Details of DPV and References
DPV NO: 303 September 1985
Family: Geminiviridae
Genus: Begomovirus
Species: Tomato golden mosaic virus | Acronym: TGMV
Tomato golden mosaic virus
K. W. Buck Department of Pure and Applied Biology, Imperial College of Science and Technology, London SW7 2BB, UK
R. H. A. Coutts Department of Pure and Applied Biology, Imperial College of Science and Technology, London SW7 2BB, UK
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
- Disease first transmitted by Costa et al. (1975). Virus first purified
by Matyis et al. (1975) and characterized by Hamilton et al. (1981).
- Synonym
- Virus do mosaico dourado do tomateiro (Rev. Pl. Path. 55: 2599)
- A virus with geminate particles c. 25 x 13 nm containing two species of circular single-stranded DNA. The known hosts are in the Solanaceae and the virus is transmitted by the whitefly Bemisia tabaci. Occurs widely in tropical areas of Brazil (Costa, 1976).
Main Diseases
Causes a type of golden mosaic of tomato (Lycopersicon esculentum) of minor economic importance (Costa, 1976; A. S. Costa, personal communication).
Geographical Distribution
Reported only from Brazil. However, similar whitefly-transmitted diseases of tomato, which are probably caused by geminiviruses, have been reported from Venezuela, Africa and Asia.
Host Range and Symptomatology
Known hosts are all species of Solanaceae and include Datura stramonium, Nicotiana benthamiana, N. clevelandii, N. debneyi, N. glutinosa, N. tabacum cvs Samsun NN, virginica, White Burley ‘Judy’s Pride’ and Xanthi-nc, and Physalis sp., which can all be infected mechanically with high efficiency (Costa et al., 1975; Hamilton et al., 1981; K. W. Buck, R. H. A. Coutts & M. Slomka, unpublished data). Mechanical transmission of the virus from tomato (Lycopersicon esculentum) to tomato has been achieved with extreme difficulty, only 1.6% of inoculated plants becoming infected. Solanum pennelli and derivatives of crosses between L. esculentum and S. pennelli were infected mechanically with relative ease (Costa et al., 1975) but potato (S. tuberosum ssp. tuberosum cv. Maris Bard) and S. nigrum were not infected either mechanically or by grafting (K. W. Buck, R. H. A. Coutts & E. J. F. Roberts, unpublished data). The symptoms are similar on all susceptible species and cultivars, consisting of stunting with severe deformation of young leaves and shoots, accompanied by a bright yellow mosaic. However, whereas younger plants routinely give these symptoms (Costa, 1976; Hamilton et al., 1981), older plants may show a partial ‘recovery’ producing apparently healthy leaves which may later develop sporadic symptoms (K. W. Buck & R. H. A. Coutts, unpublished data).
- Diagnostic species
- Nicotiana benthamiana.
Faint chlorotic mottling on inoculated leaves followed by curling and spiral distortion of systemically infected young leaves and shoots, accompanied by a bright golden yellow mosaic (Fig. 1) and stunting (Hamilton et al., 1981). - Datura stramonium. Yellow spotting on inoculated leaves followed by
a bright golden yellow mosaic on systemically infected leaves (Fig. 2)
particularly around veinal areas, together with some stunting (K. W. Buck,
R. H. A. Coutts & C. J. Knight, unpublished data).
- Propagation species
- Nicotiana benthamiana.
Plants with six to eight leaves are suitable for mechanical inoculation of the expanding upper leaves with virus, viral ssDNA or dsDNA, or cloned viral dsDNA. The most reproducible results are obtained when plants are kept in a controlled environment (18-20°C, 75% relative humidity, 16 h photoperiod, 15,000 lx light intensity).- Assay species
- The only assay is by recording the proportion of Nicotiana benthamiana plants that become systemically infected. Costa (1976) reported that the virus produced yellow local lesions on young leaves of N. glutinosa, but this was not confirmed in other tests (K. W. Buck & R. H. A. Coutts, unpublished data).
Strains
No information.
Transmission by Vectors
Transmitted by the whitefly Bemisia tabaci from tomato to tomato or to susceptible Nicotiana spp. (Costa, 1976) but data on acquisition and latent periods are lacking. However the sap-transmissible virus that causes tomato yellow mosaic in Venezuela, which is possibly distantly related to tomato golden mosaic virus (E. J. F. Roberts, R. H. A. Coutts & K. W. Buck, unpublished data), can be acquired by B. tabaci in 2 h, has a latent period of 20 h and can be transmitted by the insects for up to 7 days after they are removed from virus source plants. Female B. tabaci transmitted more frequently than males, and transmission was more frequent at 30-34°C than at 20-30°C (De Uzcategui & Lastra, 1978).
