Details of DPV and References
DPV NO: 30 October 1970
Family: Betaflexiviridae
Genus: Trichovirus
Species: Apple chlorotic leaf spot virus | Acronym: ACLSV
There is a more recent description of this virus: DPV 386
Apple chlorotic leafspot virus
R. M. Lister Purdue University, Lafayette, Indiana, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
-
Described by
Cadman (1963),
Cropley (1963,
1964) and
Lister, Bancroft & Nadakavukaren (1965).
For disease description see Luckwill & Campbell (1963).
Selected synonyms
- Pear ring pattern mosaic virus (Rev. appl. Mycol. 35: 831)
- Apple latent virus Type 1 (Rev. appl. Mycol. 45: 134)
-
An RNA-containing virus with very flexuous filamentous particles c. 600 x 12 nm. Found in woody rosaceous plants, including apple, pear, peach, plum, cherry and apricot, from which it is sap-transmissible to a limited range of herbaceous species. Natural mode of spread unknown; widespread and common in vegetatively propagated commercial fruit tree clones, especially of apple, in which it often causes no symptoms.
Main Diseases
Causes a range of disease syndromes in some natural woody hosts, including chlorotic leaf spot of Russian apple, line pattern of Malus platycarpa, ring pattern mosaic of pear, dark green mottle of peach, and unnamed diseases of quince and apricot. See Cropley (1969) for an extensive review. The virus causes no symptoms in commercial apples.
Geographical Distribution
Reported from many countries. Probably occurs wherever apples are cultivated.
Host Range and Symptomatology
Outside the Rosaceae, the host range seems rather restricted (about 15 species in 8 dicotyledonous families - Lister et al., 1965; Nemec, 1967; Cropley, 1968; Saksena & Mink, 1969b). Grafting is used for transmission between woody hosts. The virus is transmitted from woody to herbaceous hosts by inoculating extracts from buds, young leaves, petals or fruits ground in a neutral buffer at around 0.05 M. Additives such as 2% (v/v) nicotine base, or 2% (w/v) polyvinyl pyrrolidone help but are not essential. Apple petals are a better source of virus than leaves (Lister et al., 1965) and fruits may be also (Cropley, 1968).
-
Diagnostic species
- Malus sylvestris
cv. R12740-7A (Russian apple). Chlorotic blotches, usually distributed asymmetrically in young leaves: asymmetric leaf distortion and puckering (Fig. 1): stunting of shoot growth. - Malus platycarpa (Long Ashton clone). Irregular diffuse chlorotic ring
and line patterns in leaves, which are smaller than normal, distorted, and often
shed prematurely. Strains differ in virulence.
- For symptoms in other species and horticultural varieties of Malus and woody Rosaceae, see Luckwill & Campbell (1963) and Cropley (1969).
- Chenopodium quinoa. After 3-6 days, large (2 mm diam.) etched primary lesions, becoming pale brown and necrotic (Fig. 2). Later, systemic chlorotic spotting and mottling, with ring and line patterns, culminating in more general chlorosis with necrosis (Fig. 3).
- C. amaranticolor. Symptoms are similar to those in C. quinoa, but primary lesions tend to be compound, consisting of chlorotic spots surrounding several necrotic dots.
- Phaseolus vulgaris cvs. Pinto, Bountiful, Kinghorn (bean). Purple-brown necrotic spots or rings 1-3 mm in diameter. Not systemic.
- Virus transmissibility from C. quinoa to either C. quinoa or P. vulgaris is improved by using clarified C. quinoa sap or partially purified virus preparations (Saksena & Mink, 1969a).
- For symptoms in other species and horticultural varieties of Malus and woody Rosaceae, see Luckwill & Campbell (1963) and Cropley (1969).
-
Propagation species
- C. quinoa
is suitable for maintaining cultures and propagating virus for purification.Assay species
- C. quinoa
and P. vulgaris (see above).
Strains
Only a few isolates have been identified unequivocally by serology, symptomatology in woody plants, and other properties. Isolates differ in virulence towards herbaceous hosts, but not in their serological or physical properties. Variants have been distinguished by symptomatology in woody hosts (Cropley, 1969).
Transmission by Vectors
Although spread in the field has been detected, the natural mode of spread is unknown. A report of transmission by Eudorylamoid nematodes (Fritzsche & Kegler, 1968) requires confirmation. The suggestion that spread may occur in pollination (Cadman, 1965; Lister & Cadman, 1965), though not disproved, was based on an erroneous identification of this virus with raspberry bushy dwarf virus (see Relationships).
Transmission through Seed
None reported.
Transmission by Dodder
Not reported.
Serology
Moderately antigenic, giving titres up to 1/512. Ring-precipitin tests can be used with sufficiently purified virus, including virus concentrated from apple petals (Saksena & Mink, 1969c). Latex flocculation tests give specific positive results with crude buffered extracts from infected C. quinoa leaves or apple petals, but not apple leaves or fruits; and gel-diffusion tests work with virus fragments or virus protein, but not with intact virus (Lister & Hadidi, unpublished).
Relationships
No serological differences have been found between strains. The virus seems unrelated in serological and plant protection tests to the morphologically similar apple stem grooving virus, but its behaviour in plant protection tests may be anomalous (Van der Meer, 1968). A suspected relationship to raspberry bushy dwarf virus (Cadman, 1965) is now disproved (Barnett & Murant, 1970).
