Details of DPV and References
DPV NO: 305 September 1985
Family: Potyviridae
Genus: Potyvirus
Species: Bean common mosaic virus | Acronym: BCMV
This virus is now regarded as a distinct strain of bean common mosaic virus
Blackeye cowpea mosaic virus
D. Purcifull Department of Plant Pathology, University of Florida, Gainesville, Florida 32611, USA
D. Gonsalves Department of Plant Pathology, Cornell University, New York State Agricultural Experiment Station, Geneva, New York 14456, USA
Contents
- Introduction
- Main Diseases
- Geographical Distribution
- Host Range and Symptomatology
- Strains
- Transmission by Vectors
- Transmission through Seed
- Transmission by Grafting
- Transmission by Dodder
- Serology
- Nucleic Acid Hybridization
- Relationships
- Stability in Sap
- Purification
- Properties of Particles
- Particle Structure
- Particle Composition
- Properties of Infective Nucleic Acid
- Molecular Structure
- Genome Properties
- Satellite
- Relations with Cells and Tissues
- Ecology and Control
- Notes
- Acknowledgements
- Figures
- References
Introduction
Described by Anderson (1955) and Lima et al. (1979). Some isolates (Taiwo et al., 1982) were previously designated as cowpea aphid-borne mosaic virus.
- Selected synonyms
- Bean yellow mosaic virus, cowpea strain (J. gen. Virol. 15: 113, 1972)
- Cowpea aphid-borne mosaic virus, Nigeria and Kenya isolates (Rev. Pl. Path. 61: 7256)
A virus with flexuous filamentous particles c. 750 nm long, containing single-stranded RNA. It induces the formation of cytoplasmic cylindrical (pinwheel) inclusions and associated scrolls in its hosts. It is transmissible mechanically, by several aphid species in a non-persistent manner, and through the seed of cowpea. It causes a mosaic disease of cowpea and occurs in various places throughout the world.
Main Diseases
Causes mosaic diseases of cowpea (Vigna unguiculata subsp. unguiculata), asparagus bean (V. unguiculata subsp. sesquipedalis) and Crotalaria spectabilis. In cowpea, symptoms and susceptibility depend on the cultivar and on the virus strain. Some cultivars of cowpea are immune (Lima et al., 1979; Taiwo et al., 1982). Dual infections with cucumber mosaic virus result in severe stunting of cowpea (Pio-Ribeiro et al., 1978) and rugose mosaic of asparagus bean (Chang, 1983).
Geographical Distribution
Fairly wide. Reported in Africa (Bock, 1973; Taiwo et al., 1982), Brazil (Lin et al., 1981), India (Mali & Kulthe, 1980; Sekar & Sulochana, 1983), Japan (Taiwo et al., 1982), Taiwan (Chang, 1983), Thailand and Malaysia (Tsuchizaki et al., 1984) and the USA (Anderson, 1955; Zettler & Evans, 1972; Lima et al., 1979).
Host Range and Symptomatology
The virus is readily transmissible by inoculation with sap. At least 36 species in 7
dicotyledonous families are susceptible, with cowpea being a major natural host. Natural
infections are also reported in Crotalaria, asparagus bean and Desmodium.
Diagnostic species
Necrotic lesions in inoculated leaves (Fig. 1), with some
isolates (Kuhn, 1964;
Pio-Ribeiro et al., 1978).
Propagation species
Assay species
Strains
Symptom and host range variants have been reported (Bock, 1973; Bock & Conti, 1974; Taiwo et al., 1982; Murphy, 1984; Murphy et al., 1984). A major symptom variant is an isolate which causes red, necrotic ringspots and reddish veinal necrosis on cowpea cv. Knuckle Purple Hull (Murphy, 1984). Some other differences in host range between this isolate and an isolate of similar origin to that of Lima et al. (1979) also were reported. Isolates from different parts of the world are closely related serologically (Taiwo & Gonsalves, 1982; Chang, 1983; Murphy, 1984).
Transmission by Vectors
Transmitted in a non-persistent manner by Aphis craccivora and Myzus persicae (Zettler et al., 1967; Murphy, 1984). The virus was transmitted by as many as 59% of individual A. craccivora following acquisition probes of less than 1 min (Zettler et al., 1967). Individuals of M. persicae were able to acquire and transmit both blackeye cowpea mosaic virus and cucumber mosaic virus from doubly-infected asparagus bean plants (Chang, 1983).