Transmission through Seed
No information.
Serology
The virus is moderately immunogenic. An antiserum prepared in a rabbit by one intravenous and two intramuscular injections of 300 µg of purified virus had a titre of 1/256 in immunodiffusion tests and gave a single precipitin line (Stein et al., 1983).
Relationships
In gel double diffusion serological tests the virus was found to be moderately
closely related to another whitefly-transmitted geminivirus, African cassava
mosaic virus (synonym cassava latent virus) (Fig. 4) and distantly related to a
leafhopper-transmitted geminivirus, beet curly top virus, but no relationships
were found to four other leafhopper-transmitted geminiviruses: chloris striate
mosaic, maize streak, tobacco yellow dwarf and wheat dwarf viruses (Stein et
al., 1983). Similar results were obtained in immunosorbent electron
microscopy by Roberts et al. (1984) who extended the list of serologically
related whitefly-transmitted geminiviruses to include bean golden mosaic, euphorbia
mosaic and squash leaf curl viruses, but could not confirm a
relationship to beet
curly top virus.
In solid phase hybridization tests under high stringency conditions, tomato golden mosaic virus DNA was found to have sequence homology with euphorbia mosaic virus (Puerto Rican isolate kindly supplied by Dr J. Bird) (Fig. 3) and several isolates of African cassava mosaic virus (e.g. Adejare & Coutts, 1982) but not with maize streak virus or wheat dwarf virus (K. W. Buck, R. H. A. Coutts, S. W. MacDowell & G. Sunter, unpublished data); a probe for the larger DNA of African cassava mosaic virus reacted with tomato golden mosaic virus DNA whereas that for the smaller DNA of African cassava mosaic virus did not (Roberts et al., 1984). Nucleotide sequence comparisons revealed 60% homology between the larger DNA of tomato golden mosaic virus and the larger DNA of African cassava mosaic virus (Kenyan type strain) and 40% homology between the smaller DNA molecules of these two viruses; comparison of the amino acid sequences (predicted from the DNA sequences) of the coat proteins of the same two viruses revealed 71% direct homology and 85% conserved homology (Hamilton et al., 1984). Comparative hydrophilicity analyses are shown in Fig. 6.
Stability in Sap
The virus occurs in low concentration in diseased tissue and is not resistant to heat or ageing in vitro. The virus can be preserved for at least a year in N. benthamiana leaves after desiccation over calcium chloride, but infectivity is lost when aqueous extracts, which initially have an end-point of 10-3 on N. benthamiana, are stored at 4°C for 24 h (K. W. Buck, R. H. A. Coutts & C. J. Knight, unpublished data).
Purification
Purified virus particles (5 to 10 mg/kg fresh leaf) can be obtained by the following method (Stein et al., 1983). Harvest systemically infected N. benthamiana leaves 15-20 days after inoculation and homogenize in 0.1 M trisodium citrate, 0.75% sodium sulphite, 5 mM disodium EDTA, 1% 2-mercaptoethanol and 0.325% L-ascorbic acid, pH 7.0, adjusted with NaOH (2 ml buffer/g tissue). Add Triton X-100 to 2.5% by volume and stir for 16 h at 4°C before filtration through muslin. Centrifuge the filtrate at low speed and then centrifuge the supernatant fluid for 2 h at 40 000 rev/min in a Beckman Ti 45 rotor. Resuspend the virus pellets in 0.01 M trisodium citrate, 1 mM disodium EDTA with 0.05% 2-mercaptoethanol, adjust to pH 7 with NaOH, then centrifuge the virus through a ‘cushion’ of 20% (w/v) sucrose in the same buffer (2 h at 40,000 rev/min in a Beckman SW 50.1 rotor) and resuspend the pellets in the same buffer. Further purify the virus by centrifugation in gradients of 10 to 50% (w/v) sucrose for 16 h at 20,000 rev/min in a Beckman SW 28 rotor. The virus forms a sharp light-scattering zone three-quarters of the way down the gradient. Final traces of host proteins are removed by isopycnic centrifugation in self-generating density gradients of caesium sulphate (35,000 rev/min; 24 h; starting density 1.34 g/ml) (V. E. Stein, unpublished data). The infectivity of partially purified virus preparations (prior to the sucrose density gradient stage) is stable for at least 2 years when stored at -20°C.
