preparations from infected rice roots (lane 1), leaves (lane 2), and frozen leaves (lane 3) showing adverse effect of freezing on virus isolation. Lane 5 contains purified virus prepared from infected rice leaves; notice the absence of the main host protein contaminant (probably fraction 1 protein) present in lane 2. Molecular weight markers are: (lane 4) b-lactoglobulin (18,000); (lane 6) bovine serum albumin (67,000); and (lane 7) bean southern mosaic virus particle protein (30,000).
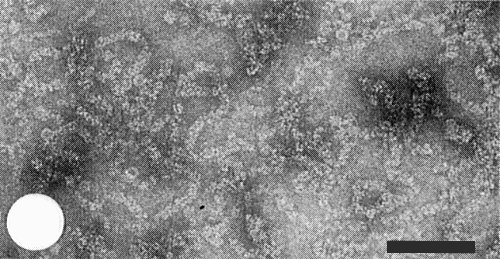
Virus-like particles observed in purified virus preparations stained in 1% uranyl acetate, pH 4.6. Bar represents 100 nm.
References list for DPV: Rice hoja blanca virus (299)
- Acuña, Ramos & Lopez, Agrotecnia 13: 23, 1958.
- Anon., A. Rep. CIAT Rice Program, 1983: 78, 1984a.
- Anon., A. Rep. CIAT Rice Program, 1983: 116, 1984b.
- Atkins & Adair, Pl. Dis. Reptr 41: 911, 1957.
- Everett & Lamey, in Viruses, Vectors and Vegetation, p. 361, ed. K. Maramorosch, New York: Interscience, 666 pp., 1969.
- Gálvez, Agronomia trop. 23: 384, 1967.
- Gálvez, Phytopathology 58: 818, 1968.
- Gálvez & Jennings, Agronomia trop. 15: 507, 1959.
- Gálvez, Jennings & Thurston, Pl. Dis. Reptr 44: 80, 1960.
- Gálvez, Thurston & Jennings, Pl. Dis. Reptr 45: 334, 1961.
- Garcés, Projects in Plant Pathology at the Palmira Experimental Station (unnumbered report to the Ministry of Economy), Bogotá, 1940.
- Gibler, Jennings & Krull, Pl. Dis. Reptr 45: 334, 1961.
- Gingery, Nault & Bradfute, Virology 112: 99, 1981.
- Koganezawa, Doi & Yora, Ann. phytopath. Soc. Japan 41: 148, 1975.
- Lamey, in The Virus Diseases of the Rice Plant, p. 293, Baltimore: Johns Hopkins, 354 pp., 1967.
- Lamey & Everett, Phytopathology 57: 227, 1967.
- Ling, Rice Virus Diseases, p. 142, International Rice Research Institute, Los Baños, Philippines, 142 pp., 1972.
- Malaguti, Diaz & Angeles, Agronomia trop. 6: 157, 1957.
- McGuire, McMillian & Lamey, Rice J. 63: 15, 1960.
- McGuire, McMillian & Lamey, Int. Rice Yb. 1961: 6, 1961.
- McMillian, McGuire & Lamey, J. econ. Ent. 55: 796, 1962.
- Morales & Niessen, Phytopathology 73: 971, 1983.
- Purcifull & Batchelor, Tech. Bull. agric. Exp. Stn. Univ. Fla 788: 39 pp., 1977.
- Shikata & Gálvez, Virology 39: 635, 1969.
- Van Hoof, Tijdschr PlZiekt. 65: 188, 1959.