Transmission through Seed
Transmitted through cowpea seed (Zettler & Evans, 1972; Bock, 1973; Lima & Purcifull, 1980), with frequencies as high as 30.9% (Zettler & Evans, 1972).
Serology
Virus particles (Bock, 1973; Lima et al., 1979; Taiwo & Gonsalves, 1982) and pyrollidine-dissociated virus (Lima et al., 1979) are good immunogens. Serological relationships have been studied by liquid precipitin tests (Corbett, 1956; Bock 1973), by sodium dodecyl sulphate (SDS)-immunodiffusion tests (Fig. 6, Fig. 7; Lima et al., 1979; Taiwo & Gonsalves, 1982), by immunodiffusion tests with ethanolamine-treated (Uyemoto et al., 1973) and pyrollidine-treated (Shepard et al., 1974) virus, by electron microscopic antibody coating tests (Taiwo & Gonsalves, 1982), and by enzyme-linked immunosorbent assays (ELISA) (Taiwo & Gonsalves, 1982; Murphy, 1984). Buffers containing 0.1 M potassium phosphate, pH 7.5, and 0.1 M ethylenediamine-tetraacetate enhance sensitivity of ELISA tests (Taiwo & Gonsalves, 1982). The virus was detected in hypocotyls of germinated cowpea seeds by single radial and double radial SDS-immunodiffusion tests and by immunosorbent electron microscopy (Lima & Purcifull, 1980). Intragel absorption tests have been useful for studying relationships (Lima, 1978; Lima et al., 1979). Antiserum to cylindrical inclusions also has been prepared and was used to detect inclusions in virus-infected hypocotyls by SDS-immunodiffusion (Lima et al., 1979; Lima & Purcifull, 1980). The cylindrical inclusions are antigenically distinct from the virus particles.
Relationships
Blackeye cowpea mosaic virus is considered a separate member of the potyvirus group
(Hollings & Brunt, 1981;
Matthews, 1982). However, it is serologically related to numerous other potyviruses (Shepard et al., 1974;
Lima et al., 1979), a phenomenon that is
common within the group. Because of this, blackeye cowpea mosaic virus was once considered
to be a cowpea-infecting strain of bean yellow mosaic virus
(Corbett, 1956;
Kuhn et al., 1966) but cytological, host range and serological tests
(Edwardson et al., 1972;
Zettler & Evans, 1972;
Lima et al., 1979) show that these two viruses should be
regarded as different. Blackeye cowpea mosaic virus is also closely serologically related
to bean common mosaic and peanut stripe viruses but it can be
distinguished from them by double diffusion tests in SDS gels
(Lima et al., 1979;
Taiwo & Gonsalves, 1982;
Demski et al., 1984; D. Purcifull, unpublished data).
There has also been considerable confusion over the relationship between blackeye cowpea
mosaic virus and cowpea aphid-borne mosaic virus. The type culture of cowpea aphid-borne
mosaic virus
(Lovisolo & Conti, 1966) has been lost and some isolates previously
assigned to that virus now seem to be strains of blackeye cowpea mosaic virus
(Taiwo et al., 1982;
Taiwo & Gonsalves, 1982). However, a virus from cowpea in Morocco has
been referred to by
Fisher & Lockhart (1976) as cowpea aphid-borne mosaic virus. This
Moroccan virus and blackeye cowpea mosaic virus are serologically related, but not
closely (Fig. 6, Fig. 7), differ in host range and are regarded as separate potyviruses
(Lima et al., 1979;
Taiwo et al., 1982;
Taiwo & Gonsalves, 1982). Certain
cowpea cultivars (e.g., Serido) may be useful as differential hosts
(Lima et al., 1979;
Taiwo et al., 1982).
Based on the morphology of scrolls and pinwheels in infected tissues, Edwardson (1974) assigned blackeye cowpea mosaic virus to subdivision I of his potyvirus classification scheme.
Stability in Sap
The thermal inactivation point in sap from infected cowpea plants is 60-65°C, longevity in vitro is 1-2 days, and the dilution end-point is between 10-3 and 10-5 (Lima et al., 1979; Murphy, 1984).