Properties of Particles
A260/A280: 1.56.
Buoyant density in caesium sulphate: 1.34 g/cm3. The particles are unstable in caesium chloride.
Particle Structure
Negatively-stained particles are predominantly ‘geminate’, c. 25 x 13 nm (Fig. 5). Unpaired quasi-isometric particles are probably formed by disruption of the geminate ones. The particles are stable between pH 4.0 and 7.0, but are disrupted at pH 8.0 or above (Matyis et al., 1975).
Particle Composition
Nucleic acid: Two segments of circular single-stranded DNA, termed A and B (Hamilton et al., 1981; Bisaro et al., 1982). DNA A has 2588 nucleotides (23.4% G, 26.0% A, 19.8% C, 30.8% T) and has M. Wt (Na salt) of 0.84 x 106. DNA B has 2508 nucleotides (21.5% G, 29.0% A, 18.7% C, 30.8% T), and has M. Wt (Na salt) of 0.82 x 106 (Hamilton et al., 1984). Particles obtained from plants infected with the native Brazilian isolate (but not from those plants infected with cloned DNA) contain a subgenomic DNA, of M. Wt c. 0.4 x 106, derived from DNA B (Hamilton et al., 1982; K. W. Buck, R. H. A. Coutts, W. D. O. Hamilton, S. W. MacDowell & V. E. Stein, unpublished data).
Protein: Only one species of polypeptide detected in the particles, of M. Wt 28,500 (Hamilton et al., 1981; V. E. Stein, unpublished data).
Genome Properties
The ssDNA from purified virus particles and the circular dsDNA of genome
length isolated from infected plants are both infective by mechanical
inoculation. Cloned linear dsDNA is also infective and DNA segments A and B
are both required for infectivity (Hamilton et al., 1981, 1983).
Analysis of the nucleotide sequences of the cloned DNA molecules showed that
DNA A and DNA B have little sequence homology except for a region of about 200
bases which is almost identical in the two molecules. Within this ‘common’
region there is a 33-base sequence capable of forming a stable hairpin structure.
Analysis of open reading frames (ORFs) revealed six potential genes for proteins
comprised of more than 100 amino acids, four in DNA A and two in DNA B. Some of
the ORFs are in the DNA strands that occur in the virus particles and some are
in the complementary strands; this suggests that transcription is bidirectional
(Fig. 7). The coat protein gene lies on DNA A and is encoded by the strand that
occurs in the virus particles (Hamilton et al., 1984 and unpublished
data).
In addition to the genomic and subgenomic ssDNA species found in the virus particles, several other virus-specific DNA species have been isolated from infected plants (Hamilton et al., 1982). These include supercoiled closed circular dsDNA of unit length with a superhelical density of -0.062 (Sunter et al., 1984), open circular and linear dsDNA species of unit length, and multimeric dsDNA species of two or more unit lengths.
Relations with Cells and Tissues
Isolated nuclei from infected cells contain all of the virus-specific DNA forms detected in whole plant extracts and are active in virus DNA replication and transcription (R. H. A. Coutts, unpublished data).
Notes
The virus may be distinguished from three other whitefly-borne geminiviruses, tobacco leaf curl virus (Osaki & Inouye, 1981) and tomato yellow leaf curl virus (Russo et al., 1980) which do not cause mosaic symptoms on infected plants and are apparently not sap-transmissible, and bean golden mosaic virus whose host range is apparently limited to certain wild and cultivated species of Leguminosae (Goodman & Bird, 1978). Isolates of tomato yellow mosaic virus from Venezuela (De Uzcategui & Lastra, 1978) and India (Verma et al., 1975) could be strains of tomato golden mosaic virus although only the former has been shown to be sap-transmissible and little comparative information is available. The limited sequence comparisons among geminiviruses so far done (Hamilton et al., 1984; Roberts et al., 1984) suggest that the degree of sequence homology of the 200-base ‘common’ regions, or of the smaller genome segment, could be useful in determining which geminiviruses are separate viruses and which are strains of existing viruses. For viruses for which cloned DNA components are available the ability or inability to form viable pseudo-recombinants is a useful criterion. Viable pseudo-recombinants were not formed when Nicotiana benthamiana plants were inoculated with a mixture of tomato golden mosaic virus DNA A and African cassava mosaic virus DNA 2 or tomato golden mosaic virus DNA B and African cassava mosaic virus DNA 1 (K. W. Buck, R. H. A. Coutts, I. T. D. Petty & J. Stanley, unpublished data). Hence these two viruses are distinct.