Stability in Sap
Much less stable than apple stem grooving virus in plant extracts. In bentonite-clarified C. quinoa sap (see Purification), thermal inactivation point (10 min) is 52-55°C, half-life at 45°C is 12±1 min and dilution end-point is c. 10-4; infectivity is abolished in 1 day at 20°C and in c. 10 days at 4°C. Infectivity is inhibited by crude C. quinoa sap (Saksena & Mink, 1969a); the inhibitor may be a ribonuclease (Lister & Hadidi, unpublished).
Purification
The most satisfactory method depends on clarifying extracts with bentonite (Cadman, 1963; Lister et al., 1965). Lister & Hadidi (unpublished) obtained no improvement using magnesium bentonite (recommended by Saksena & Mink, 1969b): they developed the following method, based on that of de Sequeira & Lister (1969):
Blend each 100 g infected C. quinoa leaf in 200 ml 0.01 M Tris-HCl buffer (pH 7.6), containing 0.01 M MgSO4. Squeeze through cheesecloth and then thoroughly clarify by cautiously adding bentonite suspension (c. 40 mg/ml in 0.01 M phosphate buffer at pH 7-8). Centrifuge at low speed. Precipitate the virus from the supernatant fluid by adding polyethylene glycol, M. Wt 6000, to 8% (w/v), and allow to stand for 1 hr. Centrifuge at low speed and resuspend virus pellets in Tris-HCl buffer. Concentrate by ultracentrifugation. Purify the virus further by density gradient centrifugation. Do all steps at c. 4°C.
Yields are 1-2 mg/100 g of leaf, depending on the strain.
Properties of Particles
Sedimentation coefficient (s20,w): about 96 S (not corrected to infinite dilution). No accessory virus particles detected.
Electrophoretic mobility: -5.1 x 10-5 cm2 sec-1 volt-1 in 0.05 M Tris/0.005 M MgCl2 buffered to pH 7.5 in 3% sucrose (determined by electrophoresis in a sucrose gradient).
A260/A280: 1.18, according to Saksena & Mink (1969b); however, Lister & Hadidi (unpublished) could not confirm this but obtained values, for very pure preparations, of 1.85 falling to 1.55 on dialysis, which can cause particle breakage.
Particle Structure
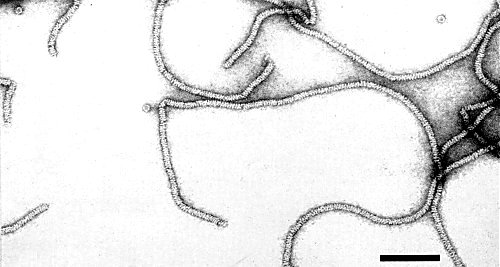
A very flexuous filament with obvious cross-banding and helical symmetry (Fig. 4). Length c. 600 nm; width c. 12 nm; pitch of helix c. 3.8 nm. Particle fragments viewed ‘end-on’ show a hollow centre, but this is not visible laterally. Best stains for electron microscopy are uranyl acetate or uranyl formate; breakage occurs in phosphotungstate (Lister et al., 1965, and unpublished).
In the absence of certain divalent cations or polyamines, particles degrade, forming fragments sedimenting at about 17 S (Lister & Hadidi, unpublished).
Particle Composition
Contains RNA (Lister & Hadidi, unpublished).
Relations with Cells and Tissues
Unknown: has been eliminated from some apple varieties and rootstocks by heat therapy (37°C for up to 3 weeks).
Notes
Cropley (1964) and Lister, Bancroft & Shay (1964) reported isolates of apple chlorotic leaf spot virus that specifically caused a chlorotic leaf spot disease in Russian apple R12740-7A. These and some other isolates have been shown, by detailed serological and other tests, to be related. However, for many assumed isolates, critical comparisons have not been made. Because strains differ in the symptoms they cause in both woody and herbaceous indicators, serological and/or thorough property testing is essential for accurate diagnosis.
Acknowledgements
Photographs: courtesy of Purdue Agricultural Experiment Station.
Figures
References list for DPV: Apple chlorotic leafspot virus (30)
- Barnett & Murant, Ann. appl. Biol. 65: 435, 1970.
- Cadman, Pl. Dis. Reptr 47: 459, 1963.
- Cadman, Pl. Dis. Reptr 49: 230, 1965.
- Cropley, Pl. Dis. Reptr 47: 165, 1963.
- Cropley, Pl. Dis. Reptr 48: 678, 1964.
- Cropley, Ann. appl. Biol. 61: 361, 1968.
- Cropley, In Tech. Commun. Commonw. Bur. Hort. Plantn Crops 30, Suppl.2/3/4: 10, 1969.
- de Sequeira & Lister, Phytopathology 59: 1740, 1969.
- Fritzsche & Kegler, TagBer. dt. Akad. Landw Wiss. Berl. 97: 289, 1968.
- Lister, Bancroft & Shay, Phytopathology 54: 1300, 1964.
- Lister, Bancroft & Nadakavukaren, Phytopathology 55: 859, 1965.
- Lister & Cadman, Zast. Bilja 16: 233, 1965.
- Luckwill & Campbell, In Tech. Commun. Commonw. Bur. Hort. Plantn Crops 30: 5, 1963.
- Nemec, Pl. Dis. Reptr 51: 283, 1967.
- Saksena & Mink, Phytopathology 59: 61, 1969a.
- Saksena & Mink, Phytopathology 59: 84, 1969b.
- Saksena & Mink, Phytopathology 59: 877, 1969c.
- Van der Meer, TagBer. dt. Akad. Landw Wiss. Berl. 97: 27, 1968.