Purification
The following method for purification of virus particles and cylindrical inclusions
from the same batch of tissue was devised by
Lima et al. (1979) and modified slightly
by
Taiwo et al. (1982). Homogenize 200-400 g systemically infected leaves of cowpea
or Nicotiana benthamiana in a mixer with 1.3 ml 0.5 M potassium phosphate buffer
(PB), pH 7.5, containing 0.5% Na2SO3, 0.3 ml chloroform, and 0.3 ml carbon
tetrachloride for each 1 g tissue. Centrifuge at 4000 g for 5 min, discard
pellet, and centrifuge supernatant fluid at 13,200 g for 15 min. Resuspend
the pellet, which contains the inclusions, in 10-20 ml 20 mM PB, pH 8.2, containing 0.1%
2-mercaptoethanol (2-ME) and purify further as described below. Precipitate the virus
from the supernatant fluid by adding polyethylene glycol (PEG, M. Wt 6000) to a
concentration of 8% (w/v). Stir for 1 h at 4°C, and centrifuge at 13,200
g for 10 min. Resuspend the precipitated virus in 20 mM PB, pH 8.2,
containing 0.1% 2-ME (PB-ME) and purify further by equilibrium centrifugation for 16-18
h at 150,000 g in 30% CsCl prepared in the same buffer. Remove the
virus-containing zone by droplet fractionation, dilute with 2 vol PB-ME, and centrifuge
at 12,000 g for 10 min. Adjust the supernatant fluid to 30% CsCl (w/w),
recentrifuge to equilibrium for 16-18 h at 150,000 g, and remove the virus
zone by droplet fractionation. Dilute the virus preparation with PB-ME, clarify by
centrifugation at 12,000 g for 10 min and re-concentrate the virus by
centrifugation at 85,000 g for 1.5 h. Resuspend the virus in 20 mM Tris
buffer, pH 8.2. Yields are up to 8 mg/100 g tissue.
Homogenize the fraction containing the cylindrical inclusions in a Sorvall Omnimixer for
2 min and add Triton X-100 to a final concentration of 5% (v/v). Stir for 1 h at 4°C,
centrifuge at 27,000 g for 15 min, resuspend the pellet containing the
inclusions in PB-ME, and homogenize for 30 sec. Re-pellet, resuspend and re-homogenize
the inclusion preparation. Layer the homogenate on sucrose step gradients containing 10
ml 80%, 7 ml 60% and 7 ml 50% (w/v) sucrose in 20 mM PB, pH 8.2. Centrifuge in a Beckman
SW 25.1 rotor for 1 h at 21,000 rev/min. Remove the inclusions, which layer on top of the
80% sucrose zone, dilute with 2 vol 20 mM PB, and concentrate by centrifugation at 27,000
g for 15 min. Resuspend in 20 mM Tris, pH 8.2. Yields are 5-20
A280 units per 100 g tissue. The inclusion proteins can be purified
further for use as immunogens by SDS-polyacrylamide gel electrophoresis following
dissociation of the inclusions, according to the methods of
Yeh & Gonsalves (1984)
and D. Gonsalves (unpublished data).
A method for purifying the virus only from infected tissue of cowpea or N. benthamiana also was described by Lima et al. (1979). It was similar to the procedure described above, with the following exceptions. Homogenize tissue in 0.5 M PB, pH 7.5, containing 0.5% Na2SO3 (2 ml buffer/g tissue). Filter through cheesecloth, add n-butanol to a final concentration of 8% (v/v), and stir overnight at 4°C. Centrifuge at 11,700 g for 10 min, precipitate the virus by adding PEG, centrifuge at 11,700 g for 10 min, and resuspend the pellet in PB-ME. Further purify by one cycle of equilibrium centrifugation in CsCl.
Properties of Particles
Sedimentation coefficient, s2O, w: 157-159 S (Lima et al., 1979).
A260/A280: 1.2.
Particle Structure
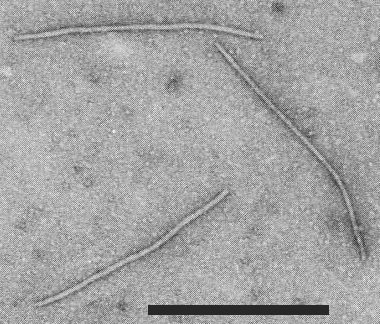
Particles are flexuous filaments (Fig. 8) with a modal length of 743-765 nm (Taiwo et al., 1982; Lima et al., 1979; Murphy, 1984).