Figures

Autoradiograph of blot-hybridizations of crude DNA extracts from (A) Euphorbia prunifolia leaves infected with euphorbia mosaic virus, (B) N. benthamiana leaves infected with tomato golden mosaic virus, to a 32P-labelled probe of a nick-translated mixture of the cloned components of tomato golden mosaic virus DNA. Bands marked 1, 2 and 3 in (B) represent the open circular, linear and supercoiled forms of tomato golden mosaic virus dsDNA band 4 is the circular ssDNA that occurs in the virus particles (Hamilton et al., 1982).

Gel immunodiffusion analysis. Wells contain: top, antiserum to tomato golden mosaic virus; bottom left, African cassava mosaic virus (Kenyan type strain); bottom right, tomato golden mosaic virus. Reproduced from Stein et al. (1983) with permission.

Electron micrograph of virus negatively stained with 1% sodium phosphotungstate, pH 5.0. Bar represents 100 nm. Courtesy of J. C. Matyis, D. M. Silva, A. R. Oliveira and A. S. Costa.

Hydrophilicity analysis of the predicted amino acid sequences of the coat protein genes of (above) tomato golden mosaic virus, (below) African cassava mosaic virus (Kenyan type strain). Reproduced from Hamilton et al. (1984) with permission.
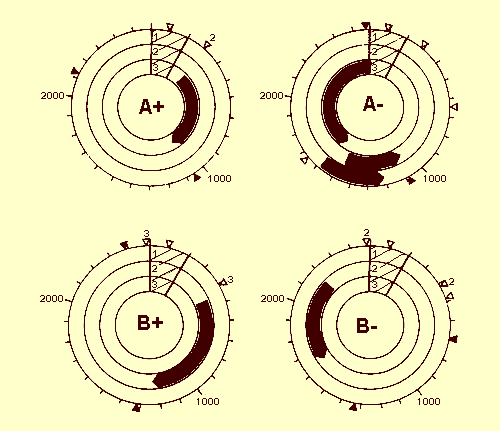
Potential coding regions in TGMV DNA A and DNA B. All ORFs starting with an ATG triplet and coding for proteins with M. Wt > 10,000 are shown as thick black arrows and the common 200-base region is shaded. Solid triangles indicate the position of AATAAA sequences and open triangles TATA boxes. More than one TATA box in the same region is indicated by a number adjacent to the open triangle. A scale is marked around the outside of each circle at 100-base intervals. The 1-kb and 2-kb positions are numbered. Reproduced from Hamilton et al. (1984) with permission.
References list for DPV: Tomato golden mosaic virus (303)
- Adejare & Coutts, Phytopath. Z. 103: 198, 1982.
- Bisaro, Hamilton, Coutts & Buck, Nucleic Acids Res. 10: 4913, 1982.
- Costa, A. Rev. Phytopath. 14: 429, 1976.
- Costa, Oliveira & Silva, 8th Ann. Congr. Soc. Bras. Fitopathol. Mossoro, Brazil (Abstr.) 1975.
- De Uzcategui & Lastra, Phytopathology 68: 985, 1978.
- Goodman & Bird, CMI/AAB Descr. Pl. Viruses 192: 4 pp., 1978.
- Hamilton, Sanders, Coutts & Buck, FEMS Microbiol. Lett. 11: 263, 1981.
- Hamilton, Bisaro & Buck, Nucleic Acids Res. 10: 4901, 1982.
- Hamilton, Bisaro, Coutts & Buck, Nucleic Acids Res. 11: 7387, 1983.
- Hamilton, Stein, Coutts & Buck, EMBO J. 3: 2197, 1984.
- Matyis, Silva, Oliveira & Costa, Summa Phytopath. 1: 267, 1975.
- Osaki & Inouye, CMI/AAB Descr. Pl. Viruses 232: 4 pp., 1981.
- Roberts, Robinson & Harrison, J. gen. Virol. 65: 1723, 1984.
- Russo, Cohen & Martelli, J. gen. Virol. 49: 209, 1980.
- Stein, Coutts & Buck, J. gen. Virol. 64: 2493, 1983.
- Sunter, Coutts & Buck, Biochem. Biophys. Res. Comm. 118: 747, 1984.
- Verma, Srivastava & Mathur, Pl. Dis. Reptr 59: 494, 1975.