Particle Composition
Nucleic acid: RNA, single-stranded, M. Wt 2.9 x 106 (Murphy, 1984). Sedimentation coefficient in sucrose gradients is c. 39-40 S (Taiwo et al., 1982). RNA is infective (Taiwo et al., 1982; Murphy, 1984).
Protein: Estimated to be 95% of particle weight, based on A260/A280 ratio. Subunit M. Wt 3.4-3.5 x 103 (Lima et al., 1979; Taiwo et al., 1982). One or two smaller proteins of M. Wt 2.9-3.1 x 103 and 2.7-2.9 x 103 were detected; these may be the result of proteolytic degradation, because preparations stored for more than 1 month contained only the smallest component.
Relations with Cells and Tissues
Cytoplasmic, cylindrical inclusions (pinwheels and scrolls: Fig. 5, Fig. 9) are detected in most tissues (Zettler et al., 1967; Edwardson et al., 1972; Edwardson, 1974; Lima et al., 1978; Lima & Purcifull, 1980; Murphy, 1984) of cowpea, crotalaria and other hosts. Nuclear inclusions have been reported in epidermal cells of crotalaria infected with some isolates (Zettler et al., 1967; Edwardson, 1974; Christie & Edwardson, 1977).
Notes
Numerous viruses infect and induce mosaic or mottle symptoms in cowpea. The presence of cytoplasmic inclusions typical of potyviruses often can be determined quickly by light microscopic examination of epidermal strips from infected plants (Christie & Edwardson, 1977). A Moroccan isolate of cowpea aphid-borne mosaic virus (Fischer & Lockhart, 1976) and blackeye cowpea mosaic virus induce similar types of cylindrical inclusion body in their hosts, but the former can be distinguished from blackeye cowpea mosaic virus by SDS-immunodiffusion tests (Lima et al., 1978; Lima et al., 1979; Taiwo & Gonsalves, 1982). Other viruses that infect cowpea naturally include cowpea mosaic, cowpea mottle, cowpea mild mottle, cowpea severe mosaic, cucumber mosaic, cowpea chlorotic mottle, southern bean mosaic, and tobamoviruses that infect legumes. Each of these viruses can be distinguished from blackeye cowpea mosaic virus by serological tests, cytology and particle morphology. Diseases in cowpea caused by blackeye cowpea mosaic virus probably can be controlled by the use of resistant varieties (Kuhn et al., 1966; Kuhn et al., 1984; Taiwo et al., 1981) and virus-free seed.
Figures

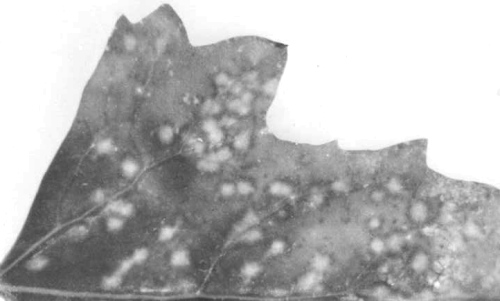
Mosaic and vein-banding symptoms in trifoliolate leaf of cowpea (Vigna unguiculata cv. Knuckle Purple Hull) seedling infected through the seed.

Vein banding mosaic on trifoliolate leaf of systemically infected cowpea cv. California Blackeye No. 5 infected by mechanical inoculation.

Photomicrograph of inclusion body (CI) consisting of mass of cylindrical inclusions in epidermal cell of infected cowpea leaf tissue. Nucleus (N). (Courtesy of R. G. Christie.) Bar represents 5 µm.

Serological detection by sodium dodecyl sulphate (SDS)-immunodiffusion tests. Antigen wells contain SDS-treated sap: B = blackeye cowpea mosaic virus-infected cowpea: C = cowpea aphid-borne mosaic virus (Moroccan isolate)-infected cowpea: H = healthy cowpea. The antiserum in wells A is to blackeye cowpea mosaic virus treated with pyrollidine (Lima et al., 1979). In Fig. 6, the cross-reacting antibodies were removed by intragel absorption with sap from plants infected with Morocco cowpea aphid-borne mosaic virus. In Fig. 7, the antiserum was absorbed by intragel absorption with sap from non-inoculated cowpea plants.

Serological detection by sodium dodecyl sulphate (SDS)-immunodiffusion tests. Antigen wells contain SDS-treated sap: B = blackeye cowpea mosaic virus-infected cowpea: C = cowpea aphid-borne mosaic virus (Moroccan isolate)-infected cowpea: H = healthy cowpea. The antiserum in wells A is to blackeye cowpea mosaic virus treated with pyrollidine (Lima et al., 1979). In Fig. 6, the cross-reacting antibodies were removed by intragel absorption with sap from plants infected with Morocco cowpea aphid-borne mosaic virus. In Fig. 7, the antiserum was absorbed by intragel absorption with sap from non-inoculated cowpea plants.
References list for DPV: Blackeye cowpea mosaic virus (305)
- Anderson, Pl. Dis. Reptr 39: 349, 1955.
- Bock, Ann. appl. Biol. 74: 75, 1973.
- Bock & Conti, CMI/AAB Descr. Pl. Viruses 134, 4 pp., 1974.
- Chang, Pl. Prot. Bull., Taiwan 25: 177, 1983.
- Christie & Edwardson, Monogr. Ser. Fla agric. Exp. Stn 9, 155 pp., 1977.
- Collins, Witcher & Barnett, Pl. Dis. 69: 18, 1985.
- Corbett, Rep. Fla agric. Exp. Stn, 1956: 117, 1956.
- Demski, Reddy & Sowell, Phytopathology 74: 627, 1984.
- Edwardson, Monogr. Ser. Fla agric. Exp. Stn 4, 398 pp., 1974.
- Edwardson, Zettler, Christie & Evans, J. gen. Virol. 15: 113, 1972.
- Fischer & Lockhart, Phytopath. Z. 85: 43, 1976.
- Hollings & Brunt, CMI/AAB Descr. Pl. Viruses 245, 7 pp., 1981.
- Kuhn, Phytopathology 54: 739, 1964.
- Kuhn, Brantley & Sowell, Pl. Dis. Reptr 49: 879, 1965.
- Kuhn, Brantley & Sowell, Bull. Ga agric. Exp. Stn 157, 22 pp., 1966.
- Kuhn, Brantley, Demski & Pio-Ribeiro, HortScience 19: 592, 1984.
- Lima, Ph.D. Diss., Univ. Fla, 154 pp., 1978.
- Lima & Purcifull, Phytopathology 70: 142, 1980.
- Lima, Purcifull & Edwardson, Abstr. 4th int. Congr. Virol., The Hague, Netherlands, p. 586, 1978.
- Lima, Purcifull & Hiebert, Phytopathology 69: 1252, 1979.
- Lin, Santos & Kitajima, Fitopatol. Bras. 6: 193, 1981.
- Lovisolo & Conti, Neth. J. Pl. Path. 72: 265, 1966.
- Mali & Kulthe, Pl. Dis. 64: 925, 1980.
- Matthews, Intervirology 17: 4, 1982.
- Murphy, M.S. Thesis, Clemson Univ., 84 pp., 1984.
- Murphy, Barnett & Witcher, Phytopathology 74: 631, 1984.
- Pio-Ribeiro, Wyatt & Kuhn, Phytopathology 68: 1260, 1978.
- Sekar & Sulochana, Ind. J. Pl. Path. 1: 38, 1983.
- Shepard, Secor & Purcifull, Virology 58: 464, 1974.
- Taiwo & Gonsalves. Phytopathology 72: 583, 1982.
- Taiwo, Provvidenti & Gonsalves, J. Hered. 72: 433, 1981.
- Taiwo, Gonsalves, Provvidenti & Thurston, Phytopathology 72: 590, 1982.
- Tsuchizaki, Senboku, Iwaki, Pholauporn, Srithongchi, Deema & Ong, Ann. phytopath. Soc. Japan 50: 461, 1984.
- Uyemoto, Provvidenti & Purcifull, Phytopathology 63: 208, 1973.
- Yeh & Gonsalves, Phytopathology 74: 1273, 1984.
- Zettler & Evans, Proc. Fla St. hort. Soc. 85: 99, 1972.
- Zettler, Christie & Edwardson, Virology 33: 549, 1967